Gluten-free has evolved into the hottest new food marketing trend in the U.S. An increasing number of food companies are now entering products into this category, and the gluten-free trend has enveloped restaurant foods, cosmetics, personal care products, beer and other alcoholic beverages.
Historically, gluten-free foods were available only from a small number of specialty foods companies and were relegated in very limited number to some obscure corner of the supermarket or obtained via mail order. Originally, gluten-free foods were developed for consumers with celiac disease, an intestinal inflammatory disease that leads to nutrient malabsorption if untreated. Celiac disease is triggered in affected individuals by consumption of gluten proteins from certain grains (wheat, rye, barley and related grains). For the affected consumer, the only effective preventive measure is the avoidance of dietary gluten. Celiac disease cannot be cured pharmacologically and is never outgrown. Since wheat (and, to a lesser extent, barley) is such a common component of food formulations, the initial gluten-free products used various strategies to substitute gluten-free ingredients for wheat-derived ingredients. Often, the quality of gluten-free products was poor by comparison with the gluten-containing counterpart. Today, the gluten-free label is attached to a much wider variety of food products, including those that are inherently gluten-free. The quality of gluten-free products has also improved considerably in recent years.
Is gluten-free just a marketing fad that could disappear in a few years? Diagnostic testing for celiac disease has improved in recent years, leading to the recognition that an increasing number of individuals have celiac disease. As many as 1 in every 133 individuals in the U.S. may have some susceptibility to the development of celiac disease, although probably fewer than 1 in 1,000 individuals suffer adverse effects upon ingestion of gluten. Another form of gluten sensitivity has become clinically recognized in recent years, but the prevalence of this condition is unknown and few consumers currently have this diagnosis. Individuals with allergies to wheat and barley may also follow gluten-free diets, but the prevalence of wheat and barley allergies is likely less than 0.1 percent. Thus, the popularity of gluten-free foods currently seems to exceed the number of individuals with a clinical diagnosis indicating that they should avoid gluten. The consumption patterns of individuals who voluntarily select gluten-free foods have not been carefully evaluated. These consumers may not always choose gluten-free for every eating occasion, whereas those consumers with celiac disease or some other clinical diagnosis must avoid gluten continuously. Will these consumers who voluntarily select gluten-free products persist with this eating preference? No one can predict at the moment. Suffice it to say that the gluten-free trend continues unabated at present. This recent popularity of gluten-free foods has induced many companies to develop and market gluten-free products.
Although consumers who have no need to completely avoid gluten may indeed purchase gluten-free foods, product developers must always remember that consumers with celiac disease or some other clinical affliction depend on these products. Thus, gluten-free foods must be made with these patients in mind. Celiac sufferers must adhere to a gluten-free diet throughout their lifetime. The availability of gluten-free foods is critical to an effective avoidance diet because gluten-containing grains and ingredients are so ubiquitous in processed food products.
Defining “Gluten-Free”
The definition of “gluten-free” is central to any discussion of gluten-free foods or gluten-free ingredients. The U.S. Food and Drug Administration (FDA) has recently defined “gluten-free” as less than 20 ppm (mg/kg) of gluten. This same definition holds in other countries as well. Some countries separately have established a category of low-gluten foods that are defined as less than 100 ppm gluten. U.S. regulations, however, don’t recognize low-gluten foods. The new U.S. gluten-free regulation also establishes several other conditions that must be met by any food labeled gluten-free:
• The food must not contain any ingredient that is a gluten-containing grain.
• The food may only contain an ingredient derived from a gluten-containing source if that ingredient has been processed in a manner to remove gluten residues to a level of less than 20 ppm.
From a clinical perspective, the ingestion of gluten-free foods containing less than 20 ppm gluten appears to be safe for celiac sufferers.
The enforcement of a regulatory gluten-free definition requires the application of test methods capable of detecting gluten residues at levels of 20 ppm or below. Currently, quantitative enzyme-linked immunosorbent assays (ELISAs) are the favored methods for the detection of residual gluten in foods or food ingredients. Gluten ELISA kits are available from several companies. ELISA kits use antibodies that specifically recognize gluten present in extracts of the food sampled for analysis. The commercial ELISA kits rely upon several distinct types of antibodies, including the R5 antibody, the G12 antibody and so-called Skerritt antibody. Analytical results may vary to some extent depending upon which ELISA kit is used for the analysis. However, the R5 and G12 antibody-based kits in particular are considered reliable for the detection of gluten residues at the levels needed to ensure the gluten-free status of food products.
