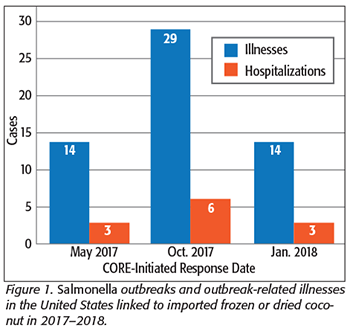
Prior to 2000, illness outbreaks linked to the consumption of coconuts and coconut products around the world had been documented, including, for example, those of cholera and paratyphoid A fever illnesses in the U.S. and Singapore, respectively.[1,2] In 1999, an outbreak of Salmonella Java phage type Dundee infections in the United Kingdom was linked to desiccated coconut.[3] However, according to the U.S. Centers for Disease Control and Prevention (CDC), between 2000 and 2017, there hadn’t been any U.S. multistate outbreaks identified linked to coconut or coconut products. That changed beginning in 2017, when U.S. officials identified three outbreaks, totaling 57 cases, in the span of 8 months (Figure 1). The outbreaks were linked to imported processed coconut consumed as a ready-to-eat food. These outbreaks offer an opportunity to highlight the challenges faced when trying to identify and remove the product from the market, as well as make recommendations for the safe processing, distribution, and consumption of imported frozen and dried coconut.
 The U.S. Food and Drug Administration (FDA)’s Office of Coordinated Outbreak Response and Evaluation (CORE), in College Park, MD, works closely with CDC to coordinate federal resources when responding to outbreaks of foodborne illnesses.[4] FDA CORE, CDC, state, and local partners use several investigative tools to identify and confirm the source of an outbreak.[5] Among these are interviews with sick people to identify possible outbreak sources, and product and environmental sampling and testing, including whole-genome sequencing (WGS) of bacterial isolates. WGS is a highly precise form of DNA analysis that can compare microbial isolates from a food with clinical isolates from patients with a foodborne illness to determine how closely they are genetically related and establish their common source.
The U.S. Food and Drug Administration (FDA)’s Office of Coordinated Outbreak Response and Evaluation (CORE), in College Park, MD, works closely with CDC to coordinate federal resources when responding to outbreaks of foodborne illnesses.[4] FDA CORE, CDC, state, and local partners use several investigative tools to identify and confirm the source of an outbreak.[5] Among these are interviews with sick people to identify possible outbreak sources, and product and environmental sampling and testing, including whole-genome sequencing (WGS) of bacterial isolates. WGS is a highly precise form of DNA analysis that can compare microbial isolates from a food with clinical isolates from patients with a foodborne illness to determine how closely they are genetically related and establish their common source.
Another important investigative tool is the traceback investigation, which is used to track shipments of food from specific retail locations back to their origins. Once a product is implicated as the source of an outbreak, FDA has several regulatory tools to remove the product from the market, such as a product recall. Many businesses may agree to a recall voluntarily, but, depending on the circumstances, FDA may consider taking other appropriate actions, such as a mandatory recall. FDA also may suspend the registration of a food facility. Registration with FDA is required for any facility that manufactures, processes, packs, or holds food for consumption in the United States. If a facility’s registration is suspended, then food from that facility cannot be imported or exported, or offered for import or export, into the U.S., or otherwise introduced into commerce in the U.S.
FDA also has tools to help prevent potentially contaminated food from entering the market in the first place, such as increased port-of-entry screening and Import Alerts.[6] For imported food, most notably, an Import Alert provides information to FDA field staff and the public that the agency has enough evidence to allow for detention without physical examination of products that appear to be in violation of the Federal Food, Drug, and Cosmetic Act of 1938. A company may provide evidence to FDA in an attempt to overcome the appearance of the violation, but if the information is not sufficient, the product is subject to refused entry into the United States.
