The role of airborne contamination of processed foods is controversial and fraught with contradictory opinions.[1,2} Some feel that airborne contamination of foods is of great public health significance, whereas others feel it is of little importance. Our perspective is that it depends upon the product, whether we are addressing a confined or an enclosed space, the type of processing equipment involved and whether indirect or direct contamination of the product or product contact surface is considered. The following article provides support for this balanced perspective. The reader is cautioned that although the following article is written from a food microbiology perspective, some aspects of air-handling unit (AHU) design are touched upon. However, a rigorous engineering treatment of concepts associated with AHUs should be sought elsewhere.
Bioaerosols Described
 Bioaerosols are solid or liquid microscopic particles suspended in air (aerosols) that carry microbes,[2] hence the term “bioaerosols.” A contaminated aerosol is likely to be comprised of injured cells (or spores), regardless of whether the aerosol is liquid (microscopic droplets) or dry (e.g., dust). In work performed in the author’s previous lab-oratory at the University of Georgia’s department of food science and Center for Food Safety, Yan et al.[3] found a 3-log reduction in the level of aerosolized Listeria monocytogenes after 20 minutes of circulating aerosols released into a 315-liter chamber at both 38 percent and 75 percent relative humidity (Figures 1 and 2).[2] However, there was only a 1-log reduction per hour after the air ceased to be circulated in the chamber. The level of cell injury was nearly 100 percent when a centrifugal air sampler was used, compared with sedimentation plates. In this study, 5.5-log CFUs of L. monocytogenes were released into the chamber, and agar strips in the centrifugal sampler were used to recover organisms released into this confined space. The agar strips were then removed from the sampler, macerated, diluted and plated onto nonselective (trypticase soy with yeast extract; TSAYE) agar, selective agar (modified Oxford medium; MOX) and TSAYE overlaid onto MOX agar. The result was the recovery of 5.8- and 5.4-log CFUs on TSAYE and TSAYE/MOX, respectively. No recovery was observed on MOX alone, which is more selective than other media that were designed to recover injured Listeria.[4] Hence, the level of cellular injury was nearly 100 percent with the centrifugal sampler. When the same study was performed with sedimentation plates, which, unlike centrifugal samplers, do not mix the air, less injury was observed. Thus, vigorous air movement correlates to vegetative cell injury. It is likely that cellular injury results from desiccation of aqueous aerosols. Dry aerosols are already desiccated, and hence, the presence of injured cells of nonspore formers can also be expected in dry aerosols.
Bioaerosols are solid or liquid microscopic particles suspended in air (aerosols) that carry microbes,[2] hence the term “bioaerosols.” A contaminated aerosol is likely to be comprised of injured cells (or spores), regardless of whether the aerosol is liquid (microscopic droplets) or dry (e.g., dust). In work performed in the author’s previous lab-oratory at the University of Georgia’s department of food science and Center for Food Safety, Yan et al.[3] found a 3-log reduction in the level of aerosolized Listeria monocytogenes after 20 minutes of circulating aerosols released into a 315-liter chamber at both 38 percent and 75 percent relative humidity (Figures 1 and 2).[2] However, there was only a 1-log reduction per hour after the air ceased to be circulated in the chamber. The level of cell injury was nearly 100 percent when a centrifugal air sampler was used, compared with sedimentation plates. In this study, 5.5-log CFUs of L. monocytogenes were released into the chamber, and agar strips in the centrifugal sampler were used to recover organisms released into this confined space. The agar strips were then removed from the sampler, macerated, diluted and plated onto nonselective (trypticase soy with yeast extract; TSAYE) agar, selective agar (modified Oxford medium; MOX) and TSAYE overlaid onto MOX agar. The result was the recovery of 5.8- and 5.4-log CFUs on TSAYE and TSAYE/MOX, respectively. No recovery was observed on MOX alone, which is more selective than other media that were designed to recover injured Listeria.[4] Hence, the level of cellular injury was nearly 100 percent with the centrifugal sampler. When the same study was performed with sedimentation plates, which, unlike centrifugal samplers, do not mix the air, less injury was observed. Thus, vigorous air movement correlates to vegetative cell injury. It is likely that cellular injury results from desiccation of aqueous aerosols. Dry aerosols are already desiccated, and hence, the presence of injured cells of nonspore formers can also be expected in dry aerosols. 
