It is not news to anyone who reads this magazine that sprouts, specifically green sprouts, have been the poster child for a fresh produce item that causes more than its share of food safety concerns. In the U.S. and Canada from 1989 to 2016, 58 illness outbreaks were attributed to green sprouts. Of the resultant 4,032 illnesses, 95 percent were caused by Salmonella and 5 percent by Shiga toxin-producing Escherichia coli.[1]
As the U.S. Food and Drug Administration (FDA)’s website states, “In outbreaks associated with sprouts, the seed is typically the source of the bacteria.”[2] Therefore, it isn’t surprising that much of the research on improving sprout safety has focused on seed disinfection. FDA’s 1999 guidance on sprout production mainly covers three areas: Good Manufacturing Practices (GMPs), seed disinfection (such as “20,000 parts per million calcium hypochlorite”) and irrigation water testing.[3–5] The 2017 draft guidance, which grew from 19 pages in 1999 to 125 pages, provides much more detail but follows the same general areas of GMPs, seed disinfection and irrigation water testing. The new draft guidance document does mention that “seed contamination, when it occurs, may be at low levels….” We’ll come back to that in a minute.
One of the tenets of creative problem solving is “a problem well defined is 80 percent solved.” This has been stated by many wise folks in a number of ways, but the idea is always the same. For example, Albert Einstein supposedly said, “If you have one hour to save the world, spend 55 minutes defining the problem and only 5 minutes finding the solution.” We are not suggesting that solving the problem of sprout safety is analogous to saving the world, but with that teaching in mind, we focused and dug deeply into why sprouts have caused so many illnesses.
Sprout Challenges
As part of defining the problem, we looked at data on pathogens found in lots of seeds that had been traced to illness outbreaks. Only two seed lots were reported to be contaminated. The levels of pathogens enumerated in these lots were Salmonella muenchen at 16.2 CFU/kg and Salmonella mbandaka at 13.3 CFU/kg. That certainly is a low level of contamination, probably not enough to make anyone ill. It works out, on average, to having to eat thirty-six 10-g servings of sprouts to ingest just one pathogenic bacterium, if there were no increase in pathogens from that level found in the seeds. So why have so many people gotten ill from eating green sprouts?
Unfortunately, pathogens do increase in numbers in sprouts that have been conventionally grown. Many studies have found a huge increase in pathogens during the process of growing the sprouts. Obviously, at least one pathogen must be present when the seeds are planted. The standard process of growing green sprouts is in a slowly rotating drum, at room temperature, with “irrigation” water added every 15–30 minutes. This is a great environment not only for growing sprouts but also for growing pathogens such as Salmonella, E. coli and even Listeria monocytogenes.
Our hypothesis is that the real “problem” is the huge increase in pathogens that can occur during the process of growing sprouts. Therefore, we set out to find a method to prevent that increase. We explored several paths, including organic acids, bacteriocins and competitive inhibition with lactic acid bacteria. All good ideas, but none of them really worked very well. What did work, and what the rest of this article will describe, was growing the sprouts at approximately 40 °F.
Preventing Pathogen Growth in Sprouts: What Worked
 The first thing we did was to move from the conventional drum system to growing sprouts in what could be a retail container. This not only made the experiments much easier to run, but it also just seemed like a better way to grow and merchandise sprouts. Once the seeds are planted, the sprouts are not touched until the end-user (consumer or foodservice) “harvests” them for use on a sandwich, salad or other use. The container was designed so that it would contain all the water the seeds would need to grow into sprouts but also keep the seeds from “drowning.” Therefore, our container held 35 g of hydrated seeds (hydrated seeds = ~1 g of air-dried seeds that had imbibed about 1 g of water) on a nonwoven hydrophobic “platform” and 60 g of water for the seeds to use as they grew. The container was also designed to provide adequate ventilation, as determined by measuring oxygen and carbon dioxide levels in the container during seed germination and growth (Figure 1).
