Several microbial contaminants appear to survive on walls and other interior surfaces of wineries, including those of presses and fermentation tanks, and within wooden barrels.[1] Bulk and bottled wines are often spoiled by fermentative species of Zygosaccharomyces, Dekkera (anamorph Brettanomyces), Saccharomyces, and Saccharomycodes. Dekkera/Brettanomyces is associated with the production of unpleasant medicinal taints, due to its production of tetrahydropyridines and volatile phenolic substances.[2] D./B. bruxellensis, Zygosaccharomyces bailii and Saccharomyces cerevisiae are spoilage yeasts. However, S. cerevisiae appears to be more problematic than indicated, as some strains isolated from dry white wines seem to be more of a potential spoilage yeast than Z. bailii, due to its sorbic acid and sulfite tolerance at high ethanol levels.[3] Both Z. bailii and S. cerevisiae can grow at low pH in the presence of acid concentrations near the legal limits.[4]
Management of these types of spoilage organisms is generally accomplished by following Good Manufacturing Practices and Good Hygiene Practices in the winery.[2] Wine cooperage, although useful for wine aging, has some disadvantages when sanitation must be performed, since the microporous structure of wood purportedly allows the penetration of microorganisms into the wood at depths that make their subsequent eradication a challenge, thereby increasing the risk of wine spoilage when cooperage is reused. Development of undesirable microbiota within cooperage may signi?cantly degrade wine, rendering both the wine and barrels unusable.[5] Certain spoilage microorganisms can survive within barrels, even under “starvation” conditions. In fact, D./B. bruxellensis can survive within the porous wood structures of barrels not filled with wine for weeks or months. Brettanomyces has been isolated from used barrels in the Finger Lakes wine region of New York.[6] Since wood is porous and a source of wood sugars, barrels are difficult to clean and sanitize, and are vectors for Brettanomyces contamination.[7,8]
While the sanitation methods used in wineries are effective on surfaces such as stainless steel, plastic, and glass, sterilization of wood surfaces has proven more difficult. Steam sanitation of wood is a standard method used for wine cooperage. However, the temperatures required to inactivate spoilage microorganisms, and the times required to reach these temperatures within the wood, are poorly understood. Heat and mass transfer in capillary porous materials such as wood has been discussed previously,[9] particularly in regard to the thermal conductivity of wooden materials and the development of thermal inactivation regimes.
Wood is a natural polymer of complex chemical composition and microstructure.[10] Its hygroscopic and porous medium results in heat transfer by conduction, convection, and radiation.[11] Various methods of heat sterilization of wood are currently under investigation as means of killing exotic insects or pathogens within imported goods. One factor being studied is the amount of time (D) required to heat wood of various cross-sectional sizes and configurations to a temperature that will kill 90 percent of the insects or pathogens.[12,13] The value of D is then used as a predictor of responses beyond the data to estimate the time required for disinfection (10-3 CFU/mL) or sterilization (10-6 CFU/mL).[14] The underlying assumption when utilizing this measure is that the relationship between the log10 number of survivors and time is linear.[14] If the logarithms of the D-values obtained at various temperatures are plotted against temperature, and the best straight line is drawn through the points, the reciprocal of the slope of this line is the value of z: the number of degrees by which the temperature has to be raised or lowered to bring about 90 percent reduction or tenfold increase in D.[13] In our research, steam treatment of barrels was used as a validation method after having obtained D- and z-values for different species of spoilage yeasts. The validation method was achieved in naturally contaminated barrels where we measured the temperatures reached at different times and depths of the staves. The understanding of temperature changes and times needed to achieve the sanitation as depth is increased, together with D- and z-values, will be useful to understand what parameters should be used when steam is the preferred sanitation method for wine cooperage.
Materials and Methods
Microorganisms
D./B. bruxellensis isolates (CE261, CE149), S. cerevisiae isolates (CE81, CE9, and CE78) and Z. bailii (4A1) were obtained from the Department of Food Science collection at Cornell University.
