In today’s 24-hour news cycle, each day seems to bring to light new chemical hazards present in our food—whether it’s arsenic in apple juice, melamine in dairy products or acrylamide in fried foods. In recent years, several of these scares have involved chemicals in food packaging materials (or other types of food contact materials), most infamously bisphenol A. Often, media reports on scientific studies lack context to help readers understand what the study results mean in the “real world,” or they distort the meaning of these studies. For instance, the interpretation of toxicology data and their use in human health risk assessment are often misunderstood by the media, resulting in inaccurate reports about what the toxicological data used in safety assessments really mean. In some cases, misguided public opinion has caused chemicals to be withdrawn from the market, even when scientific studies have demonstrated that these chemicals are safe and effective. To make matters worse, some withdrawn chemicals have been replaced by others of uncertain safety and effectiveness.
When evaluating materials for use in food packaging and other food contact applications (e.g., paper plates, cutting boards, food processing equipment), a number of considerations are important. Cost is often the driving factor, as is the effectiveness of the chemical in performing its function (e.g., preserving freshness, retaining moisture). However, the question of whether a food contact substance (FCS) presents a risk to the health of consumers is often considered later in the process, even though this question is critical to determining whether a material is suitable for food contact use.
The U.S. Food and Drug Administration (FDA) has the authority to approve or deny the notified use of an FCS under its Food Contact Notification (FCN) Program within the Center for Food Safety and Applied Nutrition. The process FDA uses to make this decision is based on a quantitative, human health risk assessment, which is a well-established scientific method used to assess whether the potential exposure or dose of a substance is safe.
With the increasing use of nanoscale materials in FCSs, this balance of functionality, human health risk assessment, risk management and risk perception appears to be at a tipping point. In this article, we outline FDA’s risk assessment process for evaluating the safety of an FCS; we describe some of the applications, benefits and human health risks associated with the use of nanoscale materials in FCSs; and we discuss the current regulatory structure specific for nanoscale materials in the U.S. under FDA and the U.S. Environmental Protection Agency (EPA).
Safety/Risk Assessment Process
“Toxicology” is defined as the study of the adverse effects of chemical or biological substances.[1] Paracelsus, considered to be the father of modern toxicology, is credited with forming its central tenet, paraphrased as “The dose makes the poison.” This concept indicates that all chemicals—whether water or oxygen or benzene—have the potential to be toxic. It is simply the amount or dose of a chemical (i.e., how much is absorbed by the body) that determines whether it is toxic.
Risk assessment is a key part of the process FDA uses to evaluate whether the notified use of an FCS is safe. The basic steps in a risk assessment include hazard identification, exposure assessment, toxicity assessment and risk characterization. Although regulatory agencies such as FDA and EPA may use slightly different nomenclature for the risk assessment process, each agency incorporates at least some form of this tool.
Each step in the risk assessment process is essential to informing the criteria that FDA uses to assess an FCN. Section 409(c) of the Federal Food, Drug, and Cosmetic Act (FD&CA) provides the legal requirements for food additives, which are applicable to an FCS and its constituents that migrate or may migrate onto/into food. Specifically, the FD&CA requires that an FCS is not a carcinogen, that the amount of the substance is no more than necessary to achieve its intended effect and that use of the substance is safe, taking into account the potential cumulative effect of the substance in contact with a food and similar substances in the diet.[2] In addition, the National Environmental Policy Act of 1969 requires each federal agency, including FDA, to assess the potential environmental impact of regulatory actions and requirements. All FCNs must contain either a claim of categorical exclusion under FDA’s environmental impact regulations under 21 C.F.R. 25.32 or an environmental assessment under 21 C.F.R. 25.40.[3]
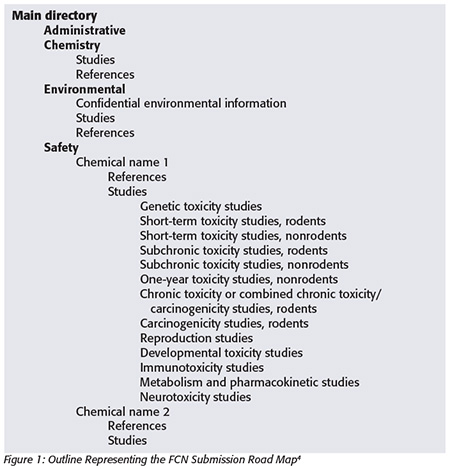 FDA guidance for industry details the type of information that may be needed for an FCN (Figure 1[4]). Depending on the nature of the FCS and its proposed use, this list may seem daunting. However, understanding the safety assessment process can help a notifier provide sufficient detail without providing extraneous information.