Epidemiology, Traceback, and International Collaboration: Precut Frozen Coconut
The first in this string of coconut-related outbreaks occurred in mid-2017. On May 19, 2017, FDA’s CORE was alerted by CDC that precut coconut was the suspected source of a group of Salmonella Chailey illnesses in the U.S.[7] The response to this multistate outbreak of Salmonella Chailey involved a partnership between FDA, CDC, local, and state public health officials in the U.S. and Canada. Independently, the epidemiological investigations in each country were hampered by the limited number of cases. However, once combined, a strong epidemiological signal implicated precut coconut purchased from a national retail food market. Nineteen cases were included in the outbreak: 14 in the U.S. and 5 cases in Canada. There were three hospitalizations, and no deaths were reported.
The precut frozen raw coconut meat was imported in packages that could hold 50 pounds or more and sent to a storage facility. The product was then repackaged into bags of just a few pounds, vacuum-sealed, and labeled with a “use-by” date of 12 months, with instructions to “keep frozen, upon thawing, refrigerate, and use within seven days.” This repackaged coconut meat was then sent to distribution centers for a U.S.-based national retail chain that also had stores in Canada. At these retail stores, the frozen coconut packages were thawed, repackaged into small clear plastic containers, and kept refrigerated, maintaining a 5-day turnaround.
FDA conducted a traceback investigation based on the available information. This investigation revealed that this product was received from a manufacturer in Indonesia through a U.S. importer. The partners in the investigation confirmed the outbreak source. They based the conclusion on strong epidemiological evidence and the identification, through traceback, of a specific lot of coconut most likely eaten by case patients. As a result, FDA increased sample collection and microbiological testing for Salmonella of imported frozen coconut chunks shipped by this manufacturer to help identify and prevent additional contaminated product from entering the country.
The Synergy of the CORE Network: Frozen, Shredded Coconut in Drinks
The second multistate outbreak investigation began in the fall of 2017. On October 3, 2017, CDC notified FDA of a group of Salmonella illnesses.
By the end of the outbreak, a total of 29 cases in nine states were identified, while the pathogens responsible for the illnesses were Salmonella I 4,[5],12:b:- and Salmonella Newport.
CDC, state, and local public health partners identified drinks purchased from various locations of a nationwide restaurant franchise specializing in Vietnamese drinks and desserts as a common exposure reported among cases. Although the identification of a common vehicle of exposure was a break in the investigation, it led to a major challenge. Reported drink ingredients included red beans, mung beans, tapioca pearls, taro, jellies, and coconut milk. Investigators then had to identify which ingredient was causing the illnesses. A traceback investigation was required to determine the path of these ingredients from the store locations to their source and determine whether there was a common source for any ingredient.
Federal and state partners conducted a traceback investigation for multiple drink ingredients from six retail locations in five states and found that each of the commodities had been imported. Upon closer review of the exposures, coconut milk was the only common ingredient in the drinks that had been described by seven case patients.
Through a review of records, FDA determined that the firms had received frozen shredded coconut exclusively from one importer, and this importer had received frozen shredded coconut exclusively from a supplier in Vietnam. The importer’s representatives reported that the coconut brand was manufactured and packaged exclusively for customers in the U.S. The traceback investigation did not implicate specific lots of frozen shredded coconut due to the lack of lot identifiers on distribution records/invoices or individual packages. Nevertheless, records collected from each point in the supply chain documented the distribution of this specific brand of frozen shredded coconut to retail locations associated with illnesses. Then, FDA increased surveillance and laboratory analysis for Salmonella at the border on this supplier’s shipments of shredded coconut.
State partners collected food samples that were critical to the investigation. One sample of prepared coconut milk was collected by New York state investigators from a New York retail location of interest, and it tested positive for Salmonella Newport. The isolate was then analyzed through WGS. The sample was closely related to the Salmonella isolated from an ill person from Massachusetts.
Massachusetts investigators discovered that the ill person had had an Asian-style dessert drink at a restaurant, so they collected multiple unopened samples of a brand of frozen shredded coconut from the restaurant for testing. The frozen shredded coconut tested positive for Salmonella, and although the samples did not match any cases in this outbreak by WGS analysis, Massachusetts state officials embargoed all remaining frozen shredded coconut at that restaurant location.
