In the April-May 2011 issue of Food Safety Magazine, an article entitled “How Should Packaging Be Addressed in Your Food Safety Program” examined the role of packaging and potential safety issues.[1] The food package, whether it is the primary or secondary package, serves a number of roles: It protects the food, provides the consumer with information, helps market the food, warns customers of potential dangers (such as from allergens), provides a container for transport, allows the product to be tracked and traced, and can even provide evidence as to how a product might have become contaminated or spoiled. Packaging was described as being able to inform a consumer or user about how fresh a product might be. The conclusion was that food packaging was generally safe, primarily because all materials used in packaging are subject to approval by regulatory agencies, such as the U.S. Food and Drug Administration (FDA) or the European Food Safety Authority.
So, the question is, has anything changed in the past 5 years? Does packaging pose a risk, and how should it be addressed in your food safety management system (FSMS)?
Hot Issues in Packaging
Consumers have developed a fear of chemicals, which is not scientifically justified. The attitude of many is “If I can’t pronounce it, I don’t want it in my food.” This has led to the clean-label movement, in which food processors are scrambling to remove the unpronounceable ingredients from their labels, whether they are harmful or not.
Five years ago, one of the big consumer concerns was a chemical called bisphenol A (BPA). BPA is an industrial chemical used to make a hard, clear plastic known as polycarbonate, which has been used in many consumer products, including reusable water bottles and baby bottles. BPA is also found in epoxy resins, which act as a protective lining inside metal-based food and beverage cans. In January 2010, FDA stated that while tests supported the safety of low-level exposure to BPA, more research was being done on its potential effects on infants and children.
By 2011, FDA scientists had determined that infant exposure to BPA through foods was lower than previously believed, but the agency still supported industry efforts to find an alternative to BPA.[2]
So, FDA has not banned the substance but understands that there are consumer concerns. Because of these concerns, packaging suppliers have been utilizing other materials in packaging.
Foreign Material Concerns
Do packaging materials pose a food safety risk for foreign materials? Packages are manufactured from glass, various metals like steel, aluminum, and tin, and a wide range of plastics and polymers. All of these materials would pose a risk if they ended up in the product, but what are the chances that they actually would do so? Quite low, but there are issues that should be addressed in production facilities.
The Institute of Packaging Professionals (IOPP) has developed a document entitled Potential Food Safety Risks and Possible Controls for Food Packaging Materials.[3] This document looks at all kinds of packaging materials, including fiber and labels. For each type of packaging, the document includes two columns: Potential Issues (Food Safety Implications) and Possible Controls (this list is not all-inclusive; alternative controls are possible). The introduction to the embedded table includes the following statement:
“Many of these hazards may be controlled by strong prerequisite programs but some may require being considered Critical Control Points (CCPs) in a HACCP plan or equivalent food safety focused control plan.”
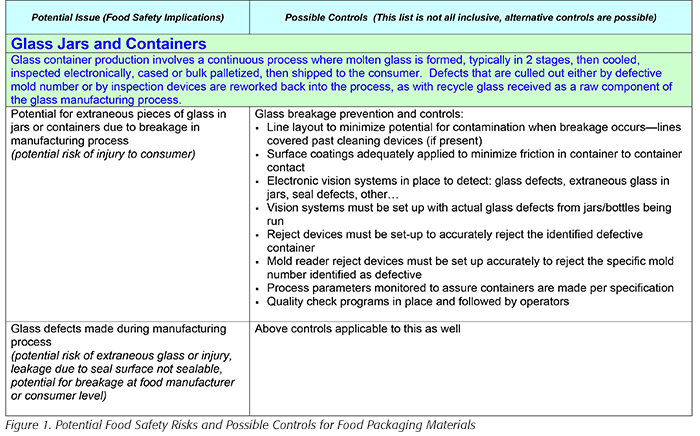 Figure 1 shows a partial screen shot of this table, which can be found on the IOPP website.[3]
Figure 1 shows a partial screen shot of this table, which can be found on the IOPP website.[3]
This part of the table was selected intentionally. Glass and glass contamination is probably the most significant potential physical hazard. This is why all processors must develop, document, implement, and maintain a glass and brittle-plastic program. For companies packaging in glass, this program is especially important. Glass packaging operations must have especially rigid programs to address breakage. Among the elements that their program must include is what to do when there is on-line breakage. Many companies will remove all product or containers within 6 to 10 feet of where the breakage occurred. Companies packing baby food in glass may have even more rigid programs. The concern is that glass shards may end up trapped under the lid of a perfectly good container.
