Over the past few years, the Food Safety Modernization Act (FSMA) and its resulting regulations and requirements have had an impact on how processors conduct their operations. In June/July 2017, at the onset of the changes spurred by FSMA, and as part of our Food Safety Insights program, we asked processors about their plans for FSMA compliance, how they saw the regulations changing their operations, how they were preparing for FDA inspections, and anything else that they saw that they would be facing.
Now that it’s 2019 and processors have had (up to) 2 years of experience with FSMA, we wanted to get an update on how they were dealing with the new rules, what changes they’ve made, what’s working and what’s not, and what they’ve learned about this new regulatory environment.
This is part one of a two-part report on a survey conducted by Strategic Consulting Inc. and Food Safety Magazine in January 2019 of more than 180 food processors from 40 U.S. states and 4 Canadian territories, and 60 processors from 36 other countries. We also asked officials at the U.S. Food and Drug Administration (FDA) for their input, comments, and viewpoints. Of the individuals who responded, 82 percent identified their position as quality assurance/quality control, food safety, plant operations, or sanitation specialists, with 18 percent in general management or regulatory/legal affairs or other related positions.
A Window into Compliance
We asked processors about their efforts to comply with FSMA, their experiences with FDA inspections (if they have had one), what their biggest issues and surprises have been, and any changes that they’ve made in their environmental monitoring and sanitation programs.
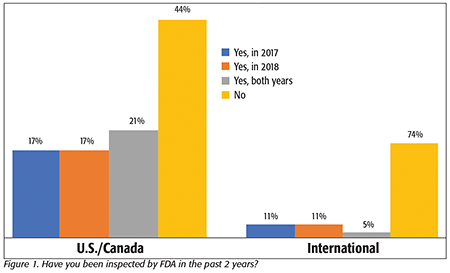 We first asked whether processors had received an FDA inspection in the past 2 years. For those in the U.S. and Canada (Figure 1), roughly one-third said that they have had one inspection in either 2017 or 2018, 21 percent said they were inspected in both years, and 44 percent reported that they have yet to receive a FSMA inspection. It is important to note that we also asked each survey respondent if they were an FDA-regulated facility, and only those FDA-regulated or “dual-regulated” facilities were included in the data. For those processors outside the U.S. and Canada (International in Figure 1), just over one in four had received an inspection, with the rest saying that they have not yet had one.
We first asked whether processors had received an FDA inspection in the past 2 years. For those in the U.S. and Canada (Figure 1), roughly one-third said that they have had one inspection in either 2017 or 2018, 21 percent said they were inspected in both years, and 44 percent reported that they have yet to receive a FSMA inspection. It is important to note that we also asked each survey respondent if they were an FDA-regulated facility, and only those FDA-regulated or “dual-regulated” facilities were included in the data. For those processors outside the U.S. and Canada (International in Figure 1), just over one in four had received an inspection, with the rest saying that they have not yet had one.
The data also showed that the likelihood that one had undergone a FSMA inspection was relatively consistent across different types of processors in the U.S. and Canada, with the key exceptions being that more respondents in the processed/prepared foods segment reported having had an inspection in both years than any other category, and more of those in the fruit/vegetable and grain segments reported that they have not yet had an inspection.
In 2017, we asked—ahead of the first FDA FSMA inspections—how processors anticipated that the agency might be changing its enforcement practices and specifically whether FDA would “educate before they regulate” as it was then claiming. At that time, only 40 percent of the respondents agreed that FDA was likely to educate first. Many of the comments we received at the time expressed views such as, “The inspectors were just in the same new FSMA classes as we were just in, so what can they know about the regulations to ‘educate us’ that we don’t already know?” and “What we do is specialized processing with specific unique requirements for our products. How can FDA inspectors, who, because they inspect so many processing plants of varying types, educate us in our processes that they cannot understand as well as we already do?”
