Undeclared traces of allergenic substances in food may cause problems for allergic individuals as they are inadvertently exposed to the offending substance. To improve consumer safety, labeling of the eight major allergens became mandatory within the U.S. in 2006. These so-called “Big Eight” include tree nuts, peanuts, soy, eggs, milk, fish, wheat and shellfish.
To comply with allergen labeling laws and to protect their own reputation and business, food producers need analytical methods to monitor the presence of allergens during production and to avoid cross-contamination in production lines. How can food producers effectively select and implement a detection method from the range of methods available? In this article, the most widely used methods available at present and upcoming ones are described, and the pros and cons of the various methods are analyzed to facilitate the selection procedure.
Immunological Screening Methods
Immunological methods are most widely used to detect allergenic products at trace levels (i.e., a low mg/kg range) in foods. These methods are based on the binding of an allergenic protein by specific antibodies. Immunological methods are available in different formats, with the more conventional formats being enzyme-linked immunosorbent assays (ELISAs) and strip tests. Over the past few decades, many ELISAs and strip tests for the detection of different allergens have been developed and have become commercially available. More recently, research has focused on multiple-allergen detection (i.e., the development of methods in which several allergens or allergenic compounds can be detected simultaneously). These assays are usually developed on biosensors and microsphere-based flow cytometric systems. It is anticipated that within the next few years, these assays will also become commercially available. With the introduction of these types of assays, routine screening of products for the presence of the “Big Eight” will become possible.
Strip tests
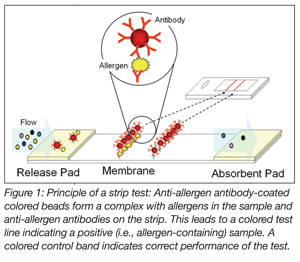 Strip tests (Figure 1) are based on the formation of complexes between anti-allergen antibody-coated colored beads with allergenic proteins in the sample and anti-allergen antibodies on the test strip. These complexes give rise to a colored test line on the strip, indicating a positive (i.e., allergen-containing) sample. In a similar way, a colored control band is formed, indicating that the test has been carried out correctly.
Strip tests (Figure 1) are based on the formation of complexes between anti-allergen antibody-coated colored beads with allergenic proteins in the sample and anti-allergen antibodies on the test strip. These complexes give rise to a colored test line on the strip, indicating a positive (i.e., allergen-containing) sample. In a similar way, a colored control band is formed, indicating that the test has been carried out correctly.
Strip tests are very easy to use, inexpensive, rapid (analysis time of a few minutes), do not require instrumentation and can therefore be used in the field. Today, most available strip tests are only qualitative; however, it is anticipated that in the near future, more and more suppliers will deliver simple handheld readers with which semi-quantitative results can be obtained. With strip tests, only single samples can be analyzed for the detection of a single allergen at one time.
ELISAs
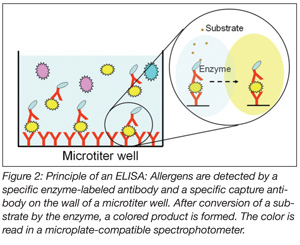 ELISAs are carried out in multiple-well strips or 96-well microtiter plates. The proteins of the allergenic compound are detected by a specific enzyme-labeled antibody and visualized by an enzymatic reaction that leads to the formation of a colored product (Figure 2). The color is read in a microplate-compatible spectrophotometer. The concentration of the allergenic compound in the sample can be determined from a calibration curve constructed by analyzing standards. To carry out an ELISA, trained laboratory personnel, standard laboratory equipment and a microtiter plate spectrophotometer are necessary. Using an ELISA, more samples (i.e., 48 or 96 including standards) can be analyzed simultaneously for a single allergen. The analysis time ranges from 30 minutes for fast ELISAs to three hours for standard ELISAs. At present, ELISAs are the most widely applied methods for detecting allergens by food processors and food control authorities.