Foreign Material Controls
Food processors utilize a number of different unit operations to minimize the potential for foreign material contamination. How these are managed and utilized can protect finished products from potential contaminants that, depending upon the contaminant, could pose a health risk. These unit operations include air washers, water washers, metal detectors, X-ray machines, magnets, cameras, scanners, lighting systems, and visual inspection. And of course, processors need to not only develop, document, and implement procedures to control potential contamination with foreign materials but also to make sure that these procedures are properly maintained. Maintenance requires management commitment to ensure that workers are properly trained, that they understand that their job is important and that records are not only maintained but also properly reviewed.
Air Washers — Air washers are used in many types of processes and with different packages. They are used with glass, cans, and plastics or pouches. The air washer performs two functions with pouches. It cleans the pouch and can ensure that the pouch is open prior to filling. Air washers must utilize filtered air to ensure that the air itself does not potentially contaminate the package. The filter should be designed to remove moisture and oil. As part of the maintenance program, filters need to be monitored regularly. In addition, a program for routinely changing filters must be established. Pressure gauges on the filter system also allow the processor to monitor filter performance. Processors should also conduct studies to validate that the air filter effectively cleans the package in question. As an example, some lines are designed to air-wash bottles of different sizes. Processors need to validate the settings for each bottle size. If the same setting is used for all sizes, make sure it is effective.
Water Washers — Cans and bottles (plastic and glass) are often washed. Wash water may be cold, hot, or contain a sanitizer such as ozone. One manufacturer of baby food with whom I worked established the glass-washing step as a Critical Control Point (CCP) in their process. Their washing step was a two-stage wash: a warm-water rinse to temper the glass and a second-stage wash done at 180 °F or above. The washing step also included water pressure as part of the CCP. The combination of water temperature and pressure ensured adequate washing of the jars.
As noted above, water washing should be validated. To validate a washing step, bottles can be seeded with glass chips or other contaminants. The bottles are then recovered after the washer, filled with distilled water and shaken vigorously. The water is then poured through a filter apparatus and the filter examined for the seeded glass.
Metal Detectors — Metal detectors are designed to detect all metal in food products above a certain size. The size of metal that is detectable depends upon the product and its package. Different types of metal detectors are available to the food industry. These include systems that pass the products through on a conveyor, in-line systems for liquids, and vertical inspection systems. Metal detectors are almost always designed to reject product found to contain metal, although there are occasional units in which the conveyor simply stops. Ideally, the best location for a metal detector is after packaging, which is why conveyor-type systems are so popular. If metal is detected, the package will be rejected. Of course, if your package contains metal, processors would have to adopt another type of metal detection system or utilize X-ray detection. If the metal was originally from the packaging and it is large enough, the unit should find the metal. However, the chances of metal coming from packaging being a hazard are probably quite low.
X-Ray Machines — X-ray technology has expanded rapidly over the last few years. It remains expensive, but costs are coming down. Most people associate X-ray technology with foreign material detection, but X-ray machines have other abilities. These include mass analysis, confirming fill levels, evaluating seal integrity, recognizing missing product and data logging, since not only is each container X-rayed, but also the photograph is maintained. X-rays machines can detect metal and hard plastics, both of which are used in packaging. One must remember that X-ray machines and metal detectors do not detect and remove all metals or other foreign materials. They are effective down to a certain size.