FDA guidance for industry details the type of information that may be needed for an FCN (Figure 1[4]). Depending on the nature of the FCS and its proposed use, this list may seem daunting. However, understanding the safety assessment process can help a notifier provide sufficient detail without providing extraneous information.
In contrast with other regulatory schemes for risk assessment, such as EPA’s approach, FDA’s scheme uses the exposure assessment [specifically, the cumulative estimated daily intake (CEDI)] to determine the toxicity data that are required. The CEDI is derived using FDA-recommended migration-testing protocols. The agency provides different migration-testing protocols depending on the type of product and end use. For instance, different protocols apply for high-temperature applications, for degradable polymers and for can coatings.
In some ways, FDA’s approach to the FCN process is quite prescribed; however, many nuances of the process can lead to objection letters or follow-up submittals. For example, shortcomings in the study data that describe the migration of the FCS, a lack of toxicity data, conflicting toxicity data or uncertainty in the cumulative exposure assessment can complicate the FCN process. Being able to communicate effectively how these potential issues affect the overall toxicity profile in the FCN is critical to a successful submission. Understanding these key issues can help a notifier avoid a rejected FCN.
In the case of an FCS with very few toxicity studies, the notifier is faced with a difficult situation. If low exposure is predicted, the FCN may still be acceptable. In any other situation, however, the notifier must decide whether to fund potentially expensive animal toxicity studies, or whether using data available for structurally similar compounds or surrogates will be acceptable. In some cases, such as with nanoscale materials, uncertainty may exist regarding what types of studies and methods are even appropriate to evaluate toxicity. Structure-activity-relationship models are available that may assist with these decisions, but in most cases, the notifier would be wise to discuss questions of this type during a prenotification meeting with FDA.
Finally, although FDA has provided guidance for many different migration scenarios, the food packaging industry continues to push forward with new substances and applications that may not be covered under any current guidance. If a notifier plans to submit an FCN for a novel substance or use, a prenotification meeting with FDA can help the notifier avoid extra time and aggravation later in the process.
Nanoscale Materials in FCSs
Nanoscale materials are increasingly being used in food packaging for a variety of applications, and the use of these materials is expected to increase. The Project on Emerging Nanotechnologies, an open-access “living” inventory of consumer products that contain nanomaterials maintained by the Wilson Center and Virginia Tech, reports that 20 products in the “food and beverage storage” category that contain nanoscale materials are currently available for sale in the U.S., including beer bottles, aluminum foil, plastic wrap and sandwich bags.[5] Also, a recent study by Persistent Market Research estimated the global market for nano-enabled food and beverage packaging materials to be $6.5 billion in 2013 and predicted that it will continue to grow rapidly over the next decade.[6]
The advantages that nanotechnology offers in food packaging are substantial and stem from the unique physical and chemical properties of materials engineered at the nanoscale. For example, nanotechnology can reduce the weight of food packaging materials while also maintaining barrier functionality. Incorporating nanoclays such as montmorillonite into plastic food packaging materials improves the gas barrier and mechanical properties of the packaging beyond what can be achieved with conventional materials. Nanocomposites reduce the migration of gases, flavor compounds and water vapor across packaging material while offering superior performance under the stresses imposed during thermal food processing (i.e., heating to eliminate potential foodborne pathogens), transportation and storage.7
Human health and environmental concerns about using petroleum-based packaging materials (i.e., plastics) have led to increased interest in sustainable alternatives, some of which take advantage of nanotechnology. For example, biopolymer nanocomposites consisting of plant-derived materials as well as animal and microbial products are currently used as natural adhesives in food packaging. This application of nanocomposites may provide a safer alternative and superior performance compared with conventional chemicals, such as polyvinyl acetate, used for this purpose.
