In our June/July article, we discussed the details of trends in food safety testing in depth. One of the areas mentioned was the ongoing focus on whole-genome sequencing (WGS). In this edition of Food Safety Insights, we want to take a closer look at the use of WGS.
Few people question the impact that WGS has had on the food safety landscape over the past few years. The increasing use of WGS by regulators to investigate and track the source of outbreaks, and especially the unique ability of WGS (in conjunction with epidemiological and food traceback data) to connect the source of foodborne illnesses with causative bacteria isolated from patients, has greatly changed the regulators’ ability to identify and solve outbreaks, and trace them to their source. This use of the technology has greatly increased the regulators’ ability to recognize what may have previously been seen as numerous, small, and unrelated outbreaks (if detected and recognized at all) as what is truly a larger and related outbreak, only scattered across a broader geography. Recognition of these outbreaks is a factor in the increase in outbreaks reported during the past few years.
Understanding that regulators will be collecting samples during inspections and using WGS to analyze those samples, many are recommending that processors also collect and use WGS to help identify and eradicate harborage organisms from their plants, not only in the interest of enhanced food safety but also as a defense measure so organisms are not there to be found, identified, and added to a database by regulators and also as a check against the regulators’ own data.
So, is this testing occurring? Are processors widely using WGS on their own samples?
In a word, no.
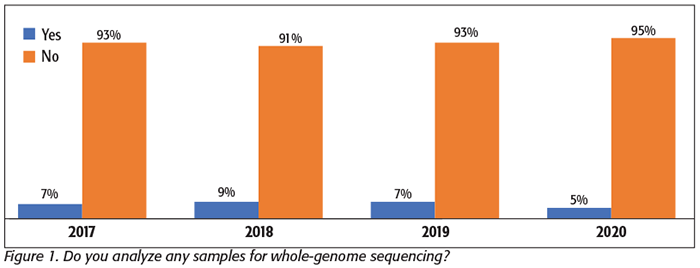 We have asked this question at least once each year since 2017, including this year’s survey in April 2020. In those 4 years, we have received data from more than 600 processors (an average of more than 150 responses per year). In each of these surveys, no more than 10 percent of respondents reported using WGS—even once—for any of their samples (Figure 1). In our most recent survey, only 5 percent reported that they are using WGS in any capacity.
We have asked this question at least once each year since 2017, including this year’s survey in April 2020. In those 4 years, we have received data from more than 600 processors (an average of more than 150 responses per year). In each of these surveys, no more than 10 percent of respondents reported using WGS—even once—for any of their samples (Figure 1). In our most recent survey, only 5 percent reported that they are using WGS in any capacity.
In the most recent survey, when we asked those using WGS how many samples per year they had submitted for sequencing, only a few reported that they sent out as many as 10 samples in the past year, with most reporting a far smaller number. No one in any of our surveys reported analyzing more than 25 samples on an annual basis. This finding confirms our previous reporting that processors are still generally not using WGS, and even those few who are submitting samples for WGS are using the technology very sparingly.
When asked about sample volume, most processors tell us that they “wanted to try” WGS to see how it might help them. But once they had the data back from the lab, 1) they thought that the data did not give them actionable information, 2) the data did not seem to be relevant to their ability to solve a problem, or 3) they were not able to effectively interpret the data.
One of the midsize processors we spoke with this year summarized their view of WGS by saying, “The technology is technologically interesting from an academic standpoint—and it certainly piques my curiosity—but it really has no practical applications in my operation. I understand the application of the technology in outbreak investigations, but I do not need to know the genome of any pathogens in my plant—what I really want is for all of them to be out of my plant or dead.”
This is a good summary of many of the comments we heard from processors.
Another processor we spoke with, who was in the very large, multinational-processor category, echoed this opinion. “We used WGS at another plant. We found the results to be not very accurate—the results of comparable samples collected at different times did not match. This made it difficult for the technology to be usable in our applications.” This processor continued, “What we really need is speed, not higher resolution. Our micro results control whether product moves from one step to another, so the faster we get the data, the better we can operate. I understand that WGS can be effective in environmental monitoring and identification of harborage organisms, but we have not had that issue, and the trials we ran with WGS were not very useful.”
Other answers we’ve heard when asking processors about their use of WGS included regulatory concerns: “Do I have to disclose the data during an inspection?” “How long do I need to retain the data?” “What will the U.S. Food and Drug Administration do with any data that I give them?” Other answers involved the long turnaround time needed for WGS, which limits its operational applicability, WGS being too complicated (e.g., presenting data that are difficult to interpret or impractical for operational decisions), and its high cost. Many processors also questioned the need to switch to a high-resolution technology like WGS when they have not had a pathogen problem in the past.
To find out more about the market response, we also spoke with an expert in sequencing technologies and in the application of WGS. In discussing the comments from our survey, he said, “I understand the comments from processors about WGS. Applications of WGS specifically as a technology to sequence the entire genome of isolates are probably not going anywhere other than its use for regulatory purposes and for outbreak investigations.”
What does this mean for sequencing technologies? Is there no use for processors for this powerful technology? Our expert offered a perspective.
“WGS is a subset of the larger category of next-generation sequencing (NGS), and many other applications of NGS can be more applicable, more versatile, and faster than full sequencing using WGS.” Added our expert, “By selecting the right applications of NGS, especially a number of applications involving shorter reads (sequencing involving fewer base pairs—typically reading target sequences typically in the range of a few hundred base pairs), the technology can have a total cost, throughput, and time-to-result to rival PCR.”
These other applications of NGS can be used to target and ID specific organisms by targeting only certain sequences and avoiding the time-consuming process of fully sequencing the entire genome of an organism. NGS can be effective at detection and identification by looking at specific characteristic sequences and can be used for tasks such as serotyping, strain typing, and 16S analysis. In higher-throughput labs, especially those with sufficient test volumes to amortize the capital costs of the sequencing instruments, plus a staff with the training and expertise to use the technology, this application of NGS may even be competitive with the use of PCR.
Assuming this is indeed the case and that these benefits can be realized, the applications of NGS would still probably be mostly limited to those larger food companies that maintain internal labs, probably specifically those that maintain a sophisticated central lab, or to commercial labs.
A move to faster and less-expensive applications may be an avenue for growth for sequencing in food safety testing. We would estimate, however, that without a breakthrough in speed, cost, and more usable data, the use of WGS and other applications of sequencing by food processors will quite likely remain infrequent and mostly take place at very large processors and commercial labs.
Bob Ferguson is president of Strategic Consulting Inc. and can be reached at foodsafetyinsights@gmail.com or on Twitter at @SCI_Ferguson.
Adoption of WGS: What Is Going On?




