Should a food processor conduct internal inspections? To those companies for which such programs are an integral part of their quality or safety programs, this may seem like a rather silly question. Self inspection is, to most food processors, an integral part of their quality assurance (QA) program. There are, however, operations that have no such programs, and have never even considered implementation.
Why should a processor conduct internal inspections? There are several very good reasons. Among them:
• Assuring product quality
• Assuring compliance with the Current Good Manufacturing Practices (cGMPs)
• Maintenance of good sanitary practices
• Having accurate records and observations of plant practices and changes can enhance operations
• Assuring that the facility is not unsafe to the workers
• Assuring the production of safe and wholesome food
The later point is one which is required by the United States government under the provisions of the Pure Food, Drug, and Cosmetic Act. This point is also the cornerstone of a Hazard Analysis and Critical Control Points (HACCP) program. Looking at it in this light, inspection almost evolves into a legal requirement. In fact, for those operations that have implemented HACCP programs, or those that are planning to do so, it is or will become mandatory.
Inspection is generally only a part, albeit an important one, of a company’s QA program. It may be even be used as a quality control (QC) check, in fact. Before going further, it is essential that everyone understand the difference between quality control and quality assurance. QC is a part of QA program, and may be defined as; “the scientific evaluation of production consisting of on-line evaluation of finished products, raw materials, and packaging materials to determine adherence to accepted standards.” By using an inspection to evaluate adherence to accepted practices, QC becomes part of the QA program.
Quality assurance has been defined as “all encompassing programs, including such aspects as quality control programs, setting of standards, evaluation of incoming materials, development of tracking and coding systems, and adherence to GMPs, designed to ensure to an established degree of confidence that products are produced, packaged, distributed, and ultimately reach the consumer in a given condition.” The key phrases are “all encompassing programs” and “degree of confidence.” These concepts reflect an organized and systematic approach towards achieving a goal; that is, safe food that meets established, well-defined quality parameters or specifications.
The final benefit of self-inspection programs is financial. It is simple, good economic sense to have programs in place that assure that your facilities are operating within regulatory guidelines and your internal operational standards. A clean and well run facility is one in which your sales staff can market as a more desirable supplier or co-packer to your customers downstream. There is usually reduced waste, and even the potential for reducing insurance premiums. The literature is full of citations where operations “got out of control” resulting in lost product, adverse publicity, product recalls, and even plant closures. These situations may never have developed if comprehensive internal inspection programs had been in place. Management, particularly in the U.S., must realize that their food safety and quality departments should not be treated as cost centers. They should be considered cost-savings centers—a point not usually recognized until too late.
Keys to the Program: Who Will Do the Work?
There are many kinds of in-house or self inspections. In this article, we will focus on how one might conduct a sanitation inspection, and provide insights into what kind of commitment management should make to provide their staff with the proper tools to do this work. The goal of a inspection is to evaluate evaluating sanitation practices, how staff is complying with the GMPs and to determine whether there are situations that may adversely affect product safety. The cGMPs should be employed as a guideline. If one is an outside or third-party investigator, such an inspection is an excellent tool for “getting a handle” on management’s commitment to proper food handling.
The first key is having someone on staff who is capable of conducting such inspections. There are different ways that companies go about developing this resource. An organization may already have one or more persons on staff that are knowledgeable about what is required to conduct a sanitation inspection. If this expertise is not on staff, the next best thing is to work with an outside expert and have that organization or individual provide the appropriate education to members of your staff. Be forewarned that proper education is not cheap and that one or two lessons does not a sanitarian make. If you elect to go this route, allow the contractor to complete the full education program. When looking for someone who can do this kind of training, look for an individual who enjoys the work, is a good communicator, understands the process and will not be offended when his or her protégé can take over on their own.
The final way to have a sanitarian on staff is simply to appoint someone. I would not recommend this approach, unless there is a commitment to provide that person with appropriate training and education. Sadly and surprisingly, this last option is employed by all too many companies. They then compound the problem by failing to provide the individual with the proper education, frustrating him or her and hamstringing the program.
