Consumer demand for responsible antibiotic use in food animal production is driving market change, and both industry and large institutional purchasers are responding. Meat and poultry products raised under policies that restrict or eliminate antibiotic use are increasingly appearing on local grocery store shelves, in popular fast-food chains and in the cafeterias of large institutions around the country, including school districts and hospital systems.
Numerous surveys have demonstrated this growing consumer preference: A 2014 poll conducted by Midan Marketing found that more than 80 percent of Americans are aware of the widespread use of antibiotics in animals, and 60 percent are concerned.[1] In the same poll, 17 percent of respondents said they stopped buying meat when they thought antibiotics (or growth hormones) were used in production. Likewise, a 2012 Consumer Reports survey[2] found that 86 percent of consumers thought that meat raised without antibiotics should be available in their local supermarket, with nearly 40 percent saying they would be willing to pay an additional $1 per pound for it.
As this demand continues to grow, it is important to have a range of antibiotic-use standards that are transparent and meaningful, and that give producers options—enabling them to take substantial action to reduce the use of antibiotics in a way that best fits their operations while ensuring that their antibiotic-use policies are ones that consumers can trust.
Public Health Threat
The use of antibiotics in both human healthcare and the production of livestock leads to the emergence of resistant pathogens, a growing and serious public health threat. The World Health Organization has declared that antibiotic resistance is “a global crisis,”[3] and according to the U.S. Centers for Disease Control and Prevention, at least 23,000 Americans die annually as a direct result of drug-resistant infections while 2 million more contract infections.[4]
Minimizing antibiotic use—including reducing inappropriate antibiotic use in human medicine and in food animals—is key to slowing the emergence of drug-resistant bacteria and preserving these lifesaving drugs. In the U.S. alone, more than 20 million pounds of medically important antibiotics (i.e., those critical to human as well as veterinary medicine), accounting for 62 percent of all antibiotics sold for use in food-producing animals, were sold for use in food animal production in 2014—a 23 percent rise since 2009.[5]
Market-Driven Approaches for Reducing Antibiotic Use in Food Animals
To respond to consumer demand, food producers have developed a range of antibiotic-use policies. Some of these restrict the use of medically important antibiotics in a meaningful way; others do not.
Verification of producer conformance to these policies varies substantially, ranging from authentication by U.S. Department of Agriculture (USDA) staff via on-farm audits to self-certification by producers of their drug-usage claims. Standards verified by independent third parties such as USDA provide greater accountability and transparency to the process than self-enforced policies.
Importantly, such policies are not static, as existing standards may be revised and new ones established. In addition to long-standing organic standards and the absolute prohibition of antibiotic use, producers and advocates are proposing new standards that allow for the responsible use of antibiotics to protect animal health while still greatly reducing overall use in food animal production. Below are comparisons of several standards now verified by USDA:
Standards available for all food producers:
Organic:[6] No use of antibiotics, beginning in the third trimester of gestation for mammals and the second day of life for poultry.
No Antibiotics Ever (NAE):[7] No antibiotics used during the entirety of the food animal’s life.
No Medically Important Antibiotics:[7] No use of antibiotics deemed important by the World Health Organization for therapeutic use in humans; however, antibiotics with no relevance to human medicine, such as ionophores, can be used.
Standards available for food producers marketing to institutional purchasers only:
Certified Responsible Antibiotic Use (CRAU):[8] Medically important antibiotics can be used only when prescribed by a veterinarian to protect the health of the animal.
These USDA-verified standards demonstrate the diversity of transparent and meaningful antibiotic-use policies, all of which can help make a positive impact on slowing the emergence of antibiotic resistance.
In the following section, two USDA-verified standards illustrate different approaches to restricting antibiotic use and how each approach can be put into practice by highlighting some of the basic criteria characteristic of transparent and meaningful standards, demonstrating the diversity of effective approaches currently available and underscoring the value of having a range of ways for producers to use antibiotics responsibly.
The “NAE” Approach
Many retailers and restaurant chains have begun to adopt NAE, which is clear-cut and easy for consumers to understand. NAE is verified by USDA and clearly defined (see Table 1). While it is more expensive to produce animals under NAE standards, data show that consumers are willing to pay more for NAE products.
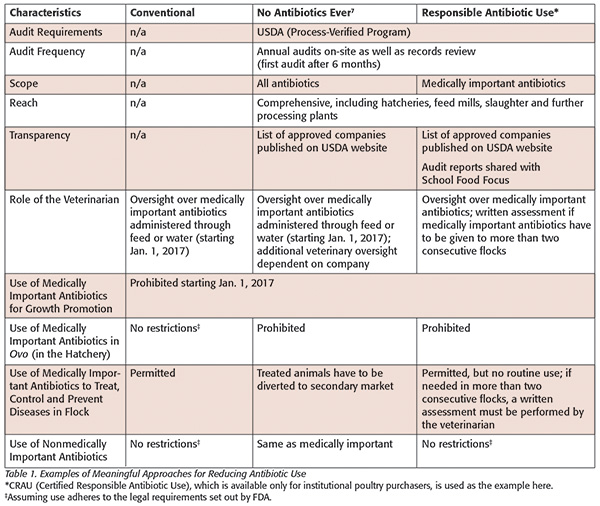 But consumers may not realize how difficult it can be for producers to meet, and then continue to operate under, the NAE standard. Making the transition to NAE production can take years, requiring continuous refinement of management practices as well as major investments, such as modifications to animal housing. Once established, daily NAE operations can be challenging for producers: For example, if a flock of chickens contracts a bacterial illness, the producer must treat the animals. However, the birds can then no longer be sold as NAE and must be diverted into another product line—meaning that NAE production depends on a secondary market for reclassifying and selling animals that have been treated for illness, usually at a lower price point.