In addition to potential health and environmental concerns, poor mechanical and barrier properties make conventional-scale biopolymers ill suited for industrial applications. To address this problem, engineered composite materials, such as biopolymers layered with nanosilica particles, have been developed that exhibit improved gas barrier properties, tensile strength and thermal stability compared with standard biopolymers. These nanocomposite packaging materials provide effective barriers against water, gases and grease while also promoting the use of sustainable source materials.[7]
Nanoscale materials are also desirable for their antimicrobial properties. Nanoscale zinc oxide and titanium oxide particles exhibit photocatalytic properties and, when incorporated into food packaging materials, help degrade harmful volatile organic compounds and kill microorganisms.8 Silver nanoparticles are also used in food packaging for their antimicrobial properties. Studies suggest that nanosilver can kill 650 disease-causing pathogens in food, whereas most antimicrobials kill only 5–6 such pathogens.[8] Moreover, preliminary evidence suggests that silver may be less susceptible than traditional antimicrobials to the development of bacterial resistance.9 Nanosilver is also readily incorporated into various materials, such as textiles and plastics. In light of these reported benefits, several food-storage products currently available on the market claim that the nanosilver particles they contain help extend the shelf life of fruits, vegetables, breads, meats and other food items.[9]
Risks to Human Health
While there is significant interest in the food packaging sector regarding the benefits of nano-enabled materials, potential risks to human health and the environment have yet to be resolved. The properties that make nanoscale materials desirable for food packaging applications (e.g., increased surface reactivity and dispersibility) can also lead to unique interactions with biological systems that often differ considerably from those demonstrated by conventional-scale forms of the same substance. This disparity is further compounded by the sheer number and variety of different nanoscale materials that require evaluation, and by a lack of standardized laboratory methods for characterizing and testing these unique materials.
To date, hazard assessment of nanoscale materials used in food packaging has focused almost entirely on the raw materials, whereby freshly generated, pure nanoscale materials are administered to animals or cells. However, consumer exposure depends entirely on whether free nanoscale particles are released into the packaged food product. Such releases would likely be associated with transformation of various nanoscale material characteristics, including particle size, agglomeration state and surface reactivity. For this reason, academic and governmental research groups have investigated the migration of nanoparticles from food packaging materials into foodstuffs to better understand the risk of human exposure. Several studies report evidence of various nanoparticles (nanosilver, nanocellulose, nanoclay) migrating out of packaging materials at low levels and indicate that the total amounts increase with time and temperature and depend on the acidity of the food sample.8 While these factors also influence the migration of conventional-scale substances used in food packaging, the impact of temperature and pH on transformations of nanoscale materials released from FCSs has yet to be fully explored.
Furthermore, several methodological limitations make it difficult to extrapolate the results of studies showing migration of nanoscale particles into foodstuffs to real-world exposures. Often, chemical solutions representing extreme environmental conditions (e.g., very high or low pH) that are not characteristic of actual foodstuffs are used to model and measure migration. Considering that nanoscale particle migration is likely related to the compatibility between the particle, its embedded food packaging matrix and the foodstuff it contacts, these extreme conditions make estimating actual human exposure difficult. These worst-case testing conditions are also a factor in the migration of conventional-scale substances used in food packaging.
Finally, as nanoscale silver particles exemplify, migration is often measured by inductively coupled plasma mass spectrometry, an experimental technique incapable of distinguishing between the elemental and ionic forms of silver. This limitation is important, because evidence suggests that the mechanism and severity of silver-induced toxicity stem largely from release of silver ions, and, therefore, measuring the ionic form of silver accurately is key.[10] Clearly, more research is necessary to adequately characterize potential human exposure and risk associated with the migration of nanoscale particles from food packaging materials.
Regulatory Status in the U.S.
The toxicological hazards associated with engineered nanoscale materials are not well understood, and characterization of potential risks posed to humans and the environment is an active area of ongoing research. In light of the current state of knowledge, FDA released guidance documents in June 2014 to outline its general approach to nanotechnology and to provide specific guidance for using nanotechnology in the food industry.[11] The general guidance document reflects FDA’s current thinking that 1) nanoscale materials are an emerging technology of major importance, with the potential for novel applications across the entire spectrum of FDA-regulated products, and 2) although these materials may not be inherently safe or harmful, FDA will continue to consider the specific characteristics of individual products. FDA encourages manufacturers to consult with the agency early in the product development process to clarify specific scientific and regulatory questions related to the safety, effectiveness and public health effects of nanotechnological products. Notably, FDA did not establish regulatory definitions for “nanotechnology,” “nanomaterial” or “nanoscale,” and instead emphasizes that new technologies will be evaluated case by case. The guidance document for the food industry describes factors to consider when determining whether a change in the manufacturing process for an FCS affects its identity and safety. Such changes may impact an FCS’s regulatory status, thereby warranting a regulatory submission to FDA. However, the guidance suggests that for a specific case, depending on the data generated by the manufacturer, FDA may conclude that replacing a conventional-scale substance with a nanoscale substance does not warrant a new regulatory submission, and that FDA’s general FCN process is applicable.