Grain Sources of Gluten
In the development of gluten-free foods, grain sources of gluten and ingredients derived from those grains must be avoided. Celiac disease is triggered by exposure to the prolamin proteins of wheat (gliadin), barley (hordein), rye (secalin) and related grains, such as durum, spelt, Kamut®, emmer, einkorn, farro and triticale. While all other foods could be considered naturally gluten-free, other grains in particular can become contaminated with gluten-bearing grains because of shared agricultural fields and shared harvesting, transporting and storage systems. In particular, oats are often contaminated with wheat, barley or both. The contamination of oats with wheat is particularly high because these two grains are grown in the same regions. Clinical research has documented that ingestion of uncontaminated oats is safe for most individuals with celiac disease, although some debate remains that a small percentage of celiac sufferers might be adversely affected by eating oats. Despite the data indicating that oats are safe, most gluten-free diets still espouse avoidance of oats because of the likelihood of contamination with wheat or other gluten-containing grains.
Gluten-Free Grains
Gluten-free oats have entered the marketplace in recent years. These oats must be grown from highly pure foundation-type seed in isolated plots and then harvested, transported and stored in a manner that does not allow contamination. The mainstays of a gluten-free diet are other grains considered inherently gluten-free, including corn (maize), rice, sorghum, buckwheat, millets, teff, amaranth and quinoa. These other grains can occasionally be contaminated with gluten-containing grains, so it is important to ensure the gluten-free status of any alternate grain intended for use in gluten-free products. It may be wise to analyze individual shipments of these grains for residual gluten to ensure that they are truly free of gluten. The risk of cross-contamination with gluten-containing grains is lowest for rice, amaranth and quinoa because these grains are typically grown in areas where gluten-containing grains are not grown.
Ingredients Derived from Gluten Sources
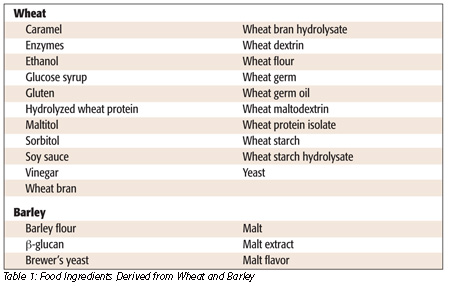 Numerous food ingredients are derived from gluten-containing grains, especially wheat and barley (Table 1). While Table 1 provides a list of ingredients that may be derived from wheat and barley, some of these ingredients occasionally may also be obtained from other sources. Due to their high gluten content, several of these ingredients are clearly hazardous for celiac sufferers, including wheat protein isolate and probably wheat bran. Other wheat- and barley-derived ingredients may have been rendered gluten-free by the process used to manufacture the ingredients. Assessing the gluten content of ingredients derived from gluten sources is obviously critical in the development of gluten-free foods. However, the appropriateness of the analytical method is a key concern in the analysis of some wheat- and barley-derived ingredients.
Numerous food ingredients are derived from gluten-containing grains, especially wheat and barley (Table 1). While Table 1 provides a list of ingredients that may be derived from wheat and barley, some of these ingredients occasionally may also be obtained from other sources. Due to their high gluten content, several of these ingredients are clearly hazardous for celiac sufferers, including wheat protein isolate and probably wheat bran. Other wheat- and barley-derived ingredients may have been rendered gluten-free by the process used to manufacture the ingredients. Assessing the gluten content of ingredients derived from gluten sources is obviously critical in the development of gluten-free foods. However, the appropriateness of the analytical method is a key concern in the analysis of some wheat- and barley-derived ingredients.
Wheat starch is actually a common ingredient in gluten-free foods. Wheat starch sometimes contains residual levels of detectable gluten even though most of the wheat protein has been removed during manufacturing. Wheat starch can be successfully incorporated into gluten-free foods if care is taken to ensure that it has sufficiently low (<20 ppm) gluten content. Analytical verification is recommended for every lot of wheat starch intended for use in gluten-free products.
