Allergen reactions cause a plethora of symptoms and are the result of an elegant and systematic cascade of immunological responses. Involving the immune system distinguishes an allergen from a food intolerance, which is not mediated by the immune system but the result of an enzyme deficiency. Unlike food allergies where avoidance is the most effective remedy, food sensitivities will allow for small portions to be ingested.
As a result of the complexity of allergen nomenclature and ingredients containing allergens, labeling laws have been enacted to help consumers understand whether a pre-packaged food contains an allergen. To assist with labeling laws and avoidance of potential allergen cross-contamination events, plant-based allergen control programs are an effective measure if managed well. However, as with other food safety control programs, allergen control programs have taken on a life of their own.
When allergen control was in its infancy, measures to mitigate cross-contamination were less complicated. The complexity has been introduced with the addition of controlling sensitizing agents and increased use of international ingredient sourcing. While some countries such as Japan have strict labeling laws, other countries such as China do not, making knowledge of ingredient composition difficult at best. Therefore, the training of purchasing agents and product developers on allergens or how to formulate without the use of allergens should be enacted to help these employees and their departments understand their part in preventing allergen cross-contamination. A minimum requirement for allergen control is the understanding and implementation of the Food Allergen Labeling and Consumer Protection Act (FALCPA). Also essential are thorough, in-house allergen control programs that are managed well on many departmental levels.
Food Allergies versus Food Sensitivities
It is important to understand the difference between a food allergy, which involves the immune system, and food intolerance, which is generally caused by an enzyme deficiency and does not involve the immune system. Food intolerance enzyme deficiencies typically result in the inability of the body to break down carbohydrates (sugars) such as lactose, fructose or sorbitol. Those who have a lactase deficiency may experience symptoms when milk or unfermented dairy products are ingested due to their inability to break down the milk sugar, lactose. In fermented dairy products, the fermentation process will reduce the concentration of lactose. Diet foods often contain non-digestible sugars such as sorbitol that will cause symptoms because the sugars are unable to be metabolized until they reach the distal small and/or proximal large intestine. When the sugars reach these areas of the intestines, the bacterial flora within these areas are able, during their growth processes, to catabolize the sugars. The byproducts of the bacterial growth—gas (carbon dioxide), acid and toxins that cause diarrhea—may be produced. Hence, the symptoms of lactose or other food intolerance will include cramping, bloating, gas and diarrhea.
How Allergens Cause Adverse Reactions
Non-specific versus specific host defense mechanisms
Allergenic reactions are due to an inappropriate immunological response to an otherwise harmless food.
The body has various defense mechanisms used to combat invaders such as viruses, bacteria or other substances it views as foreign. Any item that is non-self is termed an “antigen.” There are two main types of defense mechanisms to antigens: non-specific and specific.
Non-specific defense mechanisms are fever, inflammation (swelling, redness, pain and increased temperature at the affected area), chemical barriers (saliva, mucus, bile and gastric juices), skin and mucous membranes.
Specific defenses involve the production of antibodies in response to a foreign substance. A specific antibody is produced to attack a specific antigen and is capable of binding to and removing the antigen. There are five classes of antibodies: IgG (crosses the placenta from mother to child), IgA (tears, saliva, mucus and tissue linings), IgM (first antibody to the site when a new foreign invader is found), IgE (associated with allergic reactions) and IgD (found in membranes of immune cells). Antibodies will bind to an epitope (a short amino acid sequence on the antigen that is capable of eliciting an immune response). The amino acid sequences of many allergens are available within the scientific literature.
An allergenic reaction is a reaction between an antigen and antibody. In a sensitization step, the first time the body is exposed to a foreign substance, antibodies are produced. Typically, this will take from 5-15 days, and the amount of antibodies produced is low. However, the immune system has an elegant feature that is paramount to its success: memory. Thus, the second exposure to the same antigen elicits the production of antibodies in a much more rapid time frame and in a higher concentration compared with the first exposure. These antibodies are produced with the short, specific amino acid sequence found on the antigen so that binding and subsequent removal of the antigen are performed.