ELISAs are carried out in multiple-well strips or 96-well microtiter plates. The proteins of the allergenic compound are detected by a specific enzyme-labeled antibody and visualized by an enzymatic reaction that leads to the formation of a colored product (Figure 2). The color is read in a microplate-compatible spectrophotometer. The concentration of the allergenic compound in the sample can be determined from a calibration curve constructed by analyzing standards. To carry out an ELISA, trained laboratory personnel, standard laboratory equipment and a microtiter plate spectrophotometer are necessary. Using an ELISA, more samples (i.e., 48 or 96 including standards) can be analyzed simultaneously for a single allergen. The analysis time ranges from 30 minutes for fast ELISAs to three hours for standard ELISAs. At present, ELISAs are the most widely applied methods for detecting allergens by food processors and food control authorities.
Biosensors
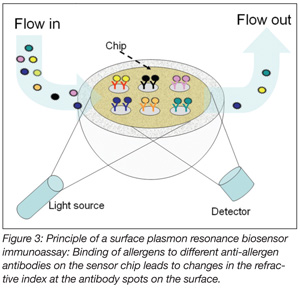 In food analysis, biosensors and, in particular, surface plasmon resonance (SPR)-based biosensors have become increasingly accepted tools. SPR detection is based on changes in the refractive index at the surface of a sensor chip, caused by the binding of an analyte to an immobilized ligand. For detection of high molecular-weight compounds such as allergens, specific antibodies are usually immobilized on the chip surface (Figure 3). The binding of allergens in the sample is followed in real time, and from the change in the signal, the concentration in the sample can be calculated from a calibration curve.
In food analysis, biosensors and, in particular, surface plasmon resonance (SPR)-based biosensors have become increasingly accepted tools. SPR detection is based on changes in the refractive index at the surface of a sensor chip, caused by the binding of an analyte to an immobilized ligand. For detection of high molecular-weight compounds such as allergens, specific antibodies are usually immobilized on the chip surface (Figure 3). The binding of allergens in the sample is followed in real time, and from the change in the signal, the concentration in the sample can be calculated from a calibration curve.
The majority of new biosensors are aimed at high-throughput and multi-analyte measurements. The major advantages of these systems are their short assay time (minutes), their high degree of automation that reduces labor time, the option to simultaneously detect several analytes and label-free detection. A major disadvantage of the majority of these systems is the relatively high price of both the machines and chips. Furthermore, only a single sample can be tested at one time, and trained laboratory personnel are needed. A few biosensor immunoassays for the detection of allergens have been developed by research groups and are described in the literature. It is expected that within the next few years, allergen test kits will become commercially available and will come to be applied in food control agencies.
Microsphere-based flow cytometric systems
 These assays are based on the flow cytometry detection of sets of differently colored micron-size beads (Figure 4). To each color-coded set of beads, antibodies against different allergenic compounds can be coupled. Specific, fluorescently labeled, second antibodies are used to visualize the binding of allergens to the beads. For analysis, different bead sets (to detect different allergens) are added simultaneously to a sample in a microtiter well. The beads are drawn up into a fluidic tube that causes the microspheres to line up in single file before they pass through the detection chamber. In this chamber, one laser identifies each bead and categorizes it into the appropriate bead set (based on which allergen is detected), while another laser scans the beads for the quantity of fluorescently labeled antibodies per bead (and determines the concentration of the detected allergen). In this way, multiple allergens can be detected simultaneously in a sample. The advantages of these types of assays are their very short analysis time (seconds), simultaneous detection of multiple allergens, small sample volume and relatively low-priced machines (compared with biosensors). However, labor times are comparable to those of ELISAs.
These assays are based on the flow cytometry detection of sets of differently colored micron-size beads (Figure 4). To each color-coded set of beads, antibodies against different allergenic compounds can be coupled. Specific, fluorescently labeled, second antibodies are used to visualize the binding of allergens to the beads. For analysis, different bead sets (to detect different allergens) are added simultaneously to a sample in a microtiter well. The beads are drawn up into a fluidic tube that causes the microspheres to line up in single file before they pass through the detection chamber. In this chamber, one laser identifies each bead and categorizes it into the appropriate bead set (based on which allergen is detected), while another laser scans the beads for the quantity of fluorescently labeled antibodies per bead (and determines the concentration of the detected allergen). In this way, multiple allergens can be detected simultaneously in a sample. The advantages of these types of assays are their very short analysis time (seconds), simultaneous detection of multiple allergens, small sample volume and relatively low-priced machines (compared with biosensors). However, labor times are comparable to those of ELISAs.