Keys to the Program: What You Need to Know
The in-house inspectors or sanitarians must develop expertise in a number of different areas. It is also quite helpful if the individual is blessed with common sense, a good sense of awareness and an ability to understand how problems can develop, which usually means that the person has the ability to solve problems. Areas of expertise include, but need not be limited to:
1. Plant Standard Operating Procedures (SOPs). The individual should understand plant and/or warehouse operations and protocols, which includes SOPs for production, cleanup and maintenance. They should have access to all manuals elucidating these operations. It is also important to understand how staff are educated, if they are. For example, are employees given basic education in food plant sanitation and GMPs? Is there a commitment to employee education?
2. cGMPs. These individuals should understand the current Good Manufacturing Practices or, if the operation is involved with meat or poultry processing, the U.S. Department of Agriculture’s (USDA) sanitation requirements. It is the cGMPs that should be used as the basis of sanitation inspections by investigators from the U.S. Food and Drug Administration (FDA). The cGMPs are what may be called “interpretive” regulations. They provide FDA investigators with a great deal of leeway in interpreting what is and what is not an adequate practice. The regulations use the terms “should” and “shall.” Practices that “should” be done are recommended; a “shall” is mandatory. In an internal inspection, the inspector, must use established company guidelines or policies to determine what constitutes a concern. He or she should then make recommendations as to how it can be remedied.
3. Pest Control. Pest management and control protocols or practices is another area that needs to be learned by the in-house sanitarian. Many companies employ outside agencies to control, discourage or kill pests. Work with these individuals and learn why they do what they do, as well as to assure yourself that they are doing their job. Many of the pest management company technicians that provide services to food manufacturing, foodservice and retail operations have a high level of expertise that you can tap into to learn about the potential problems and solutions specific to your operation.
4. Local, State and Federal Regulations. Inspectors should familiarize themselves with all regulations that affect their operation. They should also get to know the agencies and investigators who will be coming to their plant(s). Working with regulatory agencies is much easier than trying to work against or outsmart them. The agencies usually win in the end.
5. Labor Issues. Do you work in a union shop? Who is the steward? Any changes or improvements that are recommended as a result of your work will affect how the plant operates. Getting these things done may be next to impossible without the blessings and cooperation of labor.
Key Criteria for Evaluation
The next key issue is the establishment of some method or methods of evaluation. How will you weigh violations? How will you score the plant as a whole? Will individual areas be evaluated alone or as part of the whole? This is an area where there has been and will continue to be disagreement among food safety and sanitation professionals. Perhaps the most common criteria for evaluation is the use of a scoresheet in which different areas are given points and the plant is given a final grade based on the total points achieved. Scoring above a set value means the facility is considered to be “within compliance.” In fact, with some organizations, the fact that they score well on an audit is used for more than just assuring good sanitary operations. These audits are a source of pride within the company and become a means for marketing that company.
Scoring systems have their pros and cons. Everyone likes to have a target number but there are times when numbers can cause problems. For example, a plant may “pass” yet have some deficiencies. Management may feel that since the facility passed, there is really no pressing need to address problem issues. Operations degenerate, and suddenly there is a full-blown problem. As an example of why scorecards can be a problem, a colleague once described an inspection of a potential co-packer that she had conducted. The company had very poor sanitation practices but was offering a very low price for contract packing. Poor sanitary practices are usually indicative of poor management commitment to sanitation and other programs. The inspector’s company used a 0 to 100 scoresheet, with 85 considered to be passing. This company scored a 47. The questions she got from her management were: What do they have to do to pass, and could we lower the passing grade for these people, because we were offered “such a deal”?