It is the memory of the immune system that is responsible for vaccine efficacy. Vaccines are used as the sensitizing step. Thus, when a person is exposed to the same antigen after a vaccine or initial exposure, the body’s own immune system will mount a rapid and vigorous attack. Additionally, each time the body is exposed to the same antigen, the immune response has the ability to induce more and more aggressive attacks. Thus, in most cases, a healthy adult’s immune system can fight off most foreign attacks time and time again. However, the ability to fight off disease is modulated by age (immune function of the very young and elderly is impaired), race, genetics, current health status and lifestyle (diet, smoking habits, amount of exercise and sleep).
Mast cell-IgE-antigen complex
In an allergenic reaction, the body mistakenly views an ingested protein of a food as an antigen and the immune system hosts an immune reaction by the production of IgE antibodies, which will bind to the antigen and a mast cell, forming a complex. A mast cell is part of the immune cellular network, and when the antigen-antibody-mast cell complex is formed, the release of mast cell content will occur. The mast cell contains histamine, the biologically active amine that is formed by the decarboxylation of the amino acid histidine. When released by mast cells, it causes a number of activities, such as stimulating hydrochloric acid secretion in the stomach; vasodilation, which results in inflammation and leaking of fluid into tissues; bronchoconstriction, which restricts airways; the lowering of blood pressure; itching; and rashes. An immediate hypersensitivity reaction by IgE-antigen-mast cell complex formation will cause one of two types of attacks: systemic or localized. Either reaction may occur within minutes or up to a few hours after initial sensitization and subsequent ingestion of the offending foods. Systemic reactions result in vomiting, abdominal cramps, diarrhea, respiratory distress and, in severe cases, anaphylactic shock. Localized reactions center on the skin and result in rashes, hives, eczema and itching. A delayed hypersensitivity reaction may also occur greater than eight hours after ingestion, involving cellular immunity (T-lymphocytes and macrophages).
Cross-reactions
Of interest, there are documented allergic-like symptoms that arise due to cross-reactions of food and non-food allergens. Thus, when one substance is encountered, a cross-reaction occurs, causing adverse reactions. Documented cross-reactions include the following:
• Bananas and latex
• Ragweed and watermelon, cantaloupe, honeydew and bananas
• Birch pollen and hazelnuts
Common sites for allergic reactions
Common sites for allergic reactions are those areas in which mast cells are located and concentrated, such as the following:
• Mouth: swelling of the lips or tongue
• Airways: breathing restrictions, asthma, rhinitis
• Digestive tract: nausea, vomiting, diarrhea
• Skin: hives, rash, itching
Medications
There are a few medications to help reduce or eliminate the symptoms of allergies. Antihistamines block the binding of histamine to receptors on target cells and prevent effects. Epinephrine is used for severe systemic reactions; it relaxes smooth muscles, stimulates the heart and constricts blood vessels. Sodium cromolyn prevents mast cells from releasing histamines. However, for food allergies, the best defense is to practice total avoidance of ingesting the allergenic food. In that regard, the FALCPA has evolved to help those with total avoidance by facilitating allergen identification on the label declaration.
FALCPA
The FALCPA applies to packaged foods regardless of whether the food is domestically produced or imported. Within the United States, the FALCPA is preemptive of local or state laws regarding allergens. Raw agricultural commodities, except for soy lecithin, are exempt from the FALCPA. Highly refined oils are exempt from the FALCPA based on the method by which the oils are produced. The process of oil refining—specifically deodorization, decoloration and dehydrogenation—results in the destruction of the allergenic proteins within the product. Since all allergens are proteins, oil production processes eliminate the allergenicity of the oil even if the raw ingredient from which the oil comes is an allergen. To note, although soy lecithin is also subjected to these processing steps and has not been shown to contain soy proteins, no federal ruling has made it exempt from the FALCPA. FALCPA labeling requirements also do not apply to foods placed in a wrapper or container in response to a consumer’s order, such as the paper or box used to provide a sandwich ordered by a consumer.
Two labeling options are available for allergenic ingredients: the word “Contains” followed by the name of the food allergen or a parenthetical statement within the list of ingredients. Raw ingredients containing allergens, in any concentration, must be listed if present. When an allergen is in the food that is contained within an allergenic group such as “tree nuts” or “seafood,” the specific nutmeat or seafood must be listed. This is based on the fact that some people may be allergic to one tree nut or seafood item but not all within the grouping. Thus, an allergic reaction may be triggered by hazelnuts but not pecans, or shrimp but not lobster.