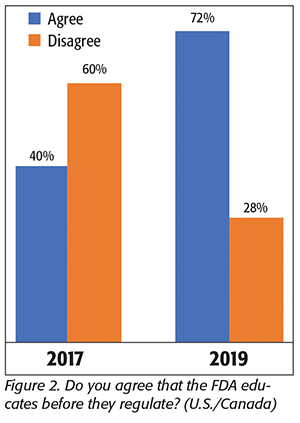 It’s now 2019, and, from what we found, something must be going right. In this survey, 72 percent of our respondents in the U.S. and Canada now said that they agree that FDA educates first (Figure 2). In 2017, many of the comments reflected processors’ apprehension about what exactly a “FSMA inspection” was going to be. This time, with more than one-half of the respondents having had experience with one or more inspections, many of the comments were far more positive about the FDA and its inspectors. One processor told us that the “local FDA inspectors in our state have been especially open with us to help with training and to discuss regulatory interpretations with us,” while another offered, “I did see that the FDA inspectors were more flexible in discussing the FSMA regulations rather than immediately looking to write up nonconformances. They also seemed to be trying harder to educate themselves as well.” One particularly positive comment that stood out was from a packaged vegetable processor who said, “We had a 3-day FDA audit and the auditor was professional and helpful, with no sense that she was there to ‘shut us down’ or find a problem. We were both well-prepared and had no findings, and the experience was one of our best FDA inspections to date.”
It’s now 2019, and, from what we found, something must be going right. In this survey, 72 percent of our respondents in the U.S. and Canada now said that they agree that FDA educates first (Figure 2). In 2017, many of the comments reflected processors’ apprehension about what exactly a “FSMA inspection” was going to be. This time, with more than one-half of the respondents having had experience with one or more inspections, many of the comments were far more positive about the FDA and its inspectors. One processor told us that the “local FDA inspectors in our state have been especially open with us to help with training and to discuss regulatory interpretations with us,” while another offered, “I did see that the FDA inspectors were more flexible in discussing the FSMA regulations rather than immediately looking to write up nonconformances. They also seemed to be trying harder to educate themselves as well.” One particularly positive comment that stood out was from a packaged vegetable processor who said, “We had a 3-day FDA audit and the auditor was professional and helpful, with no sense that she was there to ‘shut us down’ or find a problem. We were both well-prepared and had no findings, and the experience was one of our best FDA inspections to date.”
This is, of course, not to say there were no critical comments. One of the criticisms mentioned, which was a repeat of commentary we heard in our 2017 survey, was the inspectors’ lack of specific knowledge about the specific product and processing that was done in the processor’s plant. Many had similar comments about the preventive controls training that was available. One ingredient supplier mentioned, “…honestly, the training is generalized and sometimes hard to take back and implement within your facility and know that you are doing it correctly.” Another mentioned, “It would be good if (FDA inspectors) better understood where in our specific processes are our real risks, and where in our processes we do not have risks from a food safety perspective, and to put a lesser focus on documentation.”
Many of the comments were in regard to the performance of their local state agencies and the state officials who are doing these inspections on behalf of FDA. These comments were similar to those that we heard about FDA inspectors and, while they contained many critical comments, reflected the same general positive viewpoint as those for the federal agency.
I also had a chance to talk to one state official who said that they give a great deal of credit to FDA for recognizing that it did not have the network itself to get inspections done and fully implement FSMA requirements. The agency’s decision to turn to the state agencies greatly expanded FDA’s available resources and its ability to get the work done. This official added that they have been able to cooperate with their state university’s extension service and use trainers from the university, as well as the ones in their own department, to get the education component involved very early in the process. He added that they were then able to use their own network and inspectors to implement more uniform inspections and compliance throughout the state, and because of this cooperative arrangement, it was his assessment that they had been able to do an effective job implementing FSMA and believes that they are in pretty good shape in his state.
