Outsourcing your food safety laboratory testing is a sound business strategy to improve quality, increase cost savings, and boost profit margins. Outsourcing frees companies to focus on core competencies and quality managers to focus on quality products and customer service rather than managing a complicated analytical laboratory. In-house laboratories also are often plagued by substantial operating costs such as:
• Specialized, trained technical labor
• Expensive analytical and support equipment
• Maintenance contracts
• Calibration services
• Utilities
• Floor space inventory management
• Reagent and supply inventory management
• Waste management associated with disposal of biohazardous materials
Eurofins’ laboratory resource utilization rate enables us to maximize testing efficiency, and, with multiple customers, fixed capital costs are distributed across business units. Our high-volume laboratory teams include purchasing analysts who have significant leverage with vendors of equipment and consumables. Our sophisticated laboratory information management system eliminates reagent and consumables waste due to expiration.
All of these benefits mean substantially reduced testing costs for our food safety partners.
Human Resource Solutions
Hiring an unskilled analyst who delivers faulty test protocols, sloppy bench skills, and questionable data generation can severely jeopardize your brand. Yet acquiring and retaining technical talent is a growing challenge and continual cash drain in:
• Recruitment
• Headhunter fees
• Employee training
• Salaries and benefits
• Absenteeism
• Turnover
In addition, many in-house laboratories operate a single-shift, 5-day schedule, which may not be linked to production schedules. Our teams of motivated, talented technical personnel provide extensive industry and regulatory knowledge, which can accelerate new product development and reduce regulatory compliance headaches. Our food safety partners have access to expanded technical expertise and 7 days per week in multi-shift operations.
With a keen focus on analytical methods, our experts work side by side with your teams to develop client-specific methods. We are quick to adopt new technologies and pass associated cost and turnaround time savings on to our customers.
Our staff scientists are active members and leaders of professional organizations, and, as a leading global independent laboratory group, we have earned an unrivaled reputation for unbiased analysis.
Quality Solutions
In-house laboratories’ quality deficiencies are usually quickly discovered during outside regulatory or client visits; these include:
• Outdated or poorly kept equipment
• Equipment that is not calibrated or routinely monitored for performance accuracy
• Lack of quality systems
• Lack of traceable data management systems
• Lack of personnel backup
Every Eurofins laboratory maintains accreditations, registrations, and certifications that meet each unique set of industry standards, and all of our laboratories are accredited to the ISO 17025 standard. We invest heavily in our quality systems, with each laboratory staffed with full-time quality assurance personnel. And as a large-volume provider, our laboratories offer equipment and personnel redundancies that can quickly manage emergencies.
Eurofins On-Line provides our food safety partners with a secure, innovative, individualized software platform with 24/7 access to Eurofins testing laboratories and a comprehensive reporting tool for your real-time data and order management needs.
Service Solutions
As a global laboratory group with many centers of excellence, Eurofins offers over 100,000 analytical methods, and we are often first to market when a new food quality or safety threat emerges (GMOs, melamine, dioxins, etc.). We offer chemical, nutritional, residue, authenticity, and microbiological testing as well as a suite of auditing, consulting, and training services.
And with a single point of contact, you can significantly reduce administrative overhead associated with purchase orders and invoices.
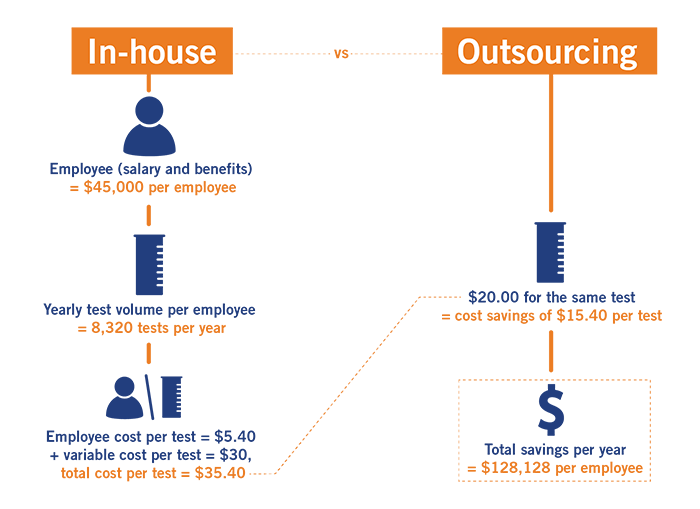 Consider this common in-house microbiology testing cost scenario (see graphic at right):
Consider this common in-house microbiology testing cost scenario (see graphic at right):
Note: We are happy to provide cost calculation comparisons using your financials!