Later in the investigation, Massachusetts investigators returned to the restaurant and collected additional samples of frozen shredded coconut. They identified various strains of Salmonella, including the main outbreak strain, Salmonella I 4,[5],12:b:-. On January 2, 2018, the importer announced a recall of its frozen shredded coconut,[8] and shortly thereafter, the supplier was added to an Import Alert identifying all their frozen coconut products,[9] while FDA, CDC, and Massachusetts state partners issued press releases to inform and protect the public.
The Impact of WGS: Bulk Dried Coconut
Before the second outbreak investigation was resolved, another was just beginning. On January 23, 2018, after receiving notification from CDC, CORE began evaluating an outbreak of Salmonella Typhimurium illnesses from six states. Ill people commonly reported eating bulk food products including almonds, dried flaked coconut, dried fruit, and granola/rolled oats.
By February 2018, CORE, in collaboration with federal, state, and local officials, initiated a traceback investigation of multiple bulk food products sold at grocery store locations associated with Salmonella illnesses in California, Colorado, Connecticut, Idaho, Oregon, and Texas. The traceback included two grocery chains. These chains received product from two importers. The traceback investigation documented the distribution of the implicated dried coconut to the retail locations associated with illnesses. However, due to the lack of lot identifiers on distribution records, the investigation could not implicate specific lots of dried coconut. Analysis of records provided by the firms identified the likely source of the outbreak as a dried coconut supplier from Sri Lanka.
On February 9, 2018, FDA collected an open consumer sample of dried coconut from a case patient’s home. The product was purchased from a retail store supplied by one of the importers identified in the traceback investigation.
Testing identified Salmonella Typhimurium in the sample, and WGS analysis showed that the bacteria from the sample were closely related genetically to the outbreak strain. An additional sample, collected from a case patient’s home by state partners, also yielded the outbreak strain.
Meanwhile, FDA collected samples of dried coconut from one distribution center and one importer. These were positive for Salmonella Typhimurium, Salmonella Weltevreden, and Salmonella Muenster. WGS analysis performed by FDA and CDC showed that the Salmonella Typhimurium bacteria from ill people and the dried coconut samples were all highly related to each other. By March 15, 2018, FDA increased laboratory testing of imported dried coconut from two manufacturers of interest for Salmonella. One manufacturer was placed on an Import Alert.[10] The importer initiated a voluntary recall of their dried coconut product,[11] and the distribution center also initiated a recall of dried coconut.[12]
Lessons Learned
Overcoming Outbreak Investigation Challenges
Each of the three outbreaks presented unique challenges, but collaborative efforts using investigative tools solved the puzzle. In the first outbreak investigation, the importance of strong epidemiological data, which informed the traceback investigation, was highlighted through the international collaboration between the U.S. and Canadian partners. Independently, the epidemiological investigations in each country lacked strength due to the limited number of cases, but when combined, the data informed stronger traceback investigations with solid conclusions.
In the second outbreak investigation, the exposure to dessert drinks with multiple shared ingredients made it difficult to identify the exact source ingredient responsible for the outbreak. However, coordination by federal, state, and local partners allowed for rapid and effective decision making to solve the outbreak. This allowed for an extensive traceback investigation and ultimately targeted sampling and analysis of the suspected ingredients. The results were quickly shared with all involved partners and allowed for timely initiation of a voluntary recall, an Import Alert, and consumer notifications to protect public health.
In the third outbreak investigation, there were, again, multiple food products considered to be suspect vehicles. The epidemiological information and traceback investigation were instrumental in finding the origin of the vehicle, but WGS was a tool that enabled the conclusive identification of the coconut as the confirmed vehicle of the outbreak.
How Can Risks Be Mitigated during the Production of Frozen and Dried Coconut?
There are many possible sources and routes of contamination in the production of processed coconut products, both on farms and in facilities. The growing, harvesting, packing, and holding of coconut is covered by the FDA Food Safety Modernization Act (FSMA)’s Produce Safety rule, if produced domestically or for import into the United States. This rule establishes science-based minimum standards for the safe growing, harvesting, packing, and holding of fruits and vegetables grown for human consumption.
During processing, coconuts may be potentially contaminated from equipment that has not been adequately maintained, cleaned, and sanitized.[13] Contamination may also occur during and after splitting, and during freezing or drying in areas that may not be properly cleaned and protected.