Magnets — Magnets have many applications in food plants. They will remove ferrous and nonferrous metals, but unlike metal detectors or X-ray machines, they can remove very small metal fragments including rust. Grate-type magnets and bar magnets are excellent for removing contaminants such as staples or clips that may be used on bins or totes. There is a reason why food safety professionals discourage people from using staples on bins, totes, or anywhere in the plant. A few inevitably end up in the product.
Cameras — Many processors employ cameras to scan containers prior to use in processing. They are often employed on can lines adjacent to the depalletizer. As the containers leave the depalletizer, they pass the camera, which is linked to a computer. The picture is compared with the standard. If the photograph deviates from the standard, the can will be removed from the line. These systems can detect can defects that might affect sealing, grease spots on a container or other defects.
Scanners — Electronic sorting or scanning systems are finding more and more acceptance in the industry. This is due in part to advances in sorting technology. These systems, whether using lasers, vision systems, or other technologies, will remove foreign materials but are really utilized more for quality purposes. Laser-sorting systems can also be used for cleaning up bulk grains, seeds, and nuts. This is especially important since bulk containers are often used for moving different products. I once watched a processor pass a load of red lentils through a laser sorter. Two passes through the system removed a significant amount of foreign material including corn, soy, and wheat. This technology could be expanded to detect packaging defects (see cameras, above). How effective such technology would be at finding small pieces of packaging materials would depend on the packaging material and the product matrix.
Lighting Systems — Many processors using glass or plastic containers pass the packaging by a light box. Backlighting the bottles illuminates defects, making them easier to see. This is especially true with glass bottles or jars. Defects such as bird’s feathers, inclusions, or bubbles may be seen. I have never seen a backlit glass inspection system that included an automatic rejection system. Processors using these systems must rely on a worker to do the actual inspection and removal.
Visual Inspection — Visual inspection may be used to detect defects as noted above. There are inherent problems with visual inspection. It must be done by people, who get fatigued. Talk with any food processor that utilizes sorting or visual inspection and ask them how often they rotate people on and off the inspection lines. Some rotate at intervals as short as every 15–30 minutes. They are aware that visual inspection and/or sorting is tiring and efficiencies drop sharply as people become tired. Visual inspection as a means for detecting container defects or contamination is not the best option. Perhaps the best kind of visual inspection is when products or packages are physically handled by a worker when loading a retort basket or tray. They can be asked to examine each package for delamination, damage, breakage, or other defects that might pose a health or safety risk.
Allergen Management
The Preventive Controls regulation that was finalized at the end of 2015[4] defines allergen management as one of the required preventive controls. An allergen management program is a complex, multidisciplinary program of which labeling is only a part, albeit an extremely important part. The allergen labeling regulations clearly state that all allergens must be clearly identified on the package. Many processors use a statement like the following after the ingredients statement to ensure that allergens in the product are clearly defined: “This Product Contains Milk and Soy.”
Many other statements are used, but the bottom line is to inform the consumer of what is or may be in the product.
One of the best tools to ensure that the right labels are used on products is a label management program. The food package and its label are, therefore, a first line of defense and an essential element for ensuring consumer safety. Label management consists of several elements, including verifying that the proper labels are ordered and delivered, ensuring that the correct labels are used in processing, which includes removing and destroying old and outdated labels, and verifying that these protocols are being followed.
A label management program might include the following elements:
• All current labels used in production will be printed and placed in a master label file. The current labels will be dated when placed in the master file.
• When a label is updated or removed from use, that label will be removed from the master file.
• The master file will include a change section. Whenever a change in label is made, that change will be noted in the change section of the master file.
• At the start of production, the label or labels to be used that day will be compared with the label in the master file to ensure it is accurate. This check will be entered into the production records.
• During production, one label from the beginning, middle, and end of production will be attached to the production records to verify that the proper label was used. The date and time that the label was removed from production and placed in the records will be noted on the label itself. The person doing this will also sign or initial the label.
Processors must also establish programs to monitor label quality from receipt through use and/or destruction. Such a program might include the following:
• When labels are received, quality assurance shall be responsible for ensuring that the labels received are properly printed and accurate. This shall be accomplished by comparing a random sampling of labels with the master maintained in the main office.
