In 1997, President Clinton launched the Administration’s Food Safety Initiative. The goal of the initiative is to improve food safety and reduce the incidence of foodborne illness to the greatest extent feasible. While industry has the primary responsibility for the safety of the food it produces and distributes, federal, state and local governments’ roles are to verify that the industry is carrying out its responsibility and to initiate appropriate regulatory action if necessary. The initiative seeks to improve coordination, communication and information exchange among federal, state and local government agencies, and enhance collaboration between the public and private sectors.
As part of the initiative, federal, state and local officials agreed there was an immediate need to improve responses to outbreaks of illness caused by contamination from bacteria, viruses and parasites through better coordination and communication during traceback investigations. A traceback investigation is a method used to determine the source and distribution of the implicated product associated with an outbreak and to identify potential points where contamination could have occurred. This article will describe the U.S. Food and Drug Administration’s (FDA) role in foodborne disease surveillance, FDA’s role in a traceback investigation, what FDA looks for and finds during a farm/source investigation, and some of the challenges facing the FDA while conducting tracebacks of contaminated produce.
Foodborne Disease Investigations
Most of the identified foodborne disease outbreaks in the United States are reported by consumers who suspect an association between a food they have eaten and an illness they are suffering. These individuals report such information to their local health departments. Linking sporadic cases or clusters of reportable diseases such as salmonellosis helps to identify other foodborne illness outbreaks. These include sporadic case surveillance of cases reported by clinical laboratories and physicians at the state and local level, and through FoodNet and PulseNet at the national level. The Foodborne Diseases Active Surveillance Network (FoodNet) is the principal foodborne disease component of the Centers for Disease Control and Prevention (CDC) Emerging Infections Program (EIP). FoodNet is a collaborative project of the CDC, nine state sites (California, Colorado, Connecticut, Georgia, New York, Maryland, Minnesota, Oregon and Tennessee), the U.S. Department of Agriculture (USDA) and the FDA. The project consists of active surveillance for foodborne diseases and related epidemiological studies designed to help public health officials better understand the epidemiology of foodborne diseases in the United States.
PulseNet is a collaborative project between CDC, FDA, USDA and state health departments and uses a national computer network to confirm outbreaks of foodborne illness and to link cases/clusters occurring in multiple states. Public health laboratories across the country perform DNA “fingerprinting” on bacteria that may be foodborne and use the system to exchange findings when outbreaks of foodborne disease occur. The network permits rapid comparison of these “fingerprint” patterns through an electronic database at CDC. The DNA “fingerprinting” method is called pulsed- field gel electrophoresis (PFGE).
FDA becomes involved in an outbreak investigation when surveillance identifies disease clusters or outbreaks and a FDA regulated product is implicated. FDA’s role in foodborne outbreak investigations include:
• Assisting in investigation and coordination in multistate outbreaks
• Reviewing epidemiological, laboratory and environmental data with CDC and state/local agencies
• Providing investigational and laboratory assistance, as needed
• Conducting tracebacks of implicated foods and removal from the market
• Monitoring recalls
• Taking other appropriate regulatory actions
• Identifying how the food became contaminate at its source (such as the farm, in the case of produce)
• Evaluating data from investigation find ing to identify trends and make recommendation to prevent similar problems
Foodborne disease investigations have three components: epidemiological, laboratory and environmental. Epidemiological investigations verify a diagnosis through case interviews and laboratory confirmation; identify the range of onset of symptoms; provide case definitions; conduct epidemiology studies (case control or cohort); and determine statistical associations between eating various foods and becoming ill. Note that this component is not designed to identify the source of contamination. The laboratory component includes analysis of clinical samples, food samples (if leftovers are available) and environmental samples. The environmental component focuses on food preparation methods and the potential for temperature abuse or cross-contamination and the location of preparation. The environmental component also identifies possible modes of contamination at the food’s source. Should the environmental investigation determine that the contamination most likely did not occur at the point of food preparation, then a traceback investigation may be initiated.
The Traceback Process
Public health agencies conduct traceback activities to determine the source and distribution of the implicated product associated with the outbreak and to subsequently identify potential points where contamination could have occurred.
This action helps prevent additional illnesses by providing a foundation for recalls of contaminated food remaining in the marketplace and identifying hazardous practices or violations.