Facilities that process coconut generally are covered by the Preventive Controls for Human Food rule if the coconut is produced domestically or for import into the United States. This rule requires food facilities to have a food safety plan in place (unless they are exempt from Subpart C of the regulation) that includes an analysis of hazards and risk-based preventive controls to minimize or prevent the identified hazards.
What Is FDA Doing about Imported Frozen and Dried Coconut?
To help focus and speed investigations into foodborne illness outbreaks, FDA is spearheading an international effort to build a network of laboratories that can sequence the genomes of foodborne pathogens and then upload the genomic sequence of the pathogen and the geographic location from which it was gathered into a publicly accessible database. The database continues to grow, increasing the ability to rapidly identify potential contamination sources in future outbreak investigations.
Specific to the lessons learned from the back-to-back-to-back outbreak investigations, CORE continues to analyze trends in outbreak sources and monitor for additional outbreaks linked to coconut and coconut products. Furthermore, CORE evaluates available outbreak data to provide support for future regulatory policy discussions and follows up on sampling results yielded by any increased screening at ports of entry.
However, consistent with FSMA, requiring an importer to have adequate verification procedures is a critical tool in managing the quality of all FDA-regulated products coming into the U.S. FDA conducted outreach to industry, consumer groups, the agency’s federal, state, local, tribal, and international regulatory counterparts, academia, and other stakeholders in developing the Foreign Supplier Verification Programs for Importers of Food for Humans and Animals rule. This rule requires that importers perform certain risk-based activities to verify that food imported into the U.S. has been produced in a manner that meets applicable U.S. safety standards.
Under the rule, importers are responsible for actions that include:
• Determining known or reasonably foreseeable hazards with each food
• Evaluating the risk posed by a food, based on the hazard analysis, and the foreign supplier’s performance
• Using the evaluation of the risk posed by an imported food and the supplier’s performance to approve suppliers and determine appropriate supplier verification activities
• Conducting supplier verification activities; importers have the flexibility to tailor supplier verification activities to unique food risks and supplier characteristics that include:
• Annual on-site audits of the supplier’s facility
• Sampling and testing
• A review of the supplier’s relevant food safety records
• Conducting corrective actions
The risk posed by the imported food and the supplier’s performance must be reevaluated at least every 3 years, or when new information comes to light about a potential hazard or the foreign supplier’s performance. Certainly, an outbreak linked to a supplier is cause for an importer to implement corrective and preventive actions to prevent future outbreaks. The appropriate corrective measure will depend on the circumstances but could include discontinuing use of the foreign supplier until the cause of noncompliance, adulteration, or misbranding has been adequately addressed.
Following these outbreaks linked to frozen or dried coconut from international suppliers, improvements are necessary in the monitoring of procedures and processes conducted by the supplier to ensure the safety of this product. FDA will continue to work to ensure that this process is followed to help prevent future outbreaks of this type.
Donald C. Obenhuber, Ph.D., is a member of FDA’s CORE Network.
References
1. www.cdc.gov/mmwr/preview/mmwrhtml/00015726.htm.
2. www.ncbi.nlm.nih.gov/pubmed/9494656.
3. www.eurosurveillance.org/content/10.2807/esw.03.12.01435-en.
4. www.fda.gov/food/outbreaks-foodborne-illness/about-core-network.
5. www.cdc.gov/foodsafety/outbreaks/pdfs/outbreak-infographic.pdf.
6. www.fda.gov/industry/actions-enforcement/import-alerts.
7. www.cdc.gov/mmwr/volumes/67/wr/mm6739a5.htm.
8. www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/evershing-
international-trading-company-recalls-frozen-shredded-coconut-because-possible-health-risk.
9. www.accessdata.fda.gov/cms_ia/importalert_71.html.
10. www.accessdata.fda.gov/cms_ia/importalert_1128.html.
11. www.fda.gov/Safety/Recalls/ucm601529.htm.
12. www.fda.gov/Safety/Recalls/ucm601687.htm.
13. www.foodstandards.gov.au/consumer/importedfoods/Documents/Dried coconut and Salmonella.pdf.
Back-to-Back Outbreaks in Frozen and Dried Imported Coconut