In vitro experiments
The yeasts were stored at –80 °C in 15% (w/v) glycerol, revitalized, and maintained on YPD agar (Difco™, Sparks, MD). All strains were grown until stationary phase (growth under agitation at 200 rpm, 30 °C) in YPD broth. Once the cultures reached stationary phase, the target inocula were verified via a viable count and injected in sterile glass capillary tubes; groups of five tubes were sealed with a direct flame and put into tubes with water already tempered in a water bath at the temperatures used for this study. The capillary tubes were removed at different times, put into tubes with ethanol (70%) to decontaminate the exterior surface, and then left in ice until diluted to assess the residual microbial activity. The dilutions were plated on YPD agar and incubated at 30 °C for 48 to 72 hours for Z. bailii and S. cerevisiae, and up to 3 to 4 weeks for D./B. bruxellensis.
Microbiological enumeration for thermal inactivation
Plates were enumerated for total microbial counts. The counts were averaged and expressed in logs. The log reduction was then calculated for each strain and expressed in logs. Each experiment was performed until the best linear correlation coefficients were obtained (r2 = 0.9).
Microbiological enumeration from barrels
The samples were analyzed to determine the initial and final Dekkera/Brettanomyces populations and general yeast populations, either by filtration (EZ-Fit™ manifold; EMD Millipore, Billerica, MA) using 0.22-µm disks and/or pertinent dilutions of the samples, since the microbial loads differed for each barrel. If samples required dilution, 0.1% (w/v) buffered peptone water (Hardy Diagnostics, Santa Maria, CA) was used.
For the filtration method, 0.22-µm nitrocellulose membranes (GE, Pittsburgh, PA) were used, the samples were filtered twice, and the results were averaged. The maximum volume filtered was 100 mL, and the results were calculated as CFU/100 mL and transformed into log10 values. The membranes were placed onto WLD and YPD agar using sterile forceps. WLD agar (Oxoid Ltd., Basingstoke, Hampshire, England) was used to detect D./B. bruxellensis after incubation at 30 °C for 3–4 weeks. WLD agar containing 10 mg/L cycloheximide (Sigma-Aldrich, St. Louis, MO) was used for the selection of D./B. bruxellensis (dissolved in 50% ethanol and filter-sterilized); 150 mg/L biphenyl (Acros Organics, Fair Lawn, NJ) were added to prevent the growth of mold (dissolved in ethanol and filter-sterilized); 30 mg/L chloramphenicol (MP Biomedicals LLC, Solon, OH) were added to prevent the growth of lactic acid bacteria (dissolved in 100% ethanol), and 25 mg/L kanamycin sulfate (AMRESCO, Solon, OH) were added to prevent the growth of acetic acid bacteria (dissolved in sterile distilled H2O). YPD agar was used to detect general yeast populations after incubation at 30 °C for 48 to 72 hours. YPD agar was supplemented with 150 mg/L biphenyl, 30 mg/L chloramphenicol, and 25 mg/L kanamycin sulfate for the same purposes described above.
Steam treatment of barrels
The 20 donated barrels used for this study were tested for the presence of` Dekkera/Brettanomyces as determined by the VINEO™ Brettanomytest PCR Kit (Bio-Rad Laboratories, Hercules, CA). These naturally contaminated barrels were treated with steam to reduce both D./B. bruxellensis and other general yeast populations that could be found there. The barrels were split in two groups of 10 barrels each and treated with steam for 5 and 10 minutes, respectively. Briefly, 7 L distilled water were added to the 20 barrels before the steam treatment. The barrels were rolled to enhance the contact between the water and the inner surface of the barrel and then stored, bung side up, for 24 hours and then sampled. Afterward, the steam treatment was achieved in a four-cabinet barrel washer (Tom Beard, Santa Rosa, CA) using a steam generator (ARS Enterprises, Santa Fe Springs, CA) with a pressure of 70 psi. The treatment was as follows: prerinsing for 30 seconds (cold rinsing) at a temperature of 15.5 °C; 5 or 10 minutes of steam; bunghole 5 minutes; and cold rinsing for 30 seconds at 15.5 °C. The temperature that was reached inside the staves of these barrels was monitored using four probes at two different depths (17 and 11 mm from the outside) and a data logger thermometer (Omega, Stamford, CT) that recorded the temperature at 1-second intervals, until 5 or 10 minutes were reached.