Wheat starch hydrolysates are glucose syrups derived from wheat, but glucose syrups can also be derived from corn. Corn-based glucose syrup should be inherently gluten-free and is the most widely used source of glucose syrup in the U.S. Wheat-derived glucose syrup is more widely used in the EU and Australia. Wheat-derived glucose syrup can be manufactured with quite low residual levels of gluten, and if gluten levels are low enough, the syrup can be used in gluten-free foods. Many other ingredients are made from glucose syrup, including sugar alcohols (e.g., sorbitol, maltitol) and caramel. These ingredients typically contain no detectable gluten and can be used in the formulation of gluten-free foods. Analytical verification of the gluten-free status of glucose syrup from wheat is recommended.
Maltodextrin can also be obtained from either wheat or corn. With maltodextrin, the starch is less fully hydrolyzed than in the case of glucose syrup. Maltodextrins typically contain very low levels of detectable gluten and can be used as ingredients in gluten-free foods if that is the case. Corn maltodextrin should be gluten-free. Analytical verification of the gluten-free status of wheat-derived maltodextrin is recommended.
Wheat bran hydrolysate is another distinct product in which the residual carbohydrate is partially hydrolyzed. This ingredient might contain detectable levels of gluten depending upon the gluten levels in the wheat bran used to make it. While wheat bran hydrolysate is not a common food ingredient, its use in gluten-free formulations is not advisable.
The gluten fraction of wheat can also be used to produce protein hydrolysates. However, protein hydrolysates may also be made from many other nongluten sources, including milk, soy, corn and peanut. Hydrolysates have varying degrees of hydrolysis—the percentage of amino acids that are broken. The detection of residual gluten peptides in these ingredients is difficult since the ELISA methods mainly detect intact protein. The safety of wheat protein hydrolysates for celiac sufferers is variable and depends upon the degree of hydrolysis. Extensively hydrolyzed wheat protein (>0.62 amino nitrogen-to-total nitrogen ratio) is likely safe, although this has not been clinically proven. However, some wheat protein hydrolysates would be less completely hydrolyzed and thus are not recommended for individuals on gluten-free diets. Because the labeling of wheat protein hydrolysates does not reveal the degree of hydrolysis, celiac sufferers should avoid these ingredients. Furthermore, in the U.S., FDA may not allow gluten-free labeling on fermented or hydrolyzed ingredients/products derived from gluten-containing grains; according to the recently released final rule, FDA indicates that it will exercise regulatory discretion on the use of gluten-free labeling of such ingredients/products, that additional rulemaking is forthcoming and that it is the manufacturer’s responsibility to ensure that a gluten-free claim is valid even when methods to reliably detect gluten peptides may not exist. Thus, considerable caution should be exercised in using gluten-free claims on such ingredients/products. Hydrolysates from other sources (e.g., soy, corn) are effective substitutes.
With soy sauce, a naturally fermented product obtained by fermentation of a mixture of soybeans and wheat, the proteins are extensively hydrolyzed. Gluten ELISA methods are not reliable for the analysis of soy sauce because these methods can only detect intact gluten. Gluten-derived peptides may still exist in soy sauce, although this possibility has not been studied. Since the safety of soy sauce for celiac sufferers is not proven, many celiac sufferers are likely to avoid this ingredient.
Ethanol (ethyl alcohol) is derived commercially from either wheat flour or cornstarch by fermentation followed by distillation. Food-grade ethanol can be used as a carrier for flavors or other ingredients. Wheat-derived ethanol does not typically contain detectable residual gluten. Since ethanol is distilled, the distillation process should serve to eliminate any residual protein or peptides. Ethanol can be used in gluten-free product formulations.
Vinegar is another product of fermentation that can be derived from wheat or malt (barley). Wheat-derived distilled vinegar does not typically contain detectable gluten residues. Again, distillation likely prevents the occurrence of gluten or gluten peptides in distilled vinegar. Distilled vinegar can likely be used in gluten-free products, although some celiac sufferers avoid it in an abundance of caution. Avoidance of malt vinegars is more commonly practiced.