Creation of Bioaerosols
A bioaerosol may be created from foot and wheeled (e.g., forklifts, handcarts) vehicle traffic through standing water in which microorganisms have grown, application of high-pressure washers to contaminated surfaces, use of compressed air lines that do not have appropriate point-of-use filters in which condensate has accumulated and various unhygienic designs of AHUs, as described below. Bioaerosols will be suspended in the air for various lengths of time. The settling rate is roughly dependent upon the square of the particle diameter. Bioaerosols of vegetative (nonspore) cells liberated into an open space will become diluted and injured as they mix with the air in the environment. These bioaerosols will move from high to low pressure on air currents in the plant. This highlights the importance of appropriately filtered, positive-pressure air in rooms requiring elevated hygienic controls, such as filling and packaging rooms, compared with basic Good Manufacturing Practice (GMP) areas of the plant.
The author’s experiences in over 25 years of in-plant microbiological troubleshooting in approximately 700 food processing facility visits have demonstrated that traditional approaches to recover specific pathogens or spoilage organisms in bioaerosols from the air have not been effective. Nontraditional approaches have worked in limited ways. For example, enrichment of plates in which large volumes (high CFM) of air were impinged for an hour with a Mattson-Garvin sampler recovered Cronobacter spp. in one processing facility.[5] It is also likely that bubbling large volumes of air through enrichment media may also recover such organisms. However, this serves only to reinforce the point that in large volumes of air in open areas, detection of pathogens or specific spoilage agents is unlikely. Nevertheless, they may be present but likely at very low levels. When such levels come into contact with food, detection is unlikely unless growth to detectable levels occurs, as shown below. Despite this, it is prudent to monitor airborne microbial populations for hygienic indicators and take appropriate corrective actions when exceeding acceptable levels. Methods for microbiological testing of air have been summarized by Evancho et al.[6] and Moberg and Kornacki.[7] Sedimentation plates are the most traditional way to measure airborne contamination, and traditional cutoff values for food processing plant air exist. However, this approach assumes a fairly steady settling rate and does not take into account air movement or airborne populations per volume of air. The presence of dripping residues in areas above sedimentation plates will also result in aberrant data.
Bioaerosol Chamber
Much of the work described below was performed in a bioaerosol chamber constructed at the University of Georgia’s Center for Food Safety at the author’s laboratory between 2001 and 2003. The chamber is depicted in Figure 1. It contained the following: attached gloves, so that one could reach inside to manipulate items without compromising the chamber’s internal environment; a peristaltic pump to recirculate air; a nebulizer used to release L. monocytogenes; pans containing saturated salt solutions to adjust the relative humidity of the atmosphere inside the chamber; and, to the far right of the chamber, a microbiological filter.
 In one experiment, sedimentation plates containing TSAYE were placed into the chamber and 200 CFUs L. monocytogenes/L were released from the nebulizer. This high level is unlikely to occur in a food processing facility. Three plates were exposed for 15 minutes at 0, 30, 60, 90 and 120 minutes, respectively. No cells of L. monocytogenes were recovered (Table 1).
In one experiment, sedimentation plates containing TSAYE were placed into the chamber and 200 CFUs L. monocytogenes/L were released from the nebulizer. This high level is unlikely to occur in a food processing facility. Three plates were exposed for 15 minutes at 0, 30, 60, 90 and 120 minutes, respectively. No cells of L. monocytogenes were recovered (Table 1).
Exposure of Product to Listeria Bioaerosols
 Commercially available ham slices were biocidally treated and cut to fit into standard-size Petri dishes and subjected to L. monocytogenes bioaerosols at various levels (Tables 2 and 3). L. monocytogenes was not detected until 3 hours of exposure to 160 and 2,200 CFUs/L in these products, when held at 75 percent and 38 percent relative humidity, respectively. It is likely that the sodium lactate and potassium diacetate inhibitors present in the ham samples prevented recovery of these injured cells. Such levels (>100 CFUs/L) are unlikely to occur in food processing environments. Higher populations of L. monocytogenes released into the chamber resulted in an increased incidence of recovery.