Sampling of barrels
Before and after the steam treatments, samples of water within the barrels were collected and placed in sterile bottles for microbiological enumeration. The water rather than the actual barrel surface was sampled to increase the probability of detecting contaminants. Before steam treatment, 7 L distilled water were placed in each barrel for 24 hours, and a fraction of the water was then collected in sterile containers. To collect the water, the bunghole was sprayed with 70% ethanol (before and after treatment), and the first fraction of water running out of the barrel was discarded. Then samples were taken from the middle portion of running water and placed at 4 °C until analysis was performed. The same procedure was followed to sample water within the barrel after steam treatment.
Statistical analysis
For the in vitro experiments, D-values were calculated as the negative reciprocal slope of the linear regression of survivor curves obtained by plotting logarithms of the survival counts versus time (minutes). Z-values were calculated using the negative reciprocal slope of the linear regression from the plots of the D-values versus temperatures. Only linear correlation coefficients of greater than or equal to 0.9 were used (r2 = 0.9).
For the reduction of Dekkera/Brettanomyces and general yeast populations in naturally contaminated barrels using steam, a Fisher’s exact test was performed to determine whether the two study groups (5 or 10 minutes) differed in the proportions of presence or absence of microorganisms. Statistical analyses were conducted using SigmaPlot 12.0 (Systat Software Inc., San Jose, CA).
Results and Discussion
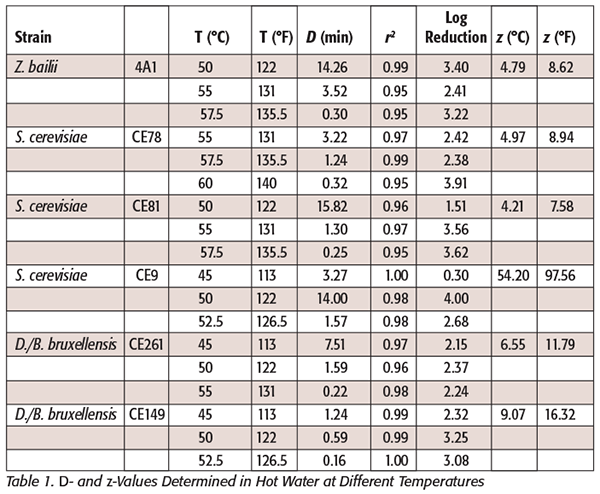 The vegetative cells of yeasts possess low heat resistance. The medium or food in which the vegetative cells are heated has a marked effect on their resistance. For instance, sugars provide protection, as do sodium chloride and citric acid.[15] Three different genera of common wine spoilage yeasts were used for this study, where thermal inactivation was achieved in hot-water baths at different temperatures. The log reduction for the yeasts studied was not increased with increasing temperature as expected in all the cases (Table 1). D-values appeared to be unaffected in some cases due to the natural variation of the response variable. A line of best fit was selected as the simplified, but nonetheless statistically correct, method of representing the data for each yeast studied.[16] Although a log-linear relationship has been applied to thermal susceptibility determinations, the straight-line relationship in such cases is not always obtained. Clumping of cells, changes in resistance to heat during treatment, or inactivation of a number of essential loci all may cause deviations from expected responses.[17] Two strains of D./B. bruxellensis were challenged at different inactivation temperatures. The highest temperature that permitted the survival of these yeasts was 55 °C (131 °F) for CE261 (Figure 1) and 52.5 °C (126.5 °F) for CE149 (Figure 2, Table 1). The lowest temperature that was capable of inactivating both yeast strains was 45 °C (113 °F). With regards to the S. cerevisiae strains used in this study, the highest temperature that permitted survival was 60 °C (140 °F) by strain CE78 (Figure 3). The lowest temperature resulting in a reduction was 45 °C (113 °F) for strain CE9 (Figure 4, Table 1). For strain CE81 (Figure 5), the highest temperature permitting survival was 57.5 °C (135.5 °F). Finally, for Z. bailii (4A1) (Figure 6), the highest survival temperature was 57.5 °C (135.5 °F) and the lowest was 50 °C (122 °F) (Table 1).