But consumers may not realize how difficult it can be for producers to meet, and then continue to operate under, the NAE standard. Making the transition to NAE production can take years, requiring continuous refinement of management practices as well as major investments, such as modifications to animal housing. Once established, daily NAE operations can be challenging for producers: For example, if a flock of chickens contracts a bacterial illness, the producer must treat the animals. However, the birds can then no longer be sold as NAE and must be diverted into another product line—meaning that NAE production depends on a secondary market for reclassifying and selling animals that have been treated for illness, usually at a lower price point.
While more and more food producers offer NAE options, it is not the right approach for every company. Other, more flexible antibiotic-use policies can also have a meaningful impact in the fight against antibiotic resistance.
The “CRAU” Approach
The concept of responsible antibiotic use allows food producers to use the drugs under the supervision of a veterinarian when necessary to protect the health of the animals and to market these animals without needing to divert them into another product line. The viability of a responsible antibiotic-use approach has been demonstrated by the development of the CRAU standard for chicken sold to institutional purchasers.
Last year, The Pew Charitable Trusts’ antibiotic resistance project collaborated with School Food Focus to develop the CRAU standard for institutional purchasers—the first USDA-verified policy to outline what a responsible-use approach can look like in practice. (CRAU is now managed solely by School Food Focus.) CRAU is designed to lessen the public health threat posed by antibiotic use, address the need to treat sick animals and help meet the growing demand for responsibly raised animal protein. CRAU was developed specifically for chicken sold to institutions, so that large-scale buyers could purchase responsibly raised poultry.
The chicken industry was a logical first choice for developing a responsible-use standard, because the demand for chicken has surpassed that of beef, pork or turkey. About 8.7 billion chickens are grown for food production in the U.S. every year,[9] and chicken is the most common protein served to children in schools—with about $1 billion spent annually.[10]
Poultry companies interested in meeting the standard must undergo regular USDA audits to verify conformance, including comprehensive reviews of plant processes and facilities such as hatcheries, feed mills, barns and processing-packaging sites. The audit must document systems for proper product identification from the farm all the way to harvest and product purchase. Two of the nation’s leading poultry producers, Tyson Foods and Perdue Chicken, have already adopted CRAU, and some of the country’s largest school districts have committed to purchasing chicken produced under this standard.
Developing responsible-use standards for other proteins may be more challenging than for chicken, because the chicken industry is more highly integrated than other protein industries. Chicken producers can control every step of the production process, from the breeder flocks that hatch the birds to the feed that the chicks consume to harvest. Additionally, producers can limit antibiotic use in chickens more easily than in other food animals because of their relatively short life cycle: Chickens produced for food typically take 42 to 45 days to reach market weight, whereas cattle, for instance, typically take as long as 2 years. Despite these challenges, the approach is feasible across species; with increasing consumer and institutional demand for responsibly raised meat, it will probably be possible to establish similar standards for pork and beef that follow a responsible-use approach.
Market Impacts Driving Reductions in Antibiotic Use
Both NAE and responsible antibiotic-use approaches have made important strides toward reducing inappropriate antibiotic use. Each goes further in restricting antibiotic use than the U.S. Food and Drug Administration policy slated to come into full effect for all food animal producers in January 2017, which will eliminate the use of medically important antibiotics for growth promotion and require veterinary oversight of these drugs when added to feed and water.[11]
While NAE may be easy for consumers to understand, approaches that emphasize responsible antibiotic use, such as the CRAU standard for institutional poultry purchasers, are more feasible for many producers to meet and sustain by allowing them to provide a more affordable product while making substantive improvements to antibiotic use.
Both approaches provide important improvements compared with conventionally produced chicken by restricting the inappropriate use of medically important antibiotics and providing transparency and accountability. Both are independently verified by expert USDA staff. Most importantly, these two approaches address increasing consumer concern about antibiotics used in food animal production by assuring customers that the products they purchase are raised under meaningful, responsible antibiotic-use policies they can trust.
Table 1 compares some of the characteristics for three approaches to antibiotic use: conventional, NAE and responsible use.
Summary
The public health threat of antibiotic resistance is driving consumer demand for responsibly produced meat and poultry. Several viable standards, such as those outlined above, reduce antibiotic use. As poultry producers look to safeguard public health and use antibiotics responsibly, approaches such as responsible antibiotic use deserve wider consideration.
Karin Hoelzer, DVM, Ph.D., is an officer in health programs supporting The Pew Charitable Trusts’ safe food and antibiotic resistance projects. She focuses on research and policies related to foodborne pathogens, the use of antibiotics in animals and other public health risks related to the food supply.
References
1. midanmarketing.com/consumers-case/antibiotics-and-growth-hormones/.
2. notinmyfood.org/wordpress/wp-content/uploads/2012/06/CR_Meat_On_Drugs_Report_06-12.pdf.
3. www.who.int/dg/speeches/2016/antimicrobial-resistance-conference/en/.
4. www.cdc.gov/drugresistance/.
5. www.fda.gov/AnimalVeterinary/NewsEvents/CVMUpdates/ucm476256.htm.
6. www.ams.usda.gov/publications/content/download-transition-organic-factsheet.
7. www.ams.usda.gov/sites/default/files/media/Official%20ListingPVP.pdf.
8. www.ams.usda.gov/services/auditing/crau.
9. usda.mannlib.cornell.edu/usda/current/PoulProdVa/PoulProdVa-04-28-2016.pdf.
10. www.schoolfoodfocus.org/procurement-change-tools/craua-betterchicken/.
11. www.fda.gov/downloads/AnimalVeterinary/GuidanceComplianceEnforcement/GuidanceforIndustry/UCM299624.pdf.
Antibiotic Use in Food Animals: A Growing Threat to Public Health