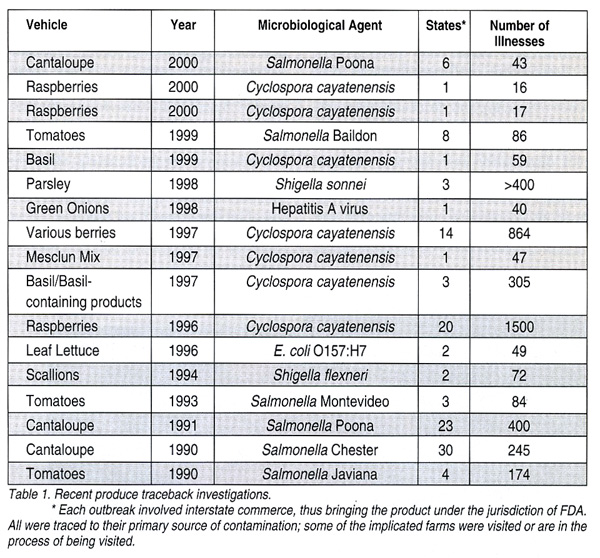 Tracebacks are not a new activity within food safety program models. Reports of tracebacks of shellfish began in the late 1800s; more recently, there have been tracebacks of canned mushrooms in the early 1970s, eggs throughout the 1980s and 1990s, and ground beef and produce in the 1990s. Produce tracebacks have involved Hepatitis A virus in sliced, frozen and sugared strawberries, Shigella in parsley, Cyclospora in raspberries, and E. coli O157:H7 in lettuce. Other examples of outbreaks are shown in Table 1 (Table 1. Recent produce traceback investigations). The primary reason for an outbreak is failure to adequately prevent contamination prior to a food’s consumption. As noted, problems on the farm have included contaminated irrigation, fumigation or processing water, infected workers and contamination by wild or domestic animals. A traceback investigation may result in a recall of product, other regulatory actions such as detention of an imported product, an injunction against a processor or grower, informing the public via press releases, closer monitoring of the product in general, domestic and foreign outreach, and “on-the-farm” investigations.
Tracebacks are not a new activity within food safety program models. Reports of tracebacks of shellfish began in the late 1800s; more recently, there have been tracebacks of canned mushrooms in the early 1970s, eggs throughout the 1980s and 1990s, and ground beef and produce in the 1990s. Produce tracebacks have involved Hepatitis A virus in sliced, frozen and sugared strawberries, Shigella in parsley, Cyclospora in raspberries, and E. coli O157:H7 in lettuce. Other examples of outbreaks are shown in Table 1 (Table 1. Recent produce traceback investigations). The primary reason for an outbreak is failure to adequately prevent contamination prior to a food’s consumption. As noted, problems on the farm have included contaminated irrigation, fumigation or processing water, infected workers and contamination by wild or domestic animals. A traceback investigation may result in a recall of product, other regulatory actions such as detention of an imported product, an injunction against a processor or grower, informing the public via press releases, closer monitoring of the product in general, domestic and foreign outreach, and “on-the-farm” investigations.
Some of the challenges facing FDA in fresh produce tracebacks include the absence of labeling and distribution records, the lack of authority for FDA to require records be kept or provided to FDA, complex distribution systems, and multiple sources of product at the point of service. Another challenge is that traceback investigations are very resource-intensive and may implicate but not confirm the cause of the contamination. While food safety agencies are working to prevent further illnesses through improved coordination and communication during a traceback investigation, the food industry has a crucial role to play. Specifically, their food safety responsibilities are to cooperate with investigators, make records available, provide information about the food (e.g., shelf life and distribution sources), and should the situation warrant, initiate a voluntary recall of an implicated food product.
Information gathered in traceback investigations is used to identify ways to make produce safer. FDA issued a guidance document entitled, “Guidance for Industry: Guide to Minimize Microbial Food Safety Hazards for Fresh Fruits and Vegetables,” in an effort to provide information to growers, packers and shippers to minimize food safety hazards potentially associated with fresh produce. As additional information is learned this guidance document may be revised. Other produce related activities may be found on the FDA website at www.cfsan.fda.gov.
Beyond Tracebacks: The Produce Farm
A farm or “source” investigation occurs after a traceback identifies the farm(s) as the source(s) of an outbreak. Note that a farm investigation is not part of the traceback investigation. Activities are varied and may include developing a map or layout diagram and locating possible sources of contamination, such as the slope of the land and drainage, windblown sources and possible problems that may be caused by flooding or other weather-related sources of contamination. Waste management is a particular concern. For example, authorities look for manure management to determine whether fresh or composted manure was applied and at what point in the growth cycle of the crop.