The vegetative cells of yeasts possess low heat resistance. The medium or food in which the vegetative cells are heated has a marked effect on their resistance. For instance, sugars provide protection, as do sodium chloride and citric acid.[15] Three different genera of common wine spoilage yeasts were used for this study, where thermal inactivation was achieved in hot-water baths at different temperatures. The log reduction for the yeasts studied was not increased with increasing temperature as expected in all the cases (Table 1). D-values appeared to be unaffected in some cases due to the natural variation of the response variable. A line of best fit was selected as the simplified, but nonetheless statistically correct, method of representing the data for each yeast studied.[16] Although a log-linear relationship has been applied to thermal susceptibility determinations, the straight-line relationship in such cases is not always obtained. Clumping of cells, changes in resistance to heat during treatment, or inactivation of a number of essential loci all may cause deviations from expected responses.[17] Two strains of D./B. bruxellensis were challenged at different inactivation temperatures. The highest temperature that permitted the survival of these yeasts was 55 °C (131 °F) for CE261 (Figure 1) and 52.5 °C (126.5 °F) for CE149 (Figure 2, Table 1). The lowest temperature that was capable of inactivating both yeast strains was 45 °C (113 °F). With regards to the S. cerevisiae strains used in this study, the highest temperature that permitted survival was 60 °C (140 °F) by strain CE78 (Figure 3). The lowest temperature resulting in a reduction was 45 °C (113 °F) for strain CE9 (Figure 4, Table 1). For strain CE81 (Figure 5), the highest temperature permitting survival was 57.5 °C (135.5 °F). Finally, for Z. bailii (4A1) (Figure 6), the highest survival temperature was 57.5 °C (135.5 °F) and the lowest was 50 °C (122 °F) (Table 1).


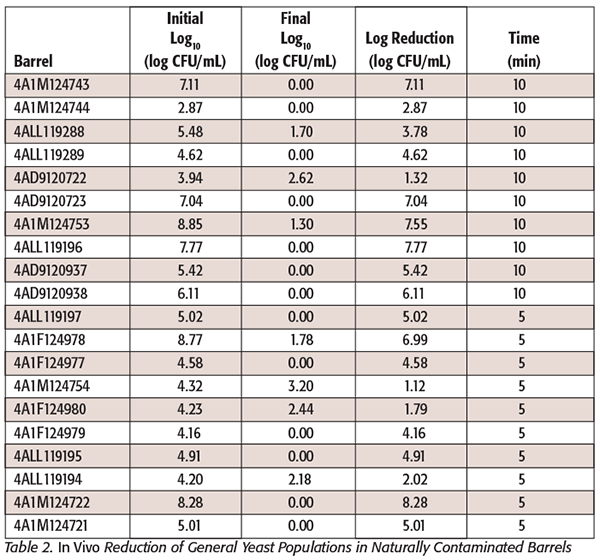
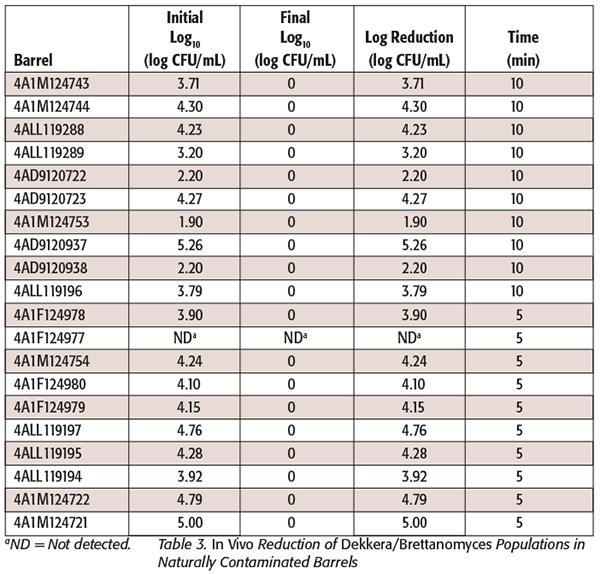
The lowest and highest temperatures obtained from the in vitro experiments that permitted survival of the strains were useful to validate the steam sanitation protocols that we achieved in wine cooperage, where steam was applied for 5 and 10 minutes to two groups of 10 barrels each, and where the presence of Dekkera/Brettanomyces and general yeast populations was analyzed. The statistical analysis showed that for general yeast populations, the 5- and 10-minute treatments were not significantly different from what is expected from random occurrence (P = 1.000); this means that the proportions of presence or absence of microorganisms are the same in 5- or 10-minute treatment. Indeed, the majority of the barrels in both treatment times had total log reduction after treatment; a few barrels had reminiscent populations of general yeast populations (Table 2). For Dekkera/Brettanomyces populations, none of the posttreatment samples were positive, so no statistical analysis was run, since both 5- and 10-minute treatments had no detectable levels of microorganisms (Table 3). In other words, there is not sufficient variability in the response to conduct statistical analysis. Since after steam treatment there was no presence of colonies on the WLD media, no further PCR analysis was performed. We found that the use of 0.22-µm filtration, combined with selective media and a long incubation time, aided detection of Brettanomyces (unpublished data). According to Starbard,[18] the recommended pore size to remove Brettanomyces is 1 µm. In our study, we incubated for 3–4 weeks to confirm or eliminate its presence. Brettanomyces strains exposed to stresses such as sulfur dioxide or nutrient starvation often do not grow for 3 days or more. Small colonies may eventually appear after 1 week (unpublished data). The relatively long incubation period used in our study was used to accommodate the time required to detect trace levels of microbial contaminants after stress treatments that could result in delayed growth. The log reduction for Dekkera/Brettanomyces and general yeast populations was substantial in both treatment times (5 and 10 minutes). This could be due to several reasons, such as the highly variable initial yeast population in those barrels and the previous sanitation practices to which those barrels were exposed. Barrels used in our study came from various wineries without uniform use, cleaning, or sanitation.