Wheat flour is sometimes used as a substrate for fermentations used to produce various food ingredients, including enzymes, bacterial cultures and yeast. Because the products are macromolecular, the separation of these products from any residual gluten is difficult. The analysis of gluten in such products by ELISA can be compromised if the fermentation involves proteolysis. In some cases, detectable gluten is found in such products. With enzymes, the very low usage levels would result in very low levels of gluten acceptable in gluten-free products. Bacterial cultures and yeast, where usage levels could be higher, should be obtained from fermentations that do not use wheat or barley, or should be avoided in gluten-free product formulations. Malt and malt extracts are derived from sprouting barley. Some proteolysis of gluten likely occurs during this process, but detectable levels of gluten are often found in malt or malt extracts. Some malt extracts contain no detectable intact gluten. However, most celiac sufferers avoid malt or malt extracts because of the frequent presence of gluten residues. Malt or malt extracts are not advisable in gluten-free foods.
The soluble fiber fraction of barley bran (β-glucan) has desirable nutritional properties. The level of gluten in the β-glucan fraction may depend upon its level of purification. Unless documented to contain no detectable gluten, the use of β-glucan in the formulation of gluten-free products is not recommended.
Detection of Gluten Residues
While many ingredients are derived from wheat and barley, some ingredients such as wheat starch can be rendered gluten-free during manufacturing. The analytical verification of gluten-free claims on all ingredients derived from gluten-containing grains is required to ensure compliance with the FDA gluten-free regulation. In fact, analysis for gluten residues is recommended for all ingredients used in gluten-free products.
The detection of gluten residues in other grains is straightforward by ELISA. Because of the frequency of contamination of other grains with gluten-bearing grains, the confirmation of the gluten-free status of all ingredients derived from grain sources is recommended. Other agricultural commodities such as legumes, including soybeans, may also be contaminated with gluten because of the commingling of commodities in storage facilities. Since commodity contamination can be quite significant and variable, successful manufacturing of gluten-free products relies upon regular testing for gluten residues.
The analysis of residual gluten levels in food ingredients derived from gluten sources can be much more challenging. ELISAs detect intact gluten or particular peptide epitopes derived from gluten. Some food ingredients contain hydrolyzed or partially hydrolyzed gluten that may not be detected in various ELISAs. Such gluten fragments might still be hazardous if they contain toxic gluten peptides capable of eliciting celiac disease. Ingredients derived by fermentation may also be challenging to analyze for gluten for similar reasons. If a gluten-containing material, for example, wheat flour, is used as a substrate for fermentation, gluten residues may remain in the final product. However, if the fermenting microorganism is proteolytic, then the concern would be for gluten peptides that are undetectable by most ELISA methods. As noted, partially hydrolyzed gluten may remain hazardous for celiac sufferers. Competitive ELISA methods can be used to detect the presence of gluten peptides in hydrolysates and fermented ingredients.
In formulating gluten-free foods, the most obvious choice is to use components that are naturally free of gluten where possible. Of course, it’s prudent also to confirm the gluten-free status of such components by analysis. Substitutes should be sought for common ingredients derived from gluten-containing grains. Still, ingredients like wheat starch can be effectively used in the formulation of gluten-free foods if care is taken to ensure the gluten-free status of such ingredients. And some other wheat-derived ingredients such as vinegar and ethanol are gluten-free because of the processes used to make them.
 Steve L. Taylor, Ph.D., is professor and codirector of the Food Allergy Research and Resource Program at the University of Nebraska. He performs active research on food allergies, including development and improvement of detection methods, assessment of effects of processing, and threshold doses and risk assessment. He maintains an active food industry outreach program in the area of food allergies, including confidential testing services and workshops, training and webinars.
Steve L. Taylor, Ph.D., is professor and codirector of the Food Allergy Research and Resource Program at the University of Nebraska. He performs active research on food allergies, including development and improvement of detection methods, assessment of effects of processing, and threshold doses and risk assessment. He maintains an active food industry outreach program in the area of food allergies, including confidential testing services and workshops, training and webinars.
 Joseph L. Baumert, Ph.D., currently serves as an assistant professor in the department of food science & technology and as codirector of the Food Allergy Research and Resource Program at the University of Nebraska. He received his Ph.D. in food science and technology at the University of Nebraska, Lincoln, specializing in food allergy research in association with the Food Allergy Research and Resource Program.
Joseph L. Baumert, Ph.D., currently serves as an assistant professor in the department of food science & technology and as codirector of the Food Allergy Research and Resource Program at the University of Nebraska. He received his Ph.D. in food science and technology at the University of Nebraska, Lincoln, specializing in food allergy research in association with the Food Allergy Research and Resource Program.
Achieving Gluten-Free Status