Commercially available ham slices were biocidally treated and cut to fit into standard-size Petri dishes and subjected to L. monocytogenes bioaerosols at various levels (Tables 2 and 3). L. monocytogenes was not detected until 3 hours of exposure to 160 and 2,200 CFUs/L in these products, when held at 75 percent and 38 percent relative humidity, respectively. It is likely that the sodium lactate and potassium diacetate inhibitors present in the ham samples prevented recovery of these injured cells. Such levels (>100 CFUs/L) are unlikely to occur in food processing environments. Higher populations of L. monocytogenes released into the chamber resulted in an increased incidence of recovery.
Product Types
Given the data above, one is tempted to conclude that airborne Listeria is of no practical significance in the industrial food processing environment, but such a conclusion could be drawn only if the product is not conducive to the recovery of injured cells and subsequent growth, or if indirect contamination events cannot occur.
Potato is generally regarded as providing an excellent microbial growth substrate, as evidenced by its use in fungal recovery media (e.g., potato dextrose agar for yeast and mold testing). On a few occasions in the past, the author has observed detectable Listeria contamination of retorted potatoes. In each case, it occurred in the cooling cycle of a retort wherein a vacuum was released, drawing contaminant across valves and aspirating Listeria onto exposed product. In each case, this happened in a confined space, wherein Listeria would not have been diluted by large volumes of air, as would occur in an unconfined space, and cells landed in product conducive to the recovery of injured cells.
Listeria and microbes closely related to Salmonella called hydrogen sulfide-producing thermoduric Enterobacteriaceae[8,9] have been recovered from condensate, oil traps and low points in compressed air lines by the author. This has occurred despite upstream air-drying equipment. Sometimes, compressed air is used to blow residue off equipment in low-moisture food processing facilities. This is not generally recommended.[10] Yeast was recovered from condensate in the compressed air line used to blow dust out of plastic bottles that were then filled with salad dressing.[11] Furthermore, some equipment is powered by compressed air, and the release of such unfiltered air into the plant environment may become a source of indirect product contamination, as discussed below. Failure to employ point-of-use filters in such lines could result in cross-contamination in the plant environment and direct contamination of product contact surfaces, in some instances.
In the author’s experience, Listeria and Salmonella have both been isolated from air intakes associated with various AHUs in food processing facilities. This highlights the need for adequate prefilters and final filters on such systems, as discussed below.
Direct vs. Indirect Contamination
Direct Contamination
Direct contamination of food from the air may or may not be significant, depending upon the type of product and process. For example, it is improbable that detectable airborne contamination will occur when exposed product has intrinsic (e.g., low aw, presence of active preservatives) or extrinsic inhibitory properties (e.g., refrigeration, in some instances). In this context, more significant sources of direct measurable environmental contamination may be sought.[9] However, should these bioaerosols contaminate a product conducive to microbial growth, measurable contamination will occur given enough time at an appropriate temperature. Hence, contamination of milk with airborne psychrotrophic strains of Pseudomonas spp. or fruit juices with airborne Alicyclobacillus spp. or vacuum-packaged raw meats with spores of Clostridium estertheticum could prove a substantial risk to product quality.
Indirect Contamination
Given the above, airborne contamination of product may seem to be of a lower priority, if a producer is manufacturing a dry product or ready-to-eat (RTE) meat products containing microbial inhibitors. However, this perspective could result in a failure to install appropriately designed or filtered AHUs or to appropriately prioritize their maintenance and repair. Since trace numbers of undesirable microorganisms may be present in the air, the presence of areas in the plant conducive to growth and recovery of injured cells should be considered. Such areas may include standing water, sandwiched structures on equipment or in the facility that entrap wet residues or exposed wet insulation. Growth to vast numbers, in such areas initially inoculated via an airborne route, may occur. Should high populations of microbes from such areas initially inoculated from air, drip, drop, slough or otherwise be transferred to the product, then direct measurable contamination becomes more likely, even in food not conducive to microbial growth.