These kind of comments underscore the importance of both management support for sanitation and the development of clear policies and procedures. Once an operation begins to compromise their quality standards, the whole program is jeopardized. A classic example is hair or beard net requirements for workers. If managers or guests fail to comply with the rules, workers will develop a “why bother” attitude. It also emphasizes the need to develop and implement a strong program from the very beginning, and to follow those guidelines to the letter. It is very difficult to tighten standards once an operation has been allowed to operate under what might be considered substandard conditions.
There are also those who simply favor using only a notebook to write down observations, both good and bad. These observations may then be written up in more detail at a later date. This is similar to what a FDA investigator will do when he or she does an inspection and files the inspection form. They will only note deficiencies.
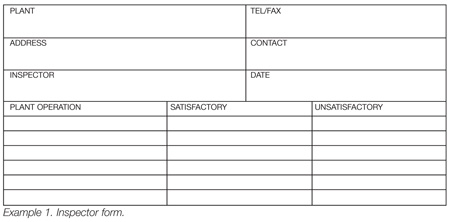 Others favor the use of checklists designed specifically for a plant. These, too, have their advantages and disadvantages. Checklists are very easy to use but can also be dangerous in that there is a potential for “dry labbing” the inspection. This means the inspector simply checks all appropriate boxes without really examining the operation. If there are only two options, as may be seen in Example 1, the potential that this might occur is higher. I have seen checklists in which the inspector simply drew a line from the top to the bottom of the page indicating that each unit operation was satisfactory. The question lingers, however, “Did that individual really look at everything?”
Others favor the use of checklists designed specifically for a plant. These, too, have their advantages and disadvantages. Checklists are very easy to use but can also be dangerous in that there is a potential for “dry labbing” the inspection. This means the inspector simply checks all appropriate boxes without really examining the operation. If there are only two options, as may be seen in Example 1, the potential that this might occur is higher. I have seen checklists in which the inspector simply drew a line from the top to the bottom of the page indicating that each unit operation was satisfactory. The question lingers, however, “Did that individual really look at everything?”
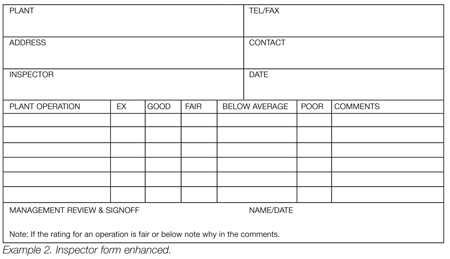 If a checklist is used, it is suggested that a scale of some sort be used for each point that must be graded, and that a place for comments be provided, as may be seen in Example 2. Look at the two types of checklist designs. Which will provide greater assurance that the inspector really looked at things?
If a checklist is used, it is suggested that a scale of some sort be used for each point that must be graded, and that a place for comments be provided, as may be seen in Example 2. Look at the two types of checklist designs. Which will provide greater assurance that the inspector really looked at things?
The Essential Key: Management Support
The final key, management support, is the most important. Without the support of management, any program, whether sanitation inspection, HACCP, or statistical quality control (SQC), is doomed to failure. If your company believes that self inspection is important, get it in writing from the top. Be sure that this person or group of persons has committed money, supports the protocols established for inspection and correction of deficiencies, and understands the importance of these programs. There is nothing more frustrating than to conduct a project believing that management is behind you, and see it die on the vine. Get their support and work to keep it.
Preparing for the Inspection
How does one go about preparing for a self inspection? This really depends upon the type of inspection that you are planning. There are two basic kinds of sanitation inspections: routine and detailed. The routine inspections are just that, routine. They may be conducted on a daily or weekly basis, and may be as simple as walking through the warehouses or processing facilities to just look at what is going on. They may involve the use of a checklist and are simply targeted at assuring yourself that the pest control agent has checked all the traps, or that cleanup was conducted as it was designed. Most plants’ employees get used to seeing their sanitarian or inspector in the plant every day. Just hope that they do not begin to take that individual for granted.