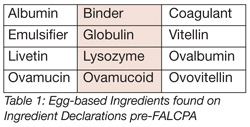 All information must be in the same font as the rest of the ingredient label. While more than 160 foods cause allergic reactions, 90% of these reactions are caused by the “top 8” (i.e., tree nuts, peanuts, soy, egg, milk, fish, wheat and shellfish) and must be listed in “plain language” on the ingredient declaration. The FALCPA ruling was designed to help consumers, particularly those who are young, to better understand the potential for allergenic ingredients in the products they are purchasing and be able to make more informed choices. Any young consumer should be able to understand the ingredients contained within a product. Table 1 lists examples of words used on ingredient declarations to describe egg products prior to implementation of the FALCPA.
All information must be in the same font as the rest of the ingredient label. While more than 160 foods cause allergic reactions, 90% of these reactions are caused by the “top 8” (i.e., tree nuts, peanuts, soy, egg, milk, fish, wheat and shellfish) and must be listed in “plain language” on the ingredient declaration. The FALCPA ruling was designed to help consumers, particularly those who are young, to better understand the potential for allergenic ingredients in the products they are purchasing and be able to make more informed choices. Any young consumer should be able to understand the ingredients contained within a product. Table 1 lists examples of words used on ingredient declarations to describe egg products prior to implementation of the FALCPA.
Exemptions to the FALCPA
Since food allergens are mainly proteins, a request can be made for an exemption to the FALCPA labeling requirement if the food produced does not contain allergenic proteins. To demonstrate the absence of allergenic proteins, test results are needed, indicating the protein is not present to the test sensitivity level. Current allergen test-kit sensitivity levels are 0.1 to 5 ppm protein. Test kits are commercially available for milk, eggs, almonds, peanuts, hazelnuts and soy flour. The remaining allergen testing can be performed by organizations, such as the Food Allergen Research and Resource Program, University of Nebraska (www.farrp.org). Sample submission requires a representative food sample and a positive control sample (sample with known allergen in question).
International Food Allergen Legislation
European food allergen legislation requires listing 12 potentially allergenic ingredients on prepackaged foods. The European Union (EU) considers the following ingredients allergenic: cereals containing gluten, fish, crustaceans, eggs, peanuts, soy, milk and dairy products, nuts, celery, mustard, sesame seeds and sulfites. The legislation requires clear labeling and information about the ingredients made from these allergens.
Similarly, Australia and New Zealand have developed a system of voluntary incidental trace allergen labeling (VITAL). This is a risk-based method for use by food manufacturers to assess allergen cross-contact. The VITAL allergen action-level grid (“Vital Grid”) was developed to determine the labeling of an allergen. Action levels are as follows: Green Zone, Action Level 1: Allergen does not need advisory labeling. Yellow Zone, Action Level 2: Allergen advisory labeling stating allergen may be present. Red Zone, Action Level 3: Significant levels of the allergen are likely, listing is advised. These action levels give manufacturers a determined level for advisory labeling.
Japan mandates the labeling of the following: eggs, milk, wheat, buckwheat and peanuts. When possible, the following 19 foods should be labeled: abalone, squid, salmon roe, shrimp/prawn, oranges, crab, kiwifruit, beef, tree nuts, salmon, mackerel, soybeans, chicken (poultry), pork, mushrooms, peaches, yams, apples and gelatin. A label of “May contain allergens” is prohibited for any product. Instead, information about the production line may be included if cross-contamination is a concern, for example, “The production plant creates products containing allergen” or “Equipment using allergen have been used in the process of production.”
China and Southeast Asia appear not to have allergen management systems in place. This places the burden of allergen testing on the importing customer.
Allergen Control Programs
In a food plant, when trying to remove the allergen from processing equipment to avoid cross-contamination, it is the cleaning step that is critical for efficacy. This is unlike sanitization being the critical step when trying to eliminate bacteria. Heating, sanitizing, irradiation or acidity will not eliminate the allergenicity of the food. Because the antibody will bind to a specific amino acid sequence, heating, sanitizers or stomach acid exposure to the allergenic proteins have not demonstrated destruction of the amino acid sequence and, therefore, the allergenicity remains.
Of interest, the FALCPA does not apply to meat, poultry or egg products that are regulated by the USDA. However, processors regulated by the USDA are to include allergen information in their HACCP plans and control measures for allergen cross-contamination avoidance during storage, production and cleaning.
