One of the hottest and fastest growing product categories in the food industry is the “nutraceuticals” or “functional foods” (as they are often called) market. As more and more consumers seek out these types of products, food manufacturers and marketers are focusing their efforts on developing and integrating functionality into their products to meet this rapidly expanding marketplace.
According to recent data, sales of functional foods currently exceed $80 billion worldwide. In the U.S., nearly $15 billion was spent on functional foods in 1997, and the rate of growth is exceeding 12% a year. Compare the statistics to the small 3% growth for traditional foods (natural foods are the other success story with a healthy 10% growth) and it’s clear why nutraceuticals are the market to target in 2001.
The term “nutraceutical,” although not defined by any regulatory agency, is any substance that may be considered a food that provides medical or health benefits such as prevention and treatment of disease. However, how a company utilizes this terminology in the marketplace can get them into regulatory trouble if they are not careful.
There are three major categories of products that can be used to bring nutraceuticals to market. These include foods, dietary supplements and drugs. Difficulties arise for many companies because of the increasingly stringent and challenging regulatory environment relating to functional claims for particular ingredients.
The Nutrition Labeling and Education Act (NLEA) resulted in FDA regulations that define the allowable health claims for foods. These include nutrient/disease relationships between diets high in calcium with osteoporosis; diets high in fiber from fruits, grains and vegetables with cancer; diets high in fruits and vegetables with high fiber; vitamins A or C with cancer; diets high in fiber from fruits, vegetables and grain products with heart disease; diets low in fat with cancer; diets low in saturated fat with cholesterol and heart disease; diets high in soy protein with coronary heart disease: and diets low in sodium with high blood pressure. The NLEA health claims offer the industry opportunities to develop products with calcium fortification, soy protein ingredients, natural fiber containing ingredients from grains, fruits and vegetables, and reduced fat, cholesterol and sodium.
Opportunities under the Dietary Supplement Health and Education Act (DSHEA) are less defined. Health related claims for dietary supplements do not need significant scientific agreement like foods under NLEA. But, supplements in food form cannot be marketed as foods. As a result, some have taken a different tact in the development of functional foods. Many companies, without making any claims, are playing on the “perceived” health benefits of ingredients that many consumers hold to be true; i.e., cranberries help urinary tract infections.
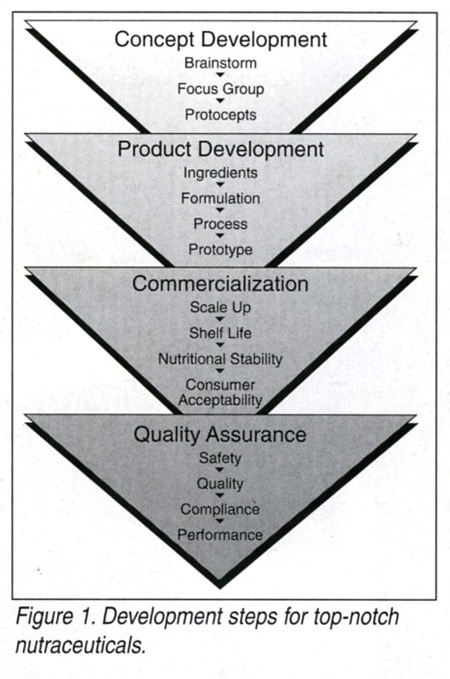
Whether a food, pharmaceutical or dietary supplement company is looking to introduce nutraceuticals that make specific health claims or simply take advantage of ingredients that consumer’s believe have perceived health benefits, the road can be quite challenging. To leverage the major opportunities these markets present, expert guidance is often needed in handling the product development, commercialization and quality assurance for these products. Here’s a brief primer on what goes into creating top-notch functional foods.
Product Development
The first critical phase in formulating a nutraceutical is finding the right ingredients. The regulatory status and quality of functional ingredients may be critical in relation to its actual function. Take herbals, for instance, where a health claim is tied to a specific ingredient Regulatory compliance as a food ingredient plus the qualification and quantification of marker compounds in the herbal extract that relate to the functionality must be validated. Coordination with the ingredient supplier for the appropriate analytical methodology and the ultimate substantiation of the functional compounds are necessary to support certificates of analysis (COAs) that must accompany each ingredient shipment. The same approach should be followed for functional foods that utilize nutrient fortification ingredients. Analysis of the actives and valid COAs will assure that the level of the nutrient(s) claimed on the label is in compliance.
Understanding the processing of ingredients is also important in the development of nutraceuticals. For example, soy protein has become one of the most popular ingredients thanks to the soy protein health claim approved in October 1999. This claim allows products that contain 6.25 grams of soy protein per sewing and are low in fat and cholesterol to make the heart healthy claim. As a result, there has been a rush to increase the amount of soy protein— an ingredient whose quality has been greatly improved over the past five years—in product formulations. However, as discussed below, there are many different options when it comes to adding soy protein to a new or existing product.