The internal temperature inside the staves of these barrels was monitored by inserting temperature probes at two depths (17 and 11 mm from the outside; the equivalent 8 and 14 mm from the inside) and a USB data logger thermometer recorded the temperature at 1-second intervals, until 5 or 10 minutes were reached. Once the steam treatment was completed, a bung was immediately inserted to generate a vacuum, which assisted in additional extraction of debris from the interior of the barrel. The analysis of the internal temperature at different depths in the barrel staves revealed that the steam application is nonuniform, and the temperature profiles showed that probes located deeper within the staves reached much lower temperatures than that reported from the point most distal to the steam source, which should be 82 °C.[19,20] In fact, much lower temperatures than the one previously mentioned were registered when less time (5-minute treatment) and deeper depths (14 mm) were used. Moreover, even when 10-minute treatment was used, none of the temperatures were more than 57.5 °C (135.5 °F). However, in vitro thermal inactivation studies showed that the highest temperature that permitted the survival of D./B. bruxellensis strains, for instance, was 55 °C (131 °F) with a corresponding D-value of 0.22 minutes and a z-value of 6.55 °C (11.79 °F) (Table 1). Our results showed that 10 minutes of steam treatment were more consistent in reaching lethal temperatures in the interior of the staves than 5 minutes of steam treatment. In fact, the highest temperature reached at 8-mm stave depth after 5 minutes of steam treatment was 47.4 °C (117.32 °F); however, using a 14-mm depth and 5 minutes of steam treatment resulted in a maximum temperature of 42.4 °C (108.32 °F). Conversely, using a 10-minute steam treatment and a depth of 8 mm, the highest temperature consistently reached was 57.5 °C (135.5 °F). Using 10 minutes and 14-mm depths, 42.5 °C (108.5 °F) was the highest temperature reached. These findings suggest that a minimum of 10-minute steam treatment is necessary to reach temperatures capable of killing harbored wine spoilage microorganisms at a depth of 8 mm. If these data were extrapolated to those from in vitro studies, the highest temperatures that permitted the survival of the genera studied ranged between 55 °C (131 °F) and 60 °C (140 °F) (Table 1).
 Consequently, if we consistently steam-treat for 10 minutes or more, it is possible to consistently reach a temperature of 57.5 °C (135.5 °F), and that is a sufficient temperature to kill wine spoilage microorganisms at a depth of 8 mm, where D./B. bruxellensis can harbor.[21] This finding is supported by the use of the D- and z-values obtained from the slope equations obtained with the in vitro experiments that in turn will help us predict information that might not be plotted in the scatter plot. Of course, deeper depths such as the one studied in this project (14 mm) also should be taken into account, since we do not know if microorganisms could reach that depth (the wine penetration is only 8 mm).[21] Since other sources of carbon, such as cellobiose, are produced during the toasting of barrels, they could serve as nutrient sources for D./B. bruxellensis and survive in the wood regardless of the level of wine penetration.[22] It is important to recognize that D./B. bruxellensis can utilize cellobiose as a carbon source for growth, as well as residual nutrients in dry, fermented wines.