Whether regulated by the USDA or the FDA, all allergen control practices should be identified in a written set of allergen-control program standard operating procedures and documentation of execution that are available for review. The allergen control program begins with a label development procedure. A checklist should be developed that is filled out during label development and is to include the rules of the FALCPA. Proof of labels and label receiving into plants need careful review for accuracy. Allergen control programs must include testing of raw materials for allergen-containing ingredients and inspection of labels to ensure that allergens contained within the raw materials are labeled on the finished product. Labeling of raw ingredients to identify allergens is necessary upon receipt and for further ground-level or rack storage of those ingredients. Additionally, multiple products should not be loaded onto the same pallet. Plant traffic flow, such as processing an allergen-containing powder in an area near where a non-allergen-containing product is also being run, must be considered. Color-coding systems for utensils used with allergens, production scheduling, cleaning, use of rework, employee education and production practices are to be reviewed and modified where needed. For example, exchange of employees from one line to another is not acceptable if they do not change their uniforms or at least their aprons and then proceed from an allergen-containing line to a non-allergen-containing line. Process control points should be added to a HACCP plan at the packaging step. The goal is to ensure that the correct label is affixed to the product containing the allergen.
Sensitizing Agents
A sensitizing agent is one that does not cause the plethora of symptoms that allergens do, but an adverse response may be mounted. Sensitizing agents can be confusing in their nomenclature. For example, monosodium glutamate’s (MSG) main component is glutamic acid, which is a sensitizing agent. Labeling of glutamic acid can be difficult to recognize because there are many different names, including hydrolyzed corn gluten, hydrolyzed wheat protein and autolyzed yeast. The word “hydrolyzed” is an indication that the product may contain MSG.
Although sulfites are not considered an allergen listed in the FALCPA, in 1986 the FDA prohibited the use of sulfites on raw fruits and vegetables or those labeled as “fresh.” Sulfites that have been added to food must be declared if added directly. If sulfites are added to the products indirectly, the level must be below 10 ppm in the food product. Sulfites may trigger a reaction in asthmatics.
The color additive FD&C Yellow No. 5 is a commonly used additive in beverages and desserts. This color is on the FDA’s list of permitted colors but has been found to cause an adverse reaction in a small subgroup of the population. An FDA advisory committee in the mid-1980s concluded the adverse reaction was not a true allergenic reaction but rather hypersensitivity. Since the issue of sensitivity was raised, the FDA now requires that labels list all certified colors.
In summary, as opposed to food sensitivities, food allergies are an immune reaction and involve the generation of IgE antibodies. By following the FALCPA, labels indicating the type of allergen allow for a wide range of customers to make informed food purchase decisions such that they avoid experiencing an immune response. This helps those who are allergic to specific foods practice continued avoidance with the goal of thwarting aggressive responses that their immune systems wage. Similarly, following an in-house plant allergen control plan will help to mitigate cross-contamination. Since food allergens cause a high percentage of product recalls, following preplanned activities will help to reduce negative media attention that no one needs.
Kara Baldus, M.B.A., joined TRAC Microbiology, Inc. in August 2005. She is involved in HACCP plan development, research experimental design and data analysis. Kara graduated from the University of Wisconsin-Madison in December 1995 with a double major in Bacteriology and Genetics. She has worked in the food industry as a microbiologist for almost ten years. During that time, she also earned her M.B.A. and Total Quality Graduate Certificate from Edgewood College.
Kristy Werronen is a Junior Consultant for TRAC Microbiology, Inc. She has been part of the TRAC team since April 2006. Previous positions held at TRAC include Scientist and Assistant Laboratory Manager. She graduated from UW-Madison in 2005 with a B.S. in Medical Microbiology and Immunology.
Virginia Deibel, Ph.D., is the CEO of TRAC Microbiology, Inc., a microbiology and chemistry contract laboratory conducting routine product and environmental testing, research, auditing and consulting. She opened TRAC in 2001, where she conducts third-party audits and performs specialized consulting including problem solving, recalls, investigations and expert-witness work. Dr. Deibel earned undergraduate and graduate degrees in Bacteriology and Microbiology from the University of Wisconsin-Madison. She has served as the Chair of the Wisconsin Association of Food Protection; is a member of IAFP, IFT, the American Society of Microbiology and AOAC; and is on the editorial board of Food Safety Magazine.
Adverse Reactions to Allergens: The Importance of Labeling and Cross-contamination Control