In-House Test Kits
Often, food producers, processors, retailers, and foodservice operators use do-it-yourself test kits or instruments to assess authenticity, quality, or safety of incoming raw materials and finished products. Two primary benefits of such devices include ease of use and rapid results, which enable users to make quick decisions on product viability.
However, there’s much to consider when you take this approach.
First, your teams can only be as effective as their tools allow. Many companies tout simplistic versions of easy-to-use devices for food analysis, but there are many caveats to their effectiveness (and as your food safety partner, Eurofins can be helpful when trying to differentiate a misleading claim from true quality).
Our recommendations for buyers of do-it-yourself test kits/instruments for food analytes like toxins, heavy metals, sanitation, etc. is to ensure they’re appropriately validated for your intended use. Some commercial kits/instruments may not have the sensitivity and/or accuracy required to detect and quantify low levels of potentially harmful substances. Also, kits/instruments may not have been validated for the products of interest. For example, a test for contaminants in water may not be a suitable test with a complex matrix like fish.
If you’re using kits/instruments to generate data for regulatory compliance, make sure the relevant regulatory authorities have approved the device for this use. Before buying, ask the kit/instrument manufacturers:
• Has the kit been validated for products and analytes of interest?
• Are you really measuring the analyte of interest or some other proxy?
• Is there instrument drift?
• What is the precision and accuracy of the kit? How were precision and accuracy established?
• Can the instrument be easily calibrated?
• How do I determine staff competency and kit applicability for the task at hand?
• Do the kits have regulatory approval?
• Do the kits have broad industry acceptance?
• What is the shelf life of the kit and required storage condition?
Once you’ve determined these factors, you should decide whether it makes sense to do analytical chemistry in a foodservice or food processing environment. Some additional considerations are:
• Do you have trained analysts who can perform the tests?
• Do you feel comfortable interpreting the results?
• Do you participate in proficiency tests to ensure you are able to achieve accurate results?
• Do you have a quality management system in place to archive data?
• Are you running positive and negative controls?
• Should you have positive controls of high-risk substances (toxins, heavy metals, pathogens) in your work environment?
• Do you have storage facilities that meet the requirements for storage of perishable kits?
• Do you have the tools and expertise needed to properly prepare a sample before analysis?
• Does speed and ease of use trump precision and accuracy?
Make sure you validate the usefulness of kits or instruments in your applications and conduct periodic checks with professional laboratories such as Eurofins. If your results aren’t sufficiently accurate, it may make sense to outsource testing to reputable laboratories. Validating your results also benefits you when auditors ask for evidence that what you are doing actually works. Eurofins Technologies provides a number of useful and validated kits for many in-house applications; we encourage you to reach out for a conversation.
At the end of the day, perhaps do-it-yourself devices can provide useful on-the-fly data; however, it is prudent to spend the additional resources required to ensure that such data are accurate, reliable, and sufficiently contribute to the minimization of risk to your brand.
Whatever you choose, and wherever you are in your testing journey, Eurofins is proud to be your local food safety partner. We know your territory, and we delight in providing fast, accurate, cost-efficient expertise to every region we serve.
Douglas Marshall is the Chief Scientific Officer of Eurofins Microbiology Laboratories, Inc., a division of the global life sciences company Eurofins Scientific.