Authorities check worker sanitation by looking for sewage use or points of exposure to find out about portable toilet use and management and if biosolids are used. Should portable toilets be serviced near the field, contaminating spillage may occur near the crops. FDA has uncovered cases in which no toilets were provided in the fields for workers, or situations in which, though provided, workers failed to use them. While children and infants have accompanied their families to the field, this practice is a problem because children are more likely than adults to be ill or to carry certain foodborne pathogens.
Farm investigators evaluate animal management as a potential source of foodborne pathogens. For example, they find out what domestic animals or wild animals were used in, or had access to, the fields. In a field where produce is growing, the evidence of deer pellets or tracks, or the presence of domestic or work animals, or manure storage adjacent to fields, all present potential microbiological hazards to producing safe produce.
During a farm or source investigation, authorities look at the tools and equipment used in (and in the vicinity of) the harvest to determine what was used and what cleaning, sanitizing and storage practices were followed. For example, FDA has observed the use of nonpotable water to wash equipment. Also, because soil contains many microorganisms of potential concern, a produce harvest knife stuck in the soil should be sanitized prior to continued use. FDA has also observed bare-hand harvesting, a potential source of contamination if the worker is ill. A farm investigation will also examine fumigation equipment for sources of improper chemical contamination. FDA has observed the use of nonpotable water to mix pesticides or wash fumigation equipment.
At the harvest site, FDA investigators check processing and packing operations for microbiological contamination, evaluate equipment construction, cleaning and sanitizing procedures, and determine if adequate chlorine residual and cooling- refrigeration requirements are being met. Observed problems include failure to wash or clean equipment/product, inadequate chlorine residuals in water, and products inadequately cooled or refrigerated.
Inspection of vehicles used on the farm to transport food is another important activity. Because these are a potential source of both chemical and microbiological contamination, FDA checks these vehicles to find out if they are clean and/ or sanitized. Investigations have revealed unclean carts and food products placed on unclean wooden surfaces.
On-the-farm investigations will inspect water sources, for example, to identify irrigation sources. Importantly, this activity reveals whether wells or surface waters are protected from contamination and whether they have been tested for safety. FDA investigations have found contaminated surface water used for spray irrigation, water systems not protected from back-siphonage, and inadequate chlorination of the public water supply.
FDA also looks at worker health and hygiene practices to determine the incidence of disease among workers and worker management practices; for example, whether ill workers are allowed to work and what hygiene training is provided to them. In addition to toilet facilities, inspectors check to see if handwashing facilities are available and convenient and whether workers are, in fact, washing their hands. Inspections have found instances where employees failed to wash hands after leaving the toilet, or ill employees were permitted and encouraged to work.
Facing The Challenges
Federal and state food safety agencies face a number of challenges responding to foodborne illness outbreaks and initiating appropriate traceback investigations. These challenges include the fact that the epidemiology of foodborne disease is changing and new pathogens have emerged, some spreading worldwide. Foodborne illnesses now occurs on a larger scale, with multistate and multinational outbreaks. They are occurring over longer periods of time, are diffuse, and are difficult to detect. In addition, foodborne illness trends are changing due to changes in consumer demands for food that is less processed, the complexity of production/processing and distribution systems, and globalization of the food supply.
Other challenges include the multiple modes of potential contamination that include animals, water and workers spreading contamination to an increasing variety of foods. Increased surveillance and better detection methods are adding to the challenges because of an increase in multistate outbreaks detected and sporadic cases from different states or regions are being linked together requiring FDA’s involvement. FDA continues to work with other agencies in spite of these challenges to improve food safety and reduce the incidence of foodborne illness.
John J. Guzewich RS., M.P.H, is the outbreak coordinator/n FDA’s Center for Food Safety and Applied Nutrition (CFSAN), where he/s the lead in investigating and responding to emergencies involving food products regulated by FDA. He also is the lead for coordinating investigation and response with other federal, as well as state and local agencies, and for developing recommendations on how outbreaks can be prevented in the future. He is past president of the International Association for Food Protection (IAFP).
Peter A. Salsbury is a science policy analyst/n FDA’s Center for Food Safety and Applied Nutrition’s Executive Operations Staff He is responsible for food safety policy issues that include initiatives with FDA’s Office of Regulatory Affairs and activities with other federal, state and local government agencies such as the National Food Safety System project.
Acknowledgments
The authors thank LeeAnne Jackson, Ph.D., John Sanders, D.V.M., Sherri McGarry (CFSAN) and Sarah Pichette (FDA’s Office of Regulatory Affairs) for their assistance on this article.
FDA's Role in Traceback Investigations for Produce