The first thing we did was to move from the conventional drum system to growing sprouts in what could be a retail container. This not only made the experiments much easier to run, but it also just seemed like a better way to grow and merchandise sprouts. Once the seeds are planted, the sprouts are not touched until the end-user (consumer or foodservice) “harvests” them for use on a sandwich, salad or other use. The container was designed so that it would contain all the water the seeds would need to grow into sprouts but also keep the seeds from “drowning.” Therefore, our container held 35 g of hydrated seeds (hydrated seeds = ~1 g of air-dried seeds that had imbibed about 1 g of water) on a nonwoven hydrophobic “platform” and 60 g of water for the seeds to use as they grew. The container was also designed to provide adequate ventilation, as determined by measuring oxygen and carbon dioxide levels in the container during seed germination and growth (Figure 1).
Results of Inoculated Pack Pathogen Studies
Seeds were disinfected (2,000 ppm NaOCl adjusted to pH 6.0 with acetic acid for 15 minutes), rinsed and hydrated as described above. Thirty-five grams of hydrated seeds and 60 g of water were planted in a container. At this point, the samples were inoculated with either a cocktail of five pathogenic strains of Salmonella or four strains of E. coli O157:H7. The target inoculation level was 103 to 105 CFU/g. Enough trays were prepared so that at each evaluation time, three individual trays could be analyzed separately. The sprouts were then grown at 70 °F and 40 °F for 2 or 21 days, respectively. Following the appropriate growth period, trays were placed at 40 °F for the shelf-life phase of the study. All studies were run at Deibel Labs in Madison, WI. The geometric mean of the three samples was calculated, and the data are displayed below.
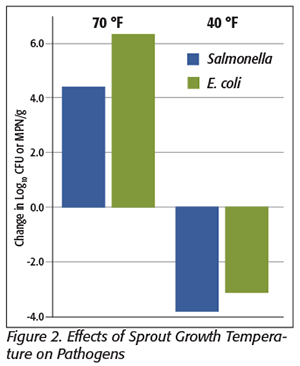 Figure 2 provides a good visualization of the results. With seeds germinated and grown at 70 °F, there was the expected 4- to 6-log increase in the population of both Salmonella and E. coli. However, when the sprouts were grown at 40 °F, there was not only no increase in the populations of either pathogen, but there was a several-log decrease in both Salmonella and E. coli.
Figure 2 provides a good visualization of the results. With seeds germinated and grown at 70 °F, there was the expected 4- to 6-log increase in the population of both Salmonella and E. coli. However, when the sprouts were grown at 40 °F, there was not only no increase in the populations of either pathogen, but there was a several-log decrease in both Salmonella and E. coli.
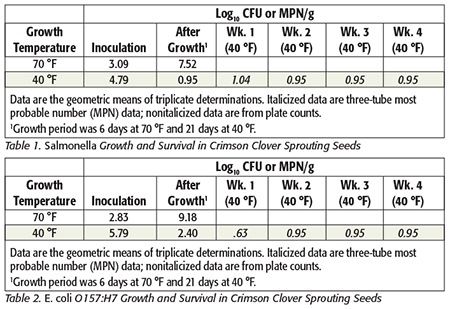 As can be seen in Tables 1 and 2, there was no increase during the subsequent 4-week shelf life of these sprouts. Interestingly, growing sprouts at 40 °F not only prevents the growth of Salmonella and E. coli, but also significantly increases product shelf life.
As can be seen in Tables 1 and 2, there was no increase during the subsequent 4-week shelf life of these sprouts. Interestingly, growing sprouts at 40 °F not only prevents the growth of Salmonella and E. coli, but also significantly increases product shelf life.
We conducted several additional inoculated pack studies with crimson clover where we looked at the addition of potential inhibitors, such as organic acids and bacteriocins, in the soak water. The results were essentially the same as reported above: Germination and growth at 40 °F was the key variable in preventing the multiplication of Salmonella or E. coli. There was no significant additional inhibition observed with the use of these inhibiting compounds.