In addition to the FCN process required by FDA, the same nanoscale materials used in packaging also face scrutiny from other regulatory agencies. Like FDA, EPA does not currently have specific regulations for manufacturing or using nanoscale materials. From January 2008 through December 2009, EPA initiated a pilot program for voluntary reporting of nanoscale materials.[12] The guiding principle behind this program was described as follows:
Nanoscale materials have a range of potentially beneficial public and commercial applications, including medicine and public health, clean energy, pollution reduction and environmental cleanup, and improved products such as stronger, lighter and more durable or conductive materials. These benefits arise from the distinctive properties of nanoscale materials, in that they are potentially more interactive or durable than other chemical substances. Altering the size of a material from conventional particle size can enhance or produce unique properties that are desirable for a variety of commercial applications. However, these unique and enhanced properties can raise new questions, such as whether the material in the smaller form may present increased hazards to humans and the environment.[13]
In 2009, EPA also developed a research strategy, identifying six nanoscale materials for toxicological testing based on their prevalence in commercial products.[14] Nanoscale silver and nanoscale titanium dioxide, two materials often used in food packaging materials, are included in this list. Currently, EPA has a preliminary framework for registering the use of nanoscale silver in consumer products. EPA has also received pressure from environmental groups to use its authority under the Federal Insecticide, Fungicide, and Rodenticide Act to regulate consumer products containing nanosilver, because of its well-known antimicrobial properties. In March 2014, EPA took action against Pathway Investment Corporation for making pesticidal claims for an unregistered use of nanosilver in plastic food containers. EPA prohibited sale of this product, stating that “Claims that mold, fungus or bacteria are controlled or destroyed by a particular product must be backed up with testing so that consumers know that the products do what the labels say.”[15]
On April 6, 2015, EPA issued a proposal for a nanoscale material reporting rule pursuant to Section 8(a) of the Toxic Substances Control Act (TSCA) and established a 90-day period for public comment.[16] This proposal, if finalized, would establish a regulatory definition for nanoscale materials, along with one-time reporting and record-keeping requirements for materials that meet this definition. Information required would include “specific chemical identity, production volume, methods of manufacture, processing, use, exposure and release information, and available health and safety data.”[16] EPA plans to use this information to decide whether further action is necessary under TSCA.
Summary and Conclusions
Human health risk assessment is the method FDA uses to evaluate whether the notified use of an FCS is safe. Risk assessment as applied by FDA to assess the safety of an FCS is quite prescribed; however, a number of nuances in this process can result in FDA denying an FCN. Aside from potential problems with the migration study data for a substance, which is relevant to the exposure assessment step in risk assessment, other challenges facing notifiers include, but are not limited to, insufficient toxicity data for a substance or conflicting data regarding the dose associated with potential health effects, and the ability to effectively communicate how these shortcomings affect the overall toxicological profile of a notified FCS.
Beyond conventional FCSs, nanoscale materials are increasingly being used in food packaging for a variety of applications, and such uses are expected to increase. The advantages that nanotechnology offers in food packaging are significant and include properties such as reduced package weight, improved gas barrier properties and superior performance under the stresses imposed on packaged food. Although there is significant interest in the food packaging sector regarding the benefits of nano-enabled food packaging materials, potential risks to human health and the environment have yet to be resolved. The science of hazard assessment for nanoscale materials and laboratory studies to estimate the amount of nanoparticles that may migrate into food are still being developed and are fraught with substantial uncertainties.
Interpreting toxicology data correctly and applying risk assessment appropriately are critical to preparing a successful FCN. Further complicating the process is the use of nanoscale chemicals as FCSs, because of the technological and scientific challenges in assessing potential exposure and toxicity of these chemicals accurately. Taking into consideration the FCS, its proposed use and the supporting data, a prenotification meeting with FDA may be a good idea. If a nanoscale chemical is the focus of an FCN, it is recommended to meet with FDA early in the process to discuss the data requirements and the best way to meet them.