Detailed inspections are designed to take a comprehensive look at the facility’s overall compliance with the cGMPs; determine if any potentially unsafe or dangerous situations are developing to determine if there are any situations that may result in compromised product; and are an integral part of HACCP verification or validation.
These inspections should examine a plant from top to bottom. It is this kind of inspection that most FDA investigators will do when they come to your plant. This is also the kind of audit that should be expected from any third party inspector, whether from a reputable consulting firm or an outside inspection agency. Before conducting such an inspection, whether in your own facility or in a sister operation, do not announce your intentions. Once people learn that they are going to be inspected, they start to clean up or change their habits. The objective of the inspection is to view normal operations, and recommend changes, if changes are in order.
The in-house inspector should dress appropriately. A three-piece suit or evening gown is not appropriate. The inspection will be from top to bottom, so the inspectors are going to get dirty. Rugged, yet clean clothes, such as “whites” or a jumpsuit are often worn. Hair nets and beard nets, if necessary, are a must. All jewelry, watches and objects that could get into food should be removed. A bump cap is highly recommended. The more one works in plants, the more one realizes this. The inspector (and management) should set an example for the plant staff. If an inspector fails to wear proper hair restraints, keeps his jewelry on or fails to follow established plant practices, workers notice. They will say to themselves, “Why should I bother obeying these rules, if management doesn’t seem to think they are important?” The bottom line is that such actions could undermine the whole program.
There are certain tools that the inspector should carry with him during the inspection. A notebook (or scoresheet) and pens will be necessary. The latter should be secured or in hand so it will not be lost. A flashlight is also a necessity, preferable one with an unbreakable lens. Other tools an investigator may want to carry are a hand lens (plastic), a knife (Swiss Army, for example), a backlight to test for rodent urine, Whirl-Pak bags or Petri dishes (plastic) for sample collection, markers, and copies of pertinent regulations. Securing these tools (flashlight, knife and pens) in a scabbard hung on a belt is one way to protect the tools from being lost and keep them easily accessible. An inspector may also want to pack his tools in a fanny pack. This will protect them, allow access and prevent their loss. If one does elect to carry a black light, remember that the lens is glass and could break, so take care to protect it and the product.
Conducting the Inspection
The first step of the sanitation program self inspection is simply to walk through the processing plant. Begin at the receiving docks and simply follow the process flow through the plant to the finished goods warehousing area. The in-house inspector should reacquaint himself or herself with the facility’s process flow chart before beginning the walk-through.
The inspection should begin with the grounds. Inspectors should walk the property lines and look at areas away from the plant. Rodent and pest infestations frequently originate off site. You want to know if your neighbors are a possible source for any problems. Look at how the grounds are maintained; how vegetation is controlled; whether areas drain properly (pests are attracted to water); if materials are stored properly; whether roads or lots are a source of dust and dirt; and if there are pest control measures being taken and applied properly.
Next, walk around each building, and if possible, get up on the roofs. Is there adequate storage for all non-food materials and are they stored properly? Are there harborage sites for insects and rodents? Are walls, windows and doors designed to exclude pests? Are screens maintained? Are there situations that could compromise your product? One area that may be used as a yardstick for management’s commitment to sanitation is the condition of the used equipment storage area, commonly called the “boneyard.” Questions related to the interior of the building and equipment design are best asked when looking at those operations.
Now, move to the receiving docks. Do not just look at how the area is set up; rather, watch what is going on. Are the employees doing their jobs properly? Are plant protocols being followed? Are there situations developing that may compromise product safety? Are recording devices functional and being maintained? This is especially important when dealing with refrigerated products. Are incoming trucks clean and well maintained? If there is rain, do the areas below the docks drain, or is there six inches of standing water sitting there?