We should also mention that 97 percent of the international respondents (i.e., outside the U.S. and Canada) agreed that FDA first educates before regulating; however, fewer than 30 percent reported having had an inspection. Most of the comments from these processors focused on the great need for additional training to help them comply with FSMA, and many mentioned that they “looked forward” to getting that training. Others mentioned that they were aware that FDA was working with their national agencies, health ministries, and other groups organizing training for FSMA compliance. It was clear, however, that their comments were mainly anticipatory and that few had yet had any formal FSMA compliance training.
Processors List Sticking Points
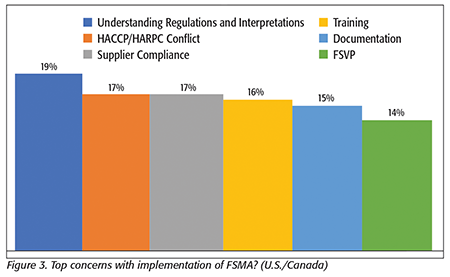 It was evident from our survey data and interviews that there are some “growing pains” with FSMA compliance. When we asked, “What are your biggest concerns with the implementation of FSMA?” the most frequent answer(s) (Figure 3) was/were related to a better understanding of the regulations and, particularly, interpretations of inspectors’ decisions and resolving conflicts with other standards. Many people mentioned conflicts their existing Hazard Analysis and Critical Control Points (HACCP) plan had with the Hazard Analysis and Risk-Based Preventive Controls (HARPC) plan that they had to create. Others mentioned that they need to maintain their HACCP plan to comply with Safe Quality Food (SQF) requirements and how this creates process and plan conflicts that they then need to resolve between SQF auditors and FDA inspectors.
It was evident from our survey data and interviews that there are some “growing pains” with FSMA compliance. When we asked, “What are your biggest concerns with the implementation of FSMA?” the most frequent answer(s) (Figure 3) was/were related to a better understanding of the regulations and, particularly, interpretations of inspectors’ decisions and resolving conflicts with other standards. Many people mentioned conflicts their existing Hazard Analysis and Critical Control Points (HACCP) plan had with the Hazard Analysis and Risk-Based Preventive Controls (HARPC) plan that they had to create. Others mentioned that they need to maintain their HACCP plan to comply with Safe Quality Food (SQF) requirements and how this creates process and plan conflicts that they then need to resolve between SQF auditors and FDA inspectors.
These issues were closely followed by others related to suppliers, including getting suppliers to comply with requirements and getting the proper documentation from those suppliers, with many respondents specifically mentioning having difficulties with the Foreign Supplier Verification Program (FSVP).
Training also showed up high on the list—at fourth with 16 percent of all respondents indicating it as an issue. But it was also clear from their comments that this was not solely due to training materials and programs, but this was also a reference to “internal execution” and specifically “…getting people to do it that way they were trained.” Many factors about the clarity, quality, and availability of the training were mentioned, although another frequently mentioned factor was related to the processors’ own efforts to keep employees properly trained and compliant while facing high levels of employee turnover.
In part two, we will continue to present the findings from this survey and discuss more data about how processors have been modifying their environmental monitoring and sanitation programs to better comply with FSMA. We will also present data from the survey on how frequently FDA is collecting environmental samples.
We will also hear directly from FDA and get their responses to our findings. We asked what processors wished FDA understood better. Not only will we discuss the data from the responses to that question, but we also (anonymously!) showed those responses to FDA and asked for their response as well as what FDA would ask that processors understood better.
I want to close by mentioning that we will be attending the Food Safety Summit in Rosemont, Illinois, in early May, and we will be discussing the findings of this survey at the Food Safety Magazine booth. We would like to hear your responses to these data and talk with you about your own experiences with FSMA, and we hope to add those responses in our next column.
See you in Rosemont!
Bob Ferguson is president of Strategic Consulting Inc. Bob can be reached at insights@food-safety.com or on Twitter at @SCI_Ferguson.
What Industry and FDA Are Thinking about FSMA Implementation – Part 1




