The Food Safety Modernization Act (FSMA), signed into law on January 4, 2011, enables the U.S. Food and Drug Administration (FDA) to better protect public health by strengthening the food safety system. FDA is able to focus more on preventing food safety problems rather than relying primarily on reacting to problems after they occur.
FSMA inspections for the largest manufacturers of human food began in 2016. The largest manufacturers include those with 500 or more full-time employees and at least $1 million worth of product. Inspections for small facilities are being phased in over several years.
What to Expect during a FSMA Compliance Inspection
The important thing to note about domestic FSMA compliance inspections is that they can be conducted at any time, without any advance notice. In some cases, facilities may get a call from an inspector stating that they will be there in an hour, but inspectors don’t even need to do that.
FDA has publicly announced that they don’t have enough internal employees to conduct all the inspections that need to be done. Therefore, they are partnering with state agencies—including agricultural departments, health departments, and tribal officials for facilities on a reservation—to conduct those inspections on behalf of FDA.
An inspector conducting an inspection on behalf of FDA will write a report with their observations of noncompliance. FDA will then determine any resulting enforcement or action. This could include a 483, a warning letter, an injunction or a seizure based on the classification (official action indicated, voluntary action indicated, no action indicated, etc.).
FSMA Compliance Inspection Process
During a FSMA compliance inspection or a Good Manufacturing Practices inspection, the inspector will analyze employee use of tools, storage practices, structural integrity, electrical equipment, and more.
Also during these inspections, the inspector may change the direction of the inspection and move to swab testing to look for environmental pathogens any time they notice something amiss, whether it be in the facility or even in the paperwork. For example, this could occur because they saw ready-to-eat products stored next to raw products.
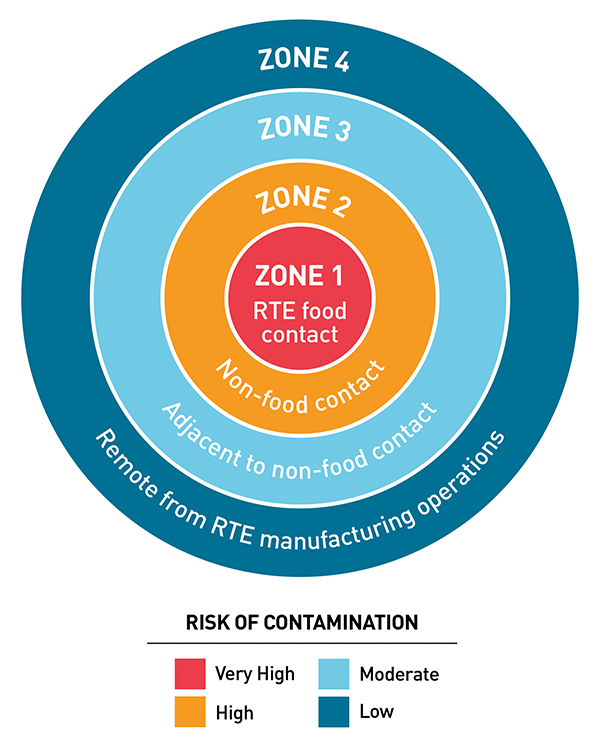 An inspector may conduct swab testing in zones 1, 2, 3, or 4. They may take a minimum of 100 samples based on the size of the facility and what zone they are in.
An inspector may conduct swab testing in zones 1, 2, 3, or 4. They may take a minimum of 100 samples based on the size of the facility and what zone they are in.
Zone 1 consists of the production/processing areas and includes the work area where employees and machinery have direct contact with food and the product is exposed until it is packaged. Zone 2 is the area immediately adjacent to production/processing and is not in direct contact with food. Zone 3 may consist of staging areas, floor drains, nearby walls and ceilings, and potentially the unpacking and quality control labs. Zone 4 typically consists of the offices, employee break room/locker areas and possibly maintenance areas, depending on their location within the facility.