The first goal is to maximize ingredient delivery. A formulator should identify a source that can deliver the highest level of the active component and can minimize an off-note in the finished product. For example, a soy protein isolate, soy protein concentrate, and soy flour may all be possible ingredient options. Of these three options, a soy protein isolate delivers the highest level of protein, which is a minimum of 90% protein on a moisture-free basis. Soy protein isolate is commonly manufactured by water extraction from defatted and dehulled soybeans. Compare this to a soy protein concentrate that is at least 65% protein on a moisture-free basis and is manufactured by the removal of a portion of carbohydrates from defatted and dehulled soybeans usually by alcohol extraction. This results in a loss in isoflavones. Finally, soy flour contains approximately 50% soy protein on a dry weight basis and is the result of grinding a whole, dehulled soybean into a fine powder. Therefore, the fat naturally occurring in the soybean is present in the flour. Each will behave differently in a finished product that contains approximately seven grams of soy protein in one serving. Ultimately, the formulator must utilize the protein source that delivers the required level of protein while providing desirable product attributes.
This is also true when formulating products that contain botanicals. A concentrated form like an extract will reduce the amount necessary to deliver the desired dosage. The more natural form (powder) requires a higher level in the finished product resulting in a greater off-note. Being well versed on the methods of manufacturing with the majority of ingredients will assist in identifying problem areas and present a variety of solutions.
Once the ingredients are in place, the actual formulation and development process begins. The key issues here are feasibility, compatibility, stability, acceptability and financial viability. In addition to the appropriate physical, chemical or microbiological assessments that are made during the process, the overall sensory quality of the product must also be confirmed. As initial prototypes are developed, they should be benchmarked with existing products in the marketplace to obtain consumer feedback. Then, final prototypes can be optimized to deliver an acceptable and profitable functional food.
Commercialization
Commercialization takes the product from its first pilot run through to manufacturing. During this phase, the product’s shelf life and stability will be put to the final test. Through innovative formulation with ingredients, and the proper use of process and package, products can be developed to meet the shelf life requirements of the appropriate distribution system. The validation of the stability of the functional ingredients in the product is a significant part of this protocol. Other issues include establishing standard operating procedures (SOPs), product specifications and distribution channels.
 It is important to note that throughout the development process, careful assessment is necessary to determine how the product complies with the regulatory requirements in regards to the claims made, to ingredients, processes, packaging and labeling. Functional foods that make label or advertising claims will be subject to regulatory compliance based on the nature of the claim. Under NLEA, there are well- defined health claims that can be made about foods. Under DSHEA, health and wellness claims can be made with appropriate scientific substantiation in the product literature. Once this type of claim is made, however, the product is no longer a food, but a dietary supplement. A remedy or cure claim makes the product a pharmaceutical, which dictates compliance to Pharmaceutical cGMPs and specific monographs for active ingredients. A good example of this is a menthol flavored hard candy with no label claims, which is a food. Add zinc and echinacea, and make a wellness claim and the product is a dietary supplement. Take the same product and make an antitussive claim (a cough drop) and the product is a drug.
It is important to note that throughout the development process, careful assessment is necessary to determine how the product complies with the regulatory requirements in regards to the claims made, to ingredients, processes, packaging and labeling. Functional foods that make label or advertising claims will be subject to regulatory compliance based on the nature of the claim. Under NLEA, there are well- defined health claims that can be made about foods. Under DSHEA, health and wellness claims can be made with appropriate scientific substantiation in the product literature. Once this type of claim is made, however, the product is no longer a food, but a dietary supplement. A remedy or cure claim makes the product a pharmaceutical, which dictates compliance to Pharmaceutical cGMPs and specific monographs for active ingredients. A good example of this is a menthol flavored hard candy with no label claims, which is a food. Add zinc and echinacea, and make a wellness claim and the product is a dietary supplement. Take the same product and make an antitussive claim (a cough drop) and the product is a drug.
Quality Assurance
Ongoing quality assurance of your product includes testing for safety, regulatory compliance, quality and performance. Ingredient suppliers should be prequalified by auditing and analytical validation of critical attributes prior to accepting ingredients just on the submission of certificates of analysis. In- process and finished product specifications must be established and continuously monitored. Product stability and expiry dating must be confirmed by the appropriate shelf life studies.
Nutraceuticals Are Here to Stay
With studies showing that nine out of 10 consumers prefer naturally nutritious foods to dietary supplements, it’s a sure bet that functional foods are going to continue to dominate the market. There are many success stories already leading the way. Tropicana has seen sales grow 173% since launching its Pure Premium juice line with calcium fortification. General Mills “Kix” breakfast cereal has seen a 17% sales increase as a result of its nutrient fortification. Gardenburger saw a 25% sales increase and a 44% distribution increase in the two months following the FDA’s allowable claim that let it call-out the benefits of the soy protein it uses in its product.
The opportunities are endless for companies that want to develop new products for the growing functional food categories. The bottom line is creating products that are safe, feasible to manufacture, stable in their intended packages, in compliance with government regulations and acceptable to the consumer.
Philip Katz is president of Shuster Laboratories Inc., a leading independent R&D firm and testing laboratory which provides comprehensive product formulation, product quality and consumer acceptability services to food, pharmaceutical and dietary supplement companies interested in developing and introducing functional foods into the marketplace. Shuster is based in Canton, MA.
Top-Notch Nutraceuticals