Consequently, if we consistently steam-treat for 10 minutes or more, it is possible to consistently reach a temperature of 57.5 °C (135.5 °F), and that is a sufficient temperature to kill wine spoilage microorganisms at a depth of 8 mm, where D./B. bruxellensis can harbor.[21] This finding is supported by the use of the D- and z-values obtained from the slope equations obtained with the in vitro experiments that in turn will help us predict information that might not be plotted in the scatter plot. Of course, deeper depths such as the one studied in this project (14 mm) also should be taken into account, since we do not know if microorganisms could reach that depth (the wine penetration is only 8 mm).[21] Since other sources of carbon, such as cellobiose, are produced during the toasting of barrels, they could serve as nutrient sources for D./B. bruxellensis and survive in the wood regardless of the level of wine penetration.[22] It is important to recognize that D./B. bruxellensis can utilize cellobiose as a carbon source for growth, as well as residual nutrients in dry, fermented wines.
Moreover, the understanding of these findings should also be referred to a more theoretical aspect, since the thermal conductivity of wood is affected by a number of basic factors that include wood density, moisture content, extractive content, grain direction, structural irregularities (checks and knots), fibril angle, and temperature. Wood conductivity increases as density, moisture content, temperature, or extractive content of the wood increases. In fact, data suggest that conductivity along the grain has been reported as 1.5 to 2.8 times greater than conductivity across the grain.[23] Therefore, when we interpret the data found with these experiments, we must understand that many different factors may affect thermal conductivity in wood; however, as we observed, we can consistently reach temperatures that effectively kill harbored wine spoilage microorganisms if steam treatments of more than 10 minutes are applied. The concept of thermal conductivity should be understood and taken into account when steam treatment is used in woody surfaces such as wine cooperage, and it should be understood as a measure of the rate of heat flow through a one-unit thickness of a material subjected to a temperature gradient.[23]
Conclusions
Steam sanitation should ensure that sufficient temperatures are reached at the deeper depths of the staves to kill any wine spoilage microorganisms that may be harbored in the pores of wood. To our knowledge, wineries in the U.S. and other parts of the world have evaluated various sanitation practices, but there was a lack of a scientific study that could validate the use of steam to sanitize wine cooperage. In this study, we employed steam since it can reach lethal temperatures, and the in vitro thermal inactivation studies showed that temperatures between 45 °C (113 °F) and 60 °C (140 °F) were sufficient to kill common spoilage yeasts found in wine environments. However, several factors must be taken into consideration to ensure that steam treatments of barrels are effective; they include treatment times, treatment temperatures, type of microorganisms we are dealing with, and penetration depth. This experiment was designed taking into account several factors to be controlled; one was penetration depth, since we know that 8 mm has been found to be the depth of wine penetration and thus where D./B. bruxellensis and other microorganisms can be found. Based on these results, steam treatment for 10 minutes should be an effective decontamination method to ensure that at a depth of 8 mm, and perhaps even deeper, the temperature inside the staves is sufficiently uniform and high enough to kill the wine spoilage microorganisms found there. Consequently, depth, time, type of microorganisms, and temperature should be the factors considered when using steam as a sanitation method for wine cooperage to consistently reach temperatures sufficient to kill target spoilage microorganisms. The findings in this article indicate that the correct use of steam as a sanitation method in wineries can control spoilage yeast including Dekkera/Brettanomyces spp. in cooperage and therefore should be reassessed as one of the preferred sanitation methods.
This work was supported by CONACYT-Mexico, federal formula multistate project under grant [NC-1023] and Cornell University Research Travel Awards. The authors thank the donors at Constellation Brands, CA, and Francis Ford Coppola Winery (Geyserville, CA).
Aguilar Solis Maria de Lourdes Alejandra, Ph.D., is a regulatory research associate in the UK.
David M. Gadoury, Ph.D., is a senior research associate in the Department of Plant Pathology and Plant-Microbe Biology, Cornell University, New York State Agricultural Experiment Station.