We also replicated this study with alfalfa seeds. We got the same results as shown with crimson clover: a several-log increase in bacteria when sprouts were germinated and grown at 70 °F, and a several-log decrease when germinated and grown at 40 °F.
Although these are very positive results, what about L. monocytogenes?
For this experiment, we used a cocktail of five strains of L. monocytogenes and inoculated at approximately 10 CFU/g. Although this level was lower than what we used for the Salmonella and E. coli challenge studies, it is very likely higher than would be encountered in seeds. Data (personal communication) from our seed supplier revealed that from 2012 to date, over 16,000 seed samples were tested; none were positive for L. monocytogenes, which is most frequently an environmental contaminant.
 The results are displayed in Table 3. At 70 °F, even at this level of inoculum, L. monocytogenes displayed more than a 6-log increase. However, when the sprouts were grown at 40 °F, there was no pathogen growth, during either the germination and growth period or the 4 weeks of shelf life at 40 °F.
The results are displayed in Table 3. At 70 °F, even at this level of inoculum, L. monocytogenes displayed more than a 6-log increase. However, when the sprouts were grown at 40 °F, there was no pathogen growth, during either the germination and growth period or the 4 weeks of shelf life at 40 °F.
Summary
In summary, we feel that the reason why sprouts have caused an inordinate number of foodborne disease outbreaks and illnesses is a function of the traditional growth process at room temperature. If any pathogens are present, their numbers increase by several log cycles and are present at infectious levels. We have developed a process for growing sprouts at 40 °F that prevents the outgrowth of any pathogens (Salmonella, E. coli) that might be present, even in disinfected seeds. In fact, in inoculation studies, the populations of Salmonella and E. coli actually decreased. L. monocytogenes, if present at approximately 10 CFU/g, also will not increase in numbers when using this process. Additionally, growth at 40 °F also extends the shelf life of the product to 4 weeks at 40 °F. It has long been recognized that the presence of L. monocytogenes in refrigerated ready-to-eat foods at levels of 100 CFU/g or less does not present a public hazard.[6] In our long research with this technology, we have not found L. monocytogenes in incoming seeds. More importantly, our experiments have shown that no growth or very minimal growth of L. monocytogenes occurs during the production and distribution of these sprouts. To differentiate them in the consumer marketplace, we call them “Cold Grown Nanoshoots,” which are being grown by RaFoods.[7]
We would like to acknowledge the technical assistance provided by Deibel Labs, Madison WI, in reviewing the design and executing these experiments. Our thanks go out to Rick Boehme, Susan Kelly and Holly Jaeger. They made working with pathogens a little less stressful!
Dennis Lonergan, Ph.D., is the chief science officer at Vista Institute LLC.
Jeffery Sholl, Ph.D., is the chief executive officer of RaFoods LLC.
William Sperber, Ph.D., is president of The Friendly Microbiologist LLC.
References
1. barfblog.com/wp-content/uploads/2014/12/sprout-associated outbreaks-12-8-14.xlsx.
2. www.foodsafety.gov/keep/types/fruits/sprouts.html.
3. Holliday, SL, AJ Scouten and LR Beuchat. 2001. “Efficacy of Chemical Treatments in Eliminating Salmonella and Escherichia coli O157:H7 on Scarified and Polished Alfalfa Seeds.” J Food Prot 64:1489–1495.
4. Stewart, DS et al. 2001. “Growth of Salmonella during Sprouting of Alfalfa Seeds Associated with Salmonellosis Outbreaks.” J Food Prot 64:618–622.
5. Beuchat, LR. 1997. “Produce Handling and Processing Practices.” Emerg Infect Dis 3:459–465.
6. Chen, Y, WH Ross, VN Scott and DE Gombas. 2003. “Listeria monocytogenes: Low Levels Equal Low Risk.” J Food Prot 66:570–577.
7. www.rananoshoots.com.
A New Approach to Solving the Problem of Sprout Safety