Some years ago, a source of Listeria contamination of an RTE meat product was determined to be from exposed insulation from an inlet window for a spiral freezer. Rising steam from hot product saturated the broken covering on exposed insulation on the inlet window, and this insulation became wet and contaminated with Listeria. Approximately every other product entering the freezer was observed to be dripped upon with this Listeria-contaminated fluid from the saturated insulation. How did the Listeria get into the insulation? The likely source was bioaerosol contamination (e.g., from spraying a contaminated floor with a high-pressure hose, traffic through contaminated standing water or release from an inappropriately designed AHU).
AHUs and HVAC Systems
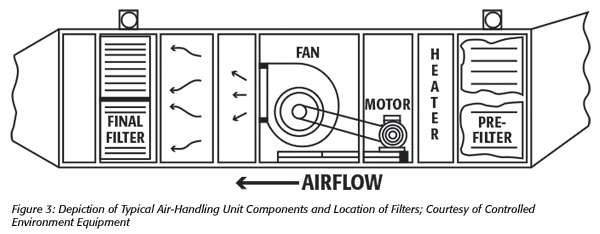 Inadequately filtered makeup air associated with AHUs could be one of several sources of pathogens and spoilage organisms in the plant environment. Makeup air that is heated and/or cooled must be filtered on both the inlet and outlet prior to entering the building envelope. Prefilters and final filters should be matched for airflow compatibility and pressure drop. The filter housing should also be designed to properly seal the filters to the filter frame and placed on an appropriate preventive maintenance inspection schedule. The inlet of the makeup air may have a minimum efficiency reporting value (MERV) 13 filter (MERV 8 in most cases is sufficient) and up to MERV 17 (HEPA) on the final outlet of the unit, as may occur with pharmaceutical production areas or “cleanrooms.” However, in many food processing facilities, these highest MERV ratings are not typically found and may not always be necessary, depending upon the product. The most restrictive filter needs to be on the outlet of the unit, since the makeup air unit can be a growth niche for pathogens, especially if the device has a cooling coil that will remove moisture from the airstream. In many food processing facilities, the final filter may be MERV 12 or 13. Figure 3 depicts a typical AHU, showing the placement of the prefilter and final filter.
Inadequately filtered makeup air associated with AHUs could be one of several sources of pathogens and spoilage organisms in the plant environment. Makeup air that is heated and/or cooled must be filtered on both the inlet and outlet prior to entering the building envelope. Prefilters and final filters should be matched for airflow compatibility and pressure drop. The filter housing should also be designed to properly seal the filters to the filter frame and placed on an appropriate preventive maintenance inspection schedule. The inlet of the makeup air may have a minimum efficiency reporting value (MERV) 13 filter (MERV 8 in most cases is sufficient) and up to MERV 17 (HEPA) on the final outlet of the unit, as may occur with pharmaceutical production areas or “cleanrooms.” However, in many food processing facilities, these highest MERV ratings are not typically found and may not always be necessary, depending upon the product. The most restrictive filter needs to be on the outlet of the unit, since the makeup air unit can be a growth niche for pathogens, especially if the device has a cooling coil that will remove moisture from the airstream. In many food processing facilities, the final filter may be MERV 12 or 13. Figure 3 depicts a typical AHU, showing the placement of the prefilter and final filter.
Other sources of contamination of the processing environment exist and may include soiled work clothes, street shoes, roof leaks, inadequately trapped floor drains, drain backups and negative air pressure in the plant drawing in air from contaminated outside areas. However, with regard to airborne particles, it is believed that liquid or solid particles of principal concern are between 5 and 20 microns. Particles smaller than 5 microns are likely to remain suspended in the air of a facility for an extended time and may be reduced by fogging with appropriate sanitizer. Particles larger than 20 microns are likely to settle quickly and can be removed by adequate cleaning and sanitization techniques applied to the plant environment.