Randy W. Worobo, Ph.D., is an associate professor of food science and microbiologist in the Department of Food Science, Cornell University, New York State Agricultural Experiment Station.
References
1. Woolfit, M et al. 2007. “Genome Survey Sequencing of the Wine Spoilage Yeast Dekkera (Brettanomyces) bruxellensis.” Eukaryot Cell 6:721–733.
2. Fleet, GH. 2003. “Yeast Interactions and Wine Flavor.” Int J Food Microbiol 86:11–22.
3. Loureiro, V and M Malfeito Ferreira. 2003. “Spoilage Yeasts in the Wine Industry.” Int J Food Microbiol 86:23–50.
4. Fernandes, AR et al. 2005. “Saccharomyces cerevisiae Adaptation to Weak Acids Involves the Transcription Factor Haa1p and Haa1p-Regulated Genes.” Biochem Biophys Res Commun 337:95–103.
5. González Arenzana, L et al. 2013. “Microwave Technology as a New Tool to Improve Microbiological Control of Oak Barrels: A Preliminary Study.” Food Cont 30:536–539.
6. grapesandwine.cals.cornell.edu/sites/grapesandwine.cals.cornell.edu/files/shared/documents/Research-Focus-Sanitation-of-wine.pdf.
7. Bisson, L. “What is ‘Brett’?” in Winemaking Problems Solved, ed. C Butzke (Boca Raton, FL: Woodhead Publishing, 2010), 290–291.
8. Van de Water, L. “How Can I Manage Brettanomyces in the Cellar?” in Winemaking Problems Solved, ed. C Butzke (Boca Raton, FL: Woodhead Publishing, 2010), 329.
9. Younsi, R et al. 2006. “Three-Dimensional Simulation of Heat and Moisture Transfer in Wood.” Appl Therm Eng 26:1274–1285.
10. Qing-Xian, Y. 2001. “Theoretical Expressions of Thermal Conductivity of Wood.” J Forestry Res 12:43–46.
11. Khattabi, A and P Steinhagen. 1993. “Analysis of Transient Nonlinear Heat Conduction in Wood Using Finite-Difference Solutions.” Holz Roh Werkst 51(272):278.
12. Simpson, WT. “Heating Times for Round and Rectangular Cross-Sections of Wood in Steam.” Gen Tech Rep FPL-GTR-130 (Madison, WI: USDA, Forest Service, Forest Products Laboratory, 2001).
13. Narang, SP. Food Microbiology (New Delhi, India: A.P.H. Publishing Corporation, 2004).
14. Sutton, SVW et al. 1991. “D-Value Determinations Are an Inappropriate Measure of Disinfecting Activity of Common Contact Lens Disinfecting Solutions.” Appl Environ Microb 57:2021–2026.
15. Splittstoesser, DF et al. 1986. “Effect of Food Composition on the Heat Resistance of Yeast Ascospores.” J Food Sci 51:1265–1267.
16. biologicalindicators.mesalabs.com/wp-content/uploads/sites/31/2014/07/Spore-News-Vol-3-No-2.pdf.
17. Forsythe, SJ. The Microbiology of Safe Food (Iowa: Blackwell Publishing, 2010).
18. Starbard, N. Beverage Industry Microfiltration (Iowa: Wiley-Blackwell, 2009).
19. www.practicalwinery.com/janfeb09/micro1.htm.
20. Fugelsang, KC. “Winery Microbiology and Sanitation,” in Winemaking Problems Solved, ed. C Butzke (Boca Raton, FL: Woodhead Publishing, 2010), 260.
21. Malfeito Ferreira, M et al. 2004. “Effect of Different Barrique Sanitation Procedures on Yeasts Isolated from the Inner Layers of Wood.” Am J Enol Viticult 55:304A.
22. Blomqvist, J et al. 2010. “Fermentation Characteristics of Dekkera bruxellensis Strains.” Appl Microbiol Biotechnol 87:1487–1497.
23. Simpson, W and A TenWolde. 2007. “Physical Properties and Moisture Relations of Wood,” in The Encyclopedia of Wood, ed. A Walker (New York: Skyhorse Publishing Inc., 2007), 15–17.
Thermal Inactivation of Wine Spoilage Yeasts to Validate Steam Sanitation Protocols in Wineries




