There is no question that implementation of Food Safety Modernization Act (FSMA) regulations will have a significant impact on food processors. This is part one of a two-part report on a survey conducted by Strategic Consulting Inc. and Food Safety Magazine of more than 100 food processors throughout the U.S. We asked about FSMA compliance, relationships with the U.S. Food and Drug Administration (FDA), environmental monitoring and sanitation practices and how they may be changing.
Our survey included companies from 32 states and 4 territories, and consisted of an online questionnaire followed by a series of telephone interviews. Of the individuals who responded, 74% identified their position as quality assurance/control, food safety or sanitation specialists, with 8% identifying as a laboratory manager. The remainder were in general management, regulatory or related positions.
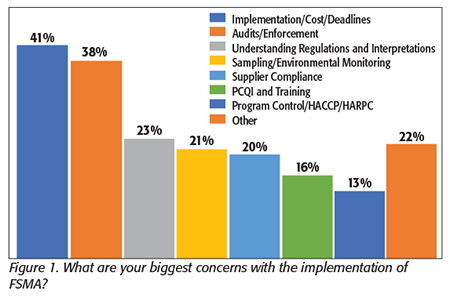 We asked what their biggest concerns were with the implementation of FSMA. The most common answer was implementation: what it will cost and how to get the resources required to get it done right and by the deadline (Figure 1). This was followed by uncertainty about inspections and enforcement, and regulatory interpretation.
We asked what their biggest concerns were with the implementation of FSMA. The most common answer was implementation: what it will cost and how to get the resources required to get it done right and by the deadline (Figure 1). This was followed by uncertainty about inspections and enforcement, and regulatory interpretation.
We explored this topic in telephone interviews, and it became clear that these top three concerns were really one major concern stated in different ways.
Concerns about Getting It Right
Processors were generally satisfied with the food safety program they had in place prior to FSMA. Because of this, most reported that the main part of their work to comply with FSMA was about writing the plans rather than instituting fundamental changes. All had confidence in their knowledge of food safety and how to produce safe products but were concerned about getting FSMA plans in place and on time with limited resources and no reduction in their daily demand (“...changing the tires on the car while still going 60,” according to one comment). But many also said that they will not know if they got it right until they have an inspection.
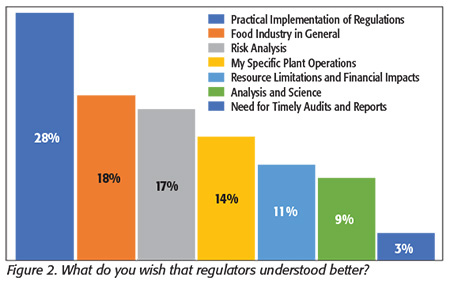 We asked, “What do you wish that regulators understood better?” The most common answer was practical regulatory interpretation (Figure 2). Many responded that regulators want companies to follow the letter of the law, but there are the practical considerations in a food processing facility. One respondent mentioned, “It is impossible to do only desk work and review policy and still have something that can be implemented in a food processing facility.” The second- and fourth-most common answers were similar in that they wished that regulators understood the food processing business in general and their specific type of processing, respectively. Many commented that regulators should have a better understanding of food processes so they can have more flexibility in inspections. Others thought there was not enough focus on risk. Several mentioned the lack of attention to the consumer’s responsibility to follow final preparation instructions, while others cited too much attention to highly unlikely risks, with one adding, “…just because regulators can imagine an unlikely-to-occur problem shouldn’t mean we have to have a program to prevent it.”
We asked, “What do you wish that regulators understood better?” The most common answer was practical regulatory interpretation (Figure 2). Many responded that regulators want companies to follow the letter of the law, but there are the practical considerations in a food processing facility. One respondent mentioned, “It is impossible to do only desk work and review policy and still have something that can be implemented in a food processing facility.” The second- and fourth-most common answers were similar in that they wished that regulators understood the food processing business in general and their specific type of processing, respectively. Many commented that regulators should have a better understanding of food processes so they can have more flexibility in inspections. Others thought there was not enough focus on risk. Several mentioned the lack of attention to the consumer’s responsibility to follow final preparation instructions, while others cited too much attention to highly unlikely risks, with one adding, “…just because regulators can imagine an unlikely-to-occur problem shouldn’t mean we have to have a program to prevent it.”