HVAC Designs of Concern
Hugenholtz and Fuerst[12] found high populations (5–7-log CFUs/cm2) of microorganisms on cooling coils of supply air. In addition, 5–7-log CFUs/mL were recovered in condensate in cooling coil catch pans in two AHUs within a five-story library. Microbes recovered from cooling coils are likely to be psychrotrophic. The predominant species found on the supply coils in that study was Blastobacter spp. Both Gram-negative and -positive microbes were recovered from various parts of the air-handling system, including Pseudomonas, Bacillus, Flavobacterium, Acinetobacter, Cedecea, Arthrobacter, Corynebacterium and Staphylococcus. It is notable that the design of this unit did not have a final filter located after the cooling coils (in the direction of airflow). Not surprisingly, microbial populations recovered from supply air were on the order of 2–3-log CFUs/m3—a very low level indeed.
Condensate Drains
It is common to find HVAC condensate drain traps plugged with residue, thus preventing them from draining. This leads to accumulation of fluid and wet residues in catch pans that will grow microorganisms. Airflow across quiescent liquid surfaces of 4 meters/s (about 9 mph) will generate fine droplets.[13]
Other Organisms and AHU Designs
Psychrotrophic pathogens, such as L. monocytogenes and Yersinia enterocolitica, are of particular concern. Some years ago, Listeria spp. was isolated from a cheese product filling area. Vector samples suggested the source was an overhead AHU duct. Further investigation revealed that cooling water in an evaporative cooler was contaminated with Listeria, and bioaerosols were exhausted directly into the confined space of this product fill area. There was no adequate microbiological filter located after Listeria-entrained droplets entered the filler room, nor was the chill water biocidally treated. These changes were implemented along with other measures to clean and sanitize the filling area and the problem was resolved.
In another example, high yeast and mold counts were obtained on a filter located after cooling coils in an HVAC unit designed to cool a grinder for an RTE dry product. Examination of the AHU revealed that its cooling coils were located so close to the final filter that condensate was blown onto the filter, thus wetting the final filter matrix and permitting the development of yeast and mold populations, which contaminated areas in downstream ductwork and product.
Penetrated Supply-Side AHU Ductwork on Rooftops
On occasion, standing water will develop in ductwork due to penetrations and introduction of rainwater. This is of particular concern with supply-side ductwork. Such standing water has been observed with high populations of undesirable microbes. Ductwork needs to be minimized wherever possible. Product dust accumulations or entrapped moisture can create microbiological harborages or growth niches. One way to distribute the air in a facility is to use transfer fans installed so that the makeup air moves from the most hygienic areas of the plant to the least hygienic (e.g., basic GMP) areas. This approach is called “cascading.” To control the airflow, it is highly recommended to include variable-frequency drives on all of the transfer fans with pressure controllers to ensure positive air pressure in the higher-hygiene rooms compared with the basic GMP areas. Control technology is more affordable today with central programmable logic systems to enable this design to work efficiently and reliably. However, facility configurations do not always allow for well-designed cascading. High-hygiene areas and basic GMP areas can be interspersed. In such cases, air locks can be designed so that the high-hygiene rooms will have entrances with double door enclosures pressurized with filtered system fans.
Some Take-Home Messages Related to AHUs
1. Use HVAC units that have been designed with final microbial filters located after the cooling coils. HEPA filters are desirable, but if this is impractical, final filters should be designed to capture particles 5 microns or larger. The suggested MERV rating minimum is 12. In addition, MERV 8 or higher prefilters ahead of the coil are also recommended.
2. Such filters should be located as close to the plant environment as practical to prevent bioaerosols that may have developed as a consequence of moisture leaking through duct penetrations (i.e., on rooftop HVAC units).
3. Ductwork, especially on the supply side of the AHU, should be maintained and kept free of penetrations that may allow entry of contaminants and moisture. Minimize ductwork by utilizing the cascading concept described earlier in this article where possible.
4. Design HVAC systems to prevent wetting the final filter with coil condensate when supply-side coils are located too close to filters. Adjustment of airflow may also be necessary to prevent this.
5. Condensate drain traps should be maintained to prevent them from becoming clogged. Such drains should also be tapped to roof drains (for rooftop AHUs) to prevent creation of standing water on roofs that may leak into the plant.