Selecting a Detection Method
When selecting a suitable detection method, the following criteria should be considered: availability of laboratory personnel and standard laboratory equipment, number of samples, number of allergens to be detected, frequency of testing, need for short analysis time, need for quantitative results, ease of use, degree of automation and the resulting costs. In Figure 5, a simplified selection scheme is presented.
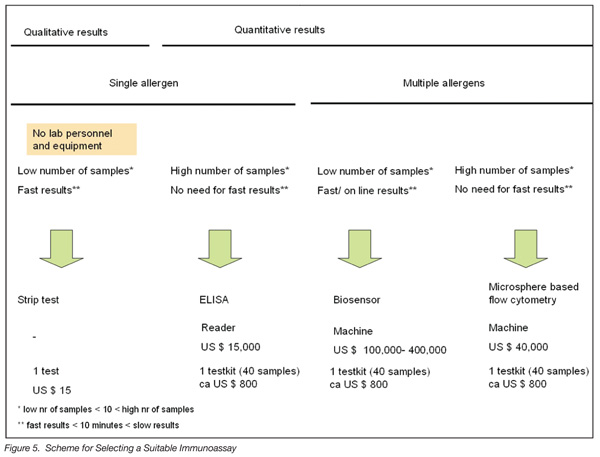
When a company doesn’t have trained lab personnel or equipment, the only tests that employees can perform by themselves are strip tests. When a high number of samples are required to be analyzed for only a single allergen, ELISA is the best option as the necessary equipment is relatively inexpensive. When multiple allergens need to be quantitatively analyzed in a few samples and when the results need to be obtained within an hour (e.g., before accepting incoming materials at receiving docks), biosensors are the method of choice. When a qualitative result is sufficient (i.e., “present at concentrations above the detection limit”), strip tests are an inexpensive and rapid alternative (no expensive machines needed). When there is no need for an especially short analysis time, microsphere-based methods are a better choice as the price of the necessary equipment is generally much lower than that of biosensors.
Before an immunological assay can be implemented, it is necessary to check whether the method is suitable for the matrix (or matrices) in which it will be used. This is important as the applied antibodies may cross-react with food-matrix components, leading to false-positive test results. Furthermore, food processing may affect the detection of allergenic proteins by antibodies as the structure and integrity of the target proteins and the recognition/binding by the antibody may be altered due to processing. In general, the kit supplier can provide validation data and is generally willing to validate the method in the required matrices or after processing. Otherwise, in-house validation for specificity and recovery is necessary. Samples suspected to contain allergens can be confirmed by using a mass spectrometry (MS) method. Using MS, parts of the unique amino acid sequence of a protein can be determined, and the protein can unambiguously be identified. MS, however, is labor-intensive, requires expensive equipment and materials and is not suitable for routine analysis.
In summary, there are several different immunological screening methods available for allergen testing and each has its advantages and disadvantages. The choice for a particular method should be based on the specific circumstances at the testing location (e.g., number of samples, number of allergens to be tested and required analysis time). The simple selection scheme presented can be used as a guide to select a suitable method. It is important that before implementation, the method is tested in the appropriate matrices after the applied production processes.
Monique Bremer, Ph.D., is a scientist in the Biomolecular Detection group of RIKILT-Institute of Food Safety, Wageningen UR The Netherlands. RIKILT-Institute of Food Safety is an independent, Dutch scientific organization that carries out research into the safety and quality of Dutch food. An expert in protein detection and identification, Dr. Bremer develops assays for the detection of allergens, processed animal proteins and growth hormone abuse. She is a member of the European Committee for Standardization Working Group CEN/TC 275/WG 12 Food Allergens.
Selecting a Suitable Food Allergen Detection Method