Follow the process flow through the plant and ask similar questions for each unit operation. Be sure to take note of the employees. They are an excellent barometer of management commitment to food safety and quality. If they appear slovenly, fail to utilize handwash stations, expectorate on the floors, are wearing jewelry or chewing gum, there is a definite problem. It is during this phase of the inspection that you should examine equipment and plant design and maintenance. Food handling and food contact equipment should be constructed of easily cleanable materials. Belts should be in good condition, constructed of cleanable materials, and properly joined. Floors should be clean; drains should drain properly; and walls, windows, screens and ceilings should be well maintained. The plant should also smell clean. Odors indicating spoilage or product degradation indicate that there is a problem somewhere. Also, use your sense of touch. If you run your finger over a piece of equipment that is supposedly clean and it appears slimy, there may be a biofilm problem.
Warehouses should be properly maintained. Corridors between products stacks and walls should be maintained at least 18 inches. This allows access to assure proper cleaning and for pest control. It also allows easy access for inventory. If one is forced to begin moving pallets to do an inventory, the cost of the inventory increases. Spills should be cleaned immediately. Temperature recording and temperature indicating devices and alarms should be maintained; temperatures should be maintained at specified levels in refrigerators, freezers and warehouses. Chemicals and cleaning materials should be properly stored in areas designed to contain spills. These areas should be locked and signs warning of potential hazards and indicating that only authorized personnel are allowed to enter should be posted.
Employee facilities must also be included in the inspection. Breakrooms should be clean; toilet and handwash stations should be clean and supplied with the necessary accouterments; signs telling employees to “Wash Hands Before Returning to Work” must be posted; lockers should be clean and elevated off the ground to allow for proper cleaning and to eliminate harborage sites. Employees should be aware of basic GMPs. Employees’ lockers have served as points where pest infestations have spread to entire plants.
The audit should also review recordkeeping procedures. Are records, particularly those related to assuring food safety, being maintained properly? Are there records of equipment, instrument and recording device maintenance and standardization? These are the questions the investigators from government agencies and that potential buyers will ask, so it behooves you to ask them first.
Finally, the in-house inspector should be sure to observe cleanup. Is the crew cleaning the plant properly? Are they being monitored? Are there any means to evaluate their performance? Are they forgetting things? Are they cleaning up in such a way that they may be compromising overall plant sanitation? Listeria has been determined to be a potential environmental problem. Blasting drains with high pressure hoses may cause the organism to become part of an aerosol, spreading it through the plant. Proper cleaning and sanitizing is essential to good plant operations.
When conducting the inspection, it is essential to keep your eyes and ears open. Talk to persons on the line or in different areas, if it is possible. These people may give you other insights into potential problems. Finally, be tough. Record all potential concerns, no matter how minor they may seem. The small ones may grow into larger ones, and if you can see them, who is to say an investigator from a regulatory agency or a third party auditor will not? And take your time. Sitting or standing in one place and watching an operation for an extended period of time may reveal that a problem is in the making.
Reporting and Implementation of Recommendations
Once the inspection is completed the inspector must then sit down with plant staff to review his or her observations. If the audit is conducted by an outsider, this meeting is referred to as an “exit interview.” The meeting should be a summation of the overall plant condition, highlighting major concerns that should be addressed immediately.
The next step is to prepare a detailed report based on observations. The report should incorporate the following:
1. Observations: All suspect and potential adulteration or safety issues should be described.
2. Degree of Concern: So that those issues of most concern are addressed first, a grading system for observations should be established. An example might be to use the terms critical, major and minor, where critical indicates a situation that will result in product adulteration requiring immediate attention; major indicates a situation that could result in adulteration in the near future; and minor indicates situations of concern that may or may not result in adulteration in the future.
Using a grading system has the inherent problem that some concerns may be ignored, but the hope is that if issues are ignored, it will be the minor concerns that receive short shrift.
3. Recommendations: Suggested actions for addressing each area of concern should be made. These should have been reviewed in the exit interview.