6. Appropriate sanitizer blocks, pellets or crystals should be placed into condensate catch pans for HVAC units.
Conclusion
Undesirable vegetative microorganisms in liquid are rapidly injured and tend to die in air at a rate dependent upon the turbulence of and time in the suspending air. However, spores of spore-forming microbes will not be injured. These injured vegetative cells or uninjured spores will be diluted in large volumes of air in open spaces, which makes direct measurable contamination unlikely. Nevertheless, indirect or secondary contamination of product from structures that permit recovery and growth of airborne microbes can pose a problem. Products in which microbial growth cannot occur are likely to be less-directly affected, whereas products that are prepared and handled in a manner to facilitate microbial growth pose a greater risk.
The significance of airborne contamination is controversial and depends upon many things, including product type, product formulation, airflow in the plant, AHU design and design of the plant itself.
The author gratefully acknowledges the technical input and HVAC diagram depicting typical air-handling unit components provided by David A. Swinehart, vice president of engineering, Controlled Environment Equipment (www.cee-corp.com).
 Jeffrey L. Kornacki, Ph.D., is president of Kornacki Microbiology Solutions Inc. in Madison, WI. Dr. Kornacki has performed in-factory microbiological investigations, risk assessments and food safety-related audits for 25 years in approximately 700 food processing facilities in a variety of capacities with different organizations.
Jeffrey L. Kornacki, Ph.D., is president of Kornacki Microbiology Solutions Inc. in Madison, WI. Dr. Kornacki has performed in-factory microbiological investigations, risk assessments and food safety-related audits for 25 years in approximately 700 food processing facilities in a variety of capacities with different organizations.
References
1. Tompkin, R.B. 2002. Control of Listeria monocytogenes in the food-processing environment. J Food Prot 65:709–725.
2. Kornacki, J.L. and J.B. Gurtler. 2007. Incidence and control of Listeria in food processing facilities. In Ryser, E.T. and E.H. Marth (eds.), Listeria, listeriosis, and food safety, 3rd ed. Boca Raton, FL: CRC Press, 681–766.
3. Yan, Z., J.L. Kornacki, C-M. Lin and M. Doyle. 2004. Fate of aerosolized Listeria monocytogenes in a closed bioaerosol chamber. International Association for Food Protection. Annual Meeting. Abstract P058.
4. Yan, Z., J.B. Gurtler and J.L. Kornacki. 2006. A solid agar overlay method for recovery of heat-injured Listeria monocytogenes. J Food Prot 69(2):428–431.
5. Gurtler, J.B., J.L. Kornacki and L.R. Beuchat. 2005. Enterobacter sakazakii: A coliform of increased concern to infant health. Int J Food Microbiol 104:1–34.
6. Evancho, G.M., et al. 2001. Microbiological monitoring of the food processing environment, Chapter 3. In Downes, F.P. and K. Ito (eds.), Compendium of methods for the microbiological examination of foods, 4th ed. Washington, DC: American Public Health Association.
7. Moberg, L. and J.L. Kornacki. 2014. Microbiological monitoring of the food processing environment, Chapter 3. In Compendium of methods for the microbiological examination of foods, 5th ed. Washington, DC: American Public Health Association. Manuscript under final review.
8. Kornacki, J.L. 2014. An environmental sampling approach to product risk assessment. Food Safety Magazine 20(1):14–19, 74.
9. Kornacki, J.L. 2010. Principles of microbiological troubleshooting in the industrial food processing environment. New York: Springer.
10. Chen, Y., et al. 2009. Control of Salmonella in low-moisture foods II: Hygiene practices to minimize Salmonella contamination and growth. Food Prot Trends 7:435–445.
11. Clavero, R. 2010. Solving microbial spoilage problems in processed foods, Chapter 3. In Kornacki, J.L. (ed.), Principles of microbiological troubleshooting in the industrial food processing environment. New York: Springer, 63–78.
12. Hugenholtz, P. and J.A. Fuerst. 1992. Heterotrophic bacteria in an air-handling system. Appl Environ Microbiol 58(12):3914–3920.
13. Mitscherlich, E., and E.H. Marth. 1984. Microbial survival in the environment: Bacteria and Rickettsiae important in human and animal health. New York: Springer-Verlag, 725.
p>