• If the new labels match the master, they will be approved.
• If the new labels do not match, the lot will be rejected and returned to the vendor at their cost or destroyed on-site.
• A record that the lot in question was destroyed shall be maintained.
• Once received labels have been approved, they shall be securely stored adjacent to the production area. The storage area shall be clearly marked as accessible to AUTHORIZED PERSONNEL ONLY. Labels shall be marked with the date received. Labels used for products containing allergens shall be clearly marked to ensure that they are not commingled with other labels.
• Labels shall be removed from the storage area as required by production. A log shall be maintained of how many labels were utilized and whether any labels were returned to storage. In addition, production shall attach a label to the production records to clearly document that the lot being packaged used the proper label.
• If a label becomes outdated or there is a change in formula requiring the development of a new label, all old or outdated labels shall be destroyed. A record that the lot in question was destroyed shall be maintained.
• Quality assurance shall conduct quarterly inspections of the label storage area. Any deficiencies in storage, usage, or recordkeeping shall be noted and subject to corrective actions.
The Global Food Safety Initiative and ISO 22000
In today’s food processing industry, third-party audits are a fact of life. Buyers expect that their suppliers will have been certified under one of the four Global Food Safety Initiative (GFSI)-benchmarked schemes, have achieved ISO 22000 certification (Food Safety Management Systems – Requirements for Any Organization in the Food Chain[5]) or been inspected using a private standard developed by a firm or laboratory. Processors should ask their customers what program they would like to see. If a company demands that a supplier adopt a GFSI scheme, the operation should be allowed to select the scheme that best suits them: BRC (British Retail Consortium), IFS (International Featured Standard), SQF (Safe Quality Foods), or FSSC 22000 (Food Safety Systems Certification). These schemes mandate that food processors employ not just a Hazard Analysis and Critical Control Points plan but also an FSMS. One element of such a program is that processors approve all suppliers, including packaging suppliers, and conduct a risk assessment on all purchased materials. This means ingredients, raw materials, and packaging materials. The expectation is that this risk assessment will include two elements: an evaluation of the severity of occurrence for all potential hazards and the likelihood that they would occur. The assessment for each type of packaging material should be recorded and incorporated into the FSMS. The results of these assessments would indicate that packaging materials are low-risk items. If the assessment said that a material was a high-risk item, the company should take steps immediately to find a low-risk alternative. The goal is safety across the board.
Summary
So, is packaging a significant risk in the overall food safety management system? The answer is no. The overall risk is probably even lower today with the emphasis on adoption of GFSI audit schemes or ISO 22000. The GFSI schemes and the ISO standard mandate that processors conduct a risk assessment on all raw materials, ingredients and packaging materials. And guess what? The U.S. Food Safety Modernization Act has the same mandates, and most food processors the world over evaluate risk on their packaging. In fact, the final paragraph from 2011 still holds true today:
“Packaging is and will remain an evolutionary science and technology. It is a discipline that constantly learns from its mistakes and has evolved rapidly over the past twenty years. Take a walk through your local market and you will see packages on the shelves that weren’t there a few years back. One word of advice to the food processor, however; when looking to change packages or materials, look to a packaging professional for help. Go to your suppliers, a consultant or someone in academia who understands the science and technology of packaging. They can help you not only find the best materials, but can help troubleshoot problems and reduce costs. Seek to do it right the first time around.”
Richard F. Stier is a consulting food scientist.
References
1. Stier, RF. 2011. “How Should Packaging Be Addressed in Your Food Safety Program?” Food Safety Magazine 17(2):30–33, 67–68.
2. www.fda.gov/forconsumers/consumerupdates/ucm297954.htm.
3. www.iopp.org/files/public/FSAP/FSAP%20Risks%20-%20Controls%20Pkg%20Materials%20May%202009.pdf.
4. www.fda.gov/Food/GuidanceRegulation/FSMA/ucm334115.htm.
5. www.iso.org/iso/catalogue_detail?csnumber=35466.