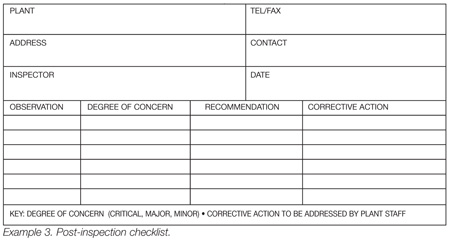 4. Timelines: An area on the reporting form allowing those persons responsible for making changes should be provided. Encourage these persons to provide a timeline as to how long it will take them to correct the problem. They should also use this form to describe their corrective actions. This term is what is used on many forms. This is one advantage of using a post-inspection checklist, shown in Example 3.
4. Timelines: An area on the reporting form allowing those persons responsible for making changes should be provided. Encourage these persons to provide a timeline as to how long it will take them to correct the problem. They should also use this form to describe their corrective actions. This term is what is used on many forms. This is one advantage of using a post-inspection checklist, shown in Example 3.
The final report should group observations by area. For example, all observations pertaining to the receiving dock should be combined. This allows that part of the report is given to the supervisor responsible for that area. It will be his or her responsibility to address the deficiencies.
No matter what format is selected, the completed report is then submitted to management. It will be management’s responsibility to call a meeting of key staff to address the concerns described in the report. Now assuming that management has already agreed to support this inspection program, the meeting date should be set quickly, perhaps within a day or so after the actual inspection was conducted.
At the staff meeting, the complete report should be reviewed. There will obviously be discussion of observations, degrees of concern and recommendations. If you are the inspector, you will probably have to defend the report. Let people know why observations were made, cite the regulations, and make sure they understand the concerns and their importance to the whole operation.
Again, the key to success is management support. If the support is there, the concerns will be addressed. The ideal situation for the inspector is for management to simply say, “Get those sections of the report related to your areas back to me within a set period with actual or proposed corrective actions and timelines for when you will or have completed the work.” This puts the onus on the different supervisors or foremen. It will be the inspector’s responsibility to evaluate whether the observations have been addressed and report to management. The inspector should also work with the different persons responsible for implementing the recommendations to ensure that they are done right the first time.
Evaluating Efficacy of the Program
This may be the hardest part of the program. How does one evaluate how effectively something is working? The first and most obvious way is during follow-up inspections, either routine or detailed. If fewer suspect practices are observed, if changes are made and maintained, and if the operations appear improved, progress is being made. This kind of observation is somewhat subjective, however. What else should one look for?
Look at the employees next. It has already been stated that they are a mirror of management’s commitment to safety and quality. Are they supportive of the program? Do they seem to take more pride in their work? Is their overall appearance improved? Ideally, the good supervisor should have gotten his employees involved in making the recommended changes. Getting them involved provides a sense of ownership and pride. This, too, is somewhat subjective.
Since management has given their support for the self-inspection program, they will need to see results. Management’s idea of results all too frequently can be distilled down to “the numbers.” How is this program improving our operation’s bottom line? So, where are these numbers? They can be found, but it will take more work on your part. There are several areas of operation where enhanced safety and quality resulting from the inspection program may show up. These include reduced rework, improved operational efficiencies, reduced downtime, fewer consumer or buyer complaints, fewer product returns, reduced waste, and the potential for eliminating recalls. Each of these areas, and there are undoubtedly more, can be measured, and the bottom line is reduced costs. Remember, operations such as plant inspection, quality assurance and food safety should be viewed as cost-savings centers, which they truly are.
Richard F. Stier is a consulting food scientist with international experience in food safety (HACCP), food plant sanitation, quality systems, process optimization, GMP compliance and food microbiology. He has worked with a wide range of processing systems and products, including canning, freezing, dehydration, deep-fat frying, aseptic systems, and seafood processing. Rick served as Director of Quality Assurance for Dole Packaged Foods North American operations. In this capacity, he was responsible for building programs targeted at ensuring the quality of Dole value-added products packed in the U.S. He is a member of the Institute of Food Technologists and an editorial advisor to Food Safety Magazine. He can be reached at rickstier4@aol.com.
Plant Self-Inspections: Keys to Developing In-House Programs