Questions about Inspections
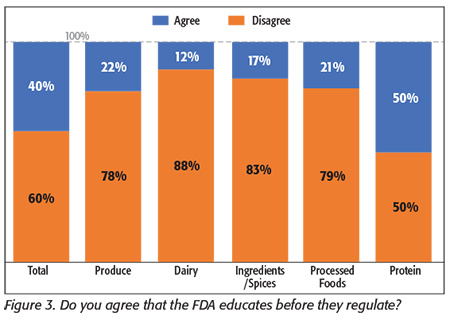 Processors indicated they are also unsure about how FDA will conduct its inspections. FDA has said that it plans to “educate before and while it regulates,” yet when we asked processors, most disagreed (Figure 3). Several respondents offered that FDA auditors were in the same FSMA training classes as they were and probably didn’t have any better information and thus doubted their ability to “educate.” Several mentioned that FDA inspectors who audit their facilities do not have enough specific experience in food production to offer any valuable assistance for their operations. One respondent commented that new inspectors must be frequently trained on what to look for and “once we train them, we get rewarded with a 483.” [An FDA form 483 is issued at the conclusion of an inspection when an investigator has observed conditions that may constitute violations of the Federal Food, Drug, and Cosmetic Act and related acts.] Other respondents expressed that since inspectors are “in a learning process,” this may lead to inconsistent inspections and enforcement.
Processors indicated they are also unsure about how FDA will conduct its inspections. FDA has said that it plans to “educate before and while it regulates,” yet when we asked processors, most disagreed (Figure 3). Several respondents offered that FDA auditors were in the same FSMA training classes as they were and probably didn’t have any better information and thus doubted their ability to “educate.” Several mentioned that FDA inspectors who audit their facilities do not have enough specific experience in food production to offer any valuable assistance for their operations. One respondent commented that new inspectors must be frequently trained on what to look for and “once we train them, we get rewarded with a 483.” [An FDA form 483 is issued at the conclusion of an inspection when an investigator has observed conditions that may constitute violations of the Federal Food, Drug, and Cosmetic Act and related acts.] Other respondents expressed that since inspectors are “in a learning process,” this may lead to inconsistent inspections and enforcement.
“This is not surprising,” according to David Acheson, former FDA associate commissioner for food and current CEO of The Acheson Group, one of the consulting companies that respondents are relying on for advice and assistance in their compliance issues. “FDA has not traditionally had a budget for education. Their effectiveness has typically been viewed by Congress based on the numbers of inspections they complete and the citations that they issue, not by more difficult-to-measure performance metrics like ‘education.’ FDA is working hard to change, but culture change is usually an uphill battle. This will be a slow ramp-up.”
The comments were not all negative, however, and several respondents said that inspectors with whom they work are good, understand the regulations, work to understand what the processor is doing and try to offer tips for better food safety during the audits.
Many wanted to know what FDA will focus on during early FSMA inspections. Processors understand that all requirements of FSMA must be met, and none expect FDA to let them “off the hook,” but they want to know if FDA will have specific areas of concern. One former FDA official said it is the agency’s goal to ensure compliance, and that FDA will be looking to see that processors have properly identified their hazards, have the correct preventive controls and scientific validation of the efficacy of those controls and proper documentation. But this official added that FDA clearly has a risk mindset, and the first inspections will be focused on processors with higher risks. Those with previous 483s, recalls and other risks should expect to be near the front of the line when inspections are scheduled.
In the next issue, we will present more data from the survey and report on changes being made to environmental monitoring programs, the use of whole-genome sequencing and changes to sanitation programs in anticipation of FSMA compliance.
Bob Ferguson is the managing director of Strategic Consulting Inc.