An inspector will look at your sanitary design and electrical equipment to make sure it is up to code and meets FSMA requirements. If the end of a cord is frayed, duct-taped, electrical-taped, exposed, hanging over a production line, or dripping due to the buildup of condensates, the inspector may move into swab testing. There could also be instances where an electrical control panel is not sealed properly and could have pathogen growth or harborage.
In food processing facility environments, the everyday use of electrical devices causes wear and tear that can result in insulation breaks, short-circuits and exposed wires. Plant managers should incorporate the process of checking their facility for deteriorating devices into their preventive maintenance program.
Electrical Equipment for Your Food Processing Facility
In many food processing facilities, high-pressure washdowns are performed multiple times per day to ensure sanitary conditions. This rough, corrosive environment can shorten the life of electrical devices and compromise the system by creating safety issues, unreliable connections, and lost productivity.
New advances in watertight, rugged plugs and connectors and receptacles that are rated for high pressure ensure more reliable power delivery in these highly sensitive areas. Many devices use multipoint sealing systems to guard against water and contaminants, creating a safer environment for workers and food production.
An open cable management system of wire mesh trays decreases the potential for bacteria to build and spread throughout the facility, preventing product contamination and potential recalls. Antimicrobial plugs and connectors feature antimicrobial additives embedded directly into the polymer components. The additives are gradually and continuously released to the surface, inhibiting the growth of dangerous microorganisms and providing long-lasting protection against bacteria, molds, mildew, and fungi.
Common Mistakes in FSMA Inspection Preparedness
FSMA compliance inspections are still a new experience for many companies, and mistakes are bound to happen. Here are the nine most common mistakes he sees when it comes to FSMA inspection preparedness:
1. Incomplete hazard analysis. This is the crux of your safety plan. Every food processing company must have a completed hazard analysis that is robust.
2. Incomplete process for preventive control validation studies. This could include studies that are incomplete or have not been conducted, or cases where they don’t have proof of review.
3. Missing signatures on their food safety plan or on associated verification records. While seemingly a simple task, make sure your records are signed appropriately.
4. Lack of facility or supply chain preventive control program.
5. Lack of recall plan. Some food processing facilities have an incomplete plan or no plan at all. Each facility is required to
have a recall plan in place.
6. Failure to practice or enforce Good Manufacturing Practices. This can be a challenge for facilities with high employee turnover rates.
7. Failure to treat human and animal foods properly. Any human food destined for animal food needs to be properly identified, handled and stored.
8. Lack of or incomplete letter assurance. Companies that are not controlling a known hazard and are depending on an entity downstream to control the hazard need to have detailed letters of assurance between both parties.
9. Failure to address environmental pathogens. They are either not considered in the hazard analysis or their environmental monitoring program is too weak. There should be a very robust seek-and-destroy approach to environmental pathogens. If you go several years without finding an environmental pathogen, that will be an open invitation for a swab-a-thon by FDA.
Best Practices for FSMA Inspection Preparedness
To help make sure you avoid common mistakes, here are best practices for making sure your facility is FSMA compliant and that you’re prepared for an inspection, whenever it may occur.
1. First of all, don’t work off checklists. Checking off boxes on a list doesn’t always align with what is really happening on the floor and can lead to regulatory actions you’re trying to avoid. Instead, take a holistic look at your facility’s operation, including food safety culture.
2. Make sure food safety is practiced through your whole facility and that your employees take ownership of it. They have to be trained to understand not only what they have to do, but why they have to do it. Give them a reason why and they will take ownership.
3. Have representatives from multiple departments on your food safety team. They should all have input when you’re developing and carrying out the food safety plan.
4. Hire a third-party consultant to come in and take a holistic look to see if you’re meeting the intent of the law and protecting your brand. Work with them to review your facility plans. Be open to change and input, and be willing to accept constructive criticism.
5. Finally, keep an open ear and eye for any new guidance that comes from FDA. Make sure you understand it and, if you don’t, reach out and ask for help.
The author thanks Jeff Seabury, Industrial Product Manager, Legrand, for contributing to this article.
Christopher Snabes, M.Sc., is a director of food safety with The Acheson Group.