Joel Cohen, Sci.D., is a toxicologist with Gradient Corporation, an environmental and risk sciences consulting firm. Dr. Cohen earned his doctoral degree at the Harvard Chan School of Public Health, where he investigated particle-cell interactions using various in vitro models for nanomaterial toxicity, cellular uptake and particle translocation. He developed a newly patented method for characterizing a critical particle parameter that drives transport in liquid suspension, enabling accurate estimation of delivered-to-cell doses for in vitro studies. He has authored several peer-reviewed articles and presented his work in the field of nanotoxicology to academic and general audiences.
Eric Dube, M.P.H., is a senior environmental health scientist at Gradient. He has over 20 years of experience in exposure study design and oversight, product- and site-specific risk assessment, safety evaluation of food additives, pesticide risk assessment and regulatory analysis. He has applied his technical and management expertise to a number of projects, including designing and presenting exposure studies to help refine the EPA’s risk assessment of registered wood preservatives, preparing and presenting studies to streamline the agency’s regulatory approval of fragrances used in registered pesticide products for food contact use, coordinating ad hoc industry coalitions to improve an EPA-required efficacy test method for antimicrobial pesticides and obtaining additional expert review of EPA’s draft assessment of inorganic arsenic.
Michael Peterson, M.E.M., DABT, is a senior toxicologist at Gradient with 17 years of experience specializing in human health toxicology and risk assessment and the application of these skills in the area of food safety. His experience includes evaluation of cancer and noncancer endpoints, critical and weight-of-evidence analyses of human and animal toxicology and epidemiology studies, and multimedia assessment of exposure to chemicals. His previous experience includes the evaluation of risks from food contaminants and additives, development of chemical toxicity profiles, assessment of the toxicity of novel chemical and commercial products, and determining microbial and chemical contamination risks associated with various food safety best practices. While earning a master’s degree in environmental management at Duke University, he researched the oral bioavailability of polycyclic aromatic hydrocarbons from soil.
References
1. Eaton, DL and CD Klaassen. 2001. Principles of toxicology. In Casarett and Doull’s Toxicology, The Basic Science of Poisons, 6th ed., ed. CD Klaassen. New York: McGraw-Hill, 11–34.
2. www.gpo.gov/fdsys/pkg/USCODE-2013-title21/pdf/USCODE-2013-title21-chap9-subchapIV-sec348.pdf.
3. www.gpo.gov/fdsys/pkg/CFR-2014-title21-vol1/pdf/CFR-2014-title21-vol1-part25.pdf.
4. www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/
IngredientsAdditivesGRASPackaging/ucm2021277.htm.
5. www.nanotechproject.org/cpi/browse/categories/food-and-beverage/storage.
6. www.persistencemarketresearch.com/mediarelease/nano-enabled-packaging-market.asp.
7. Arora, A and GW Padua. 2010. Review: Nanocomposites in food packaging. J Food Sci 75(1):R43–R49. doi:10.1111/
j.1750-3841.2009.01456.x.
8. Bumbudsanpharoke, N and S Ko. 2015. Nano-food packaging: An overview of market, migration research, and safety regulations. J Food Sci doi:10.1111/1750-3841.12861.
9. Duncan, TV. 2011. Applications of nanotechnology in food packaging and food safety: Barrier materials, antimicrobials and sensors. J Colloid Interface Sci 363(1):1–24. doi:10.1016/j.jcis.2011.07.017.
10. Kim, S and DY Ryu. 2013. Silver nanoparticle-induced oxidative stress, genotoxicity and apoptosis in cultured cells and animal tissues. J Appl Toxicol 33(2):78–89.
11. www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/ucm300661.htm.
12. www.gpo.gov/fdsys/pkg/FR-2008-10-31/pdf/E8-26026.pdf.
13. www.gpo.gov/fdsys/pkg/FR-2015-04-06/html/2015-07497.htm.
14. www2.epa.gov/chemical-research/research-evaluating-nanomaterials-chemical-safety.
15. yosemite.epa.gov/opa/admpress.nsf/0/6469952CDBC19A4585257CAC0053E637.
16. www.epa.gov/oppt/nano.
Nanoscale Materials: The Dose Makes the Poison