Government regulations often seems like a burden to the industry as they typically come with new and sometimes challenging reporting requirements. But new regulations also present opportunities because they help to establish clear benchmarks for accountability that can promote higher quality. In industries where regulation is pervasive––such as the food industry––the most successful companies are typically those that develop systems and practices that maximize these opportunities for process improvement, higher quality and ultimately greater insight into the data capture necessary for regulatory reporting and control over finished product.
Governments worldwide hold their food industries to a higher standard, and rightly so. Even small errors in food production can do serious damage to public health and consumer confidence. Numerous rules exist to enforce quality and safety, from voluntary standards such as ISO 22000 to mandatory regulations such as the U.S. Food Safety Modernization Act (FSMA) or the European Union Regulation (EC) No. 178/2002. Additional regulations exist for specific food and food production processes, such as the Federal Meat Inspection Act or the Egg Products Inspection Act. Most of these regulations are based on trusted quality assurance and quality control paradigms such as the Hazard Analysis and Critical Control Points (HACCP) methodology, which has been widely used in food production for several decades.
Managing this myriad of regulations and standards is hard work, but food producers that take a systematic and software-driven approach to achieving compliance with them can actually make all these rules work in their favor.
Automating Compliance Data Management
FSMA Section 103, entitled “Hazard Analysis and Risk-Based Preventive Controls,” outlines the structure of a “preventive controls plan” based on the HACCP methodology.
The main challenge of implementing these controls is dealing with the incredible volume of data they produce. A comprehensive HACCP program produces thousands of data points each day, and this data is only as useful as the system that manages them. One of the best solutions for managing this flow of data is a laboratory information management system, or LIMS.
A LIMS collects and manages data over a food product’s entire lifecycle––from incoming ingredient shipments to finished product deliveries and all HACCP points in between. The LIMS helps food producers establish a new HACCP program or strengthen an existing one by guiding them through five critical steps: evaluating hazards, defining preventive steps, establishing monitoring controls, maintaining monitoring records and specifying corrective actions.
Hazard Evaluation
 Every food production process is different, so the first step in any compliance program is always identifying the unique HACCP points in a given facility. Food safety and food contamination risks most commonly occur where raw materials are introduced, vessels are opened, product is extracted, materials are added or finished products are packaged. Especially dangerous are the HACCP points that involve employee interaction––and by extension, the potential for human error.
Every food production process is different, so the first step in any compliance program is always identifying the unique HACCP points in a given facility. Food safety and food contamination risks most commonly occur where raw materials are introduced, vessels are opened, product is extracted, materials are added or finished products are packaged. Especially dangerous are the HACCP points that involve employee interaction––and by extension, the potential for human error.
Figure 1 shows a virtual facility map generated by a LIMS which lays out all hazard points within the production plant. Supplier data and outgoing shipments can also be mapped, allowing the facility to organize data by supplier, batch date, outgoing shipment or any other criteria. This capability makes it much easier to identify and manage the effects of process errors.
Defining Preventive Steps
Nowhere is the classic adage “an ounce of prevention is worth a pound of cure” truer than in the food production industry. Though a comprehensive HACCP program may be expensive, its cost pales in comparison to the costs of a wide-ranging product recall.
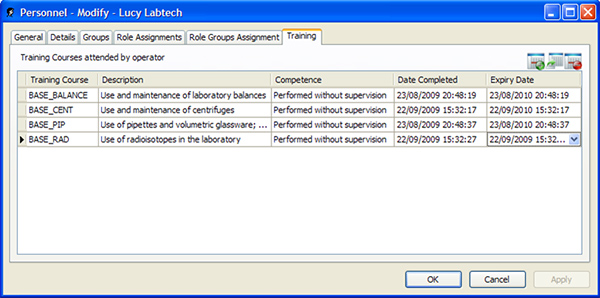 Improper instrument maintenance is one of the most common causes of food production errors. A LIMS can help food producers prevent these errors by providing staff with automated maintenance reminders. The system can also keep track of operator competency records, which allows both management and external auditors to ensure that all staff have received the appropriate training (Figure 2). Lastly, the LIMS can monitor the quality of raw and processed materials as they move through the production facility, identifying out of specification materials at the earliest HACCP point possible.
Improper instrument maintenance is one of the most common causes of food production errors. A LIMS can help food producers prevent these errors by providing staff with automated maintenance reminders. The system can also keep track of operator competency records, which allows both management and external auditors to ensure that all staff have received the appropriate training (Figure 2). Lastly, the LIMS can monitor the quality of raw and processed materials as they move through the production facility, identifying out of specification materials at the earliest HACCP point possible.
Establishing Monitoring Controls
Identifying hazard points and defining preventive steps represent only the initial planning phase of the compliance process. The bulk of the work occurs once monitoring actually begins. Fortunately, a LIMS that catalogues all hazard points and preventive steps enables food safety professionals to simplify monitoring by scheduling measurements, setting alerts, applying control limits and analyzing data for each one of a facility’s HACCP points.
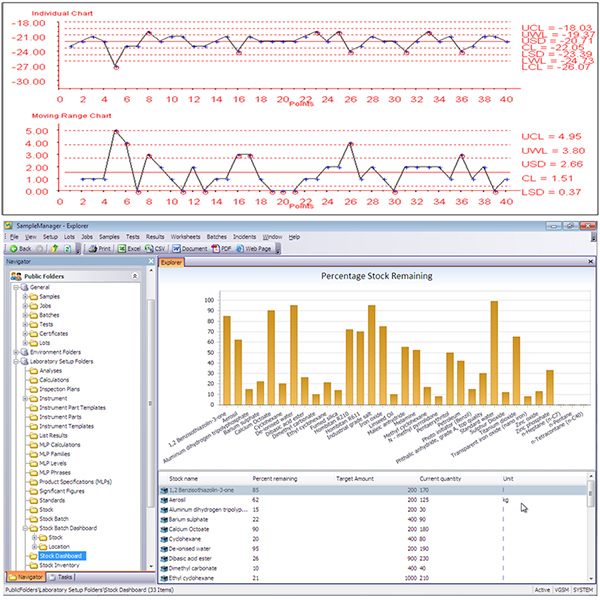 All data generated by process monitoring instruments installed at HACCP points are stored in a relational database built into the LIMS. This makes it easy to present information in whatever way is most useful for the intended audience(s). Options include graphs, process map overlays, real-time alerts and more (Figure 3). Control points can also be organized into groups, enabling users to further segment the data and establish consistent sampling plans for each subset.
All data generated by process monitoring instruments installed at HACCP points are stored in a relational database built into the LIMS. This makes it easy to present information in whatever way is most useful for the intended audience(s). Options include graphs, process map overlays, real-time alerts and more (Figure 3). Control points can also be organized into groups, enabling users to further segment the data and establish consistent sampling plans for each subset.
Maintaining Monitoring Records
HACCP monitoring data are only as good as the system that manages them. Data lose most of its value if it’s not organized into clear, comprehensive and easily understandable records. A LIMS simplifies record keeping and retrieval processes by enabling completely paperless data collection and reporting. Networking the LIMS with all HACCP monitoring instruments virtually eliminates manual data collection as well as the enormous potential for human error that manual work creates.
All records stored within the LIMS can be searched and organized according to many variables––including electronic signatures, origin and batch––making both internal and external audits much easier.
Specifying Corrective Actions
A food production facility’s error response time is often the difference between a controlled food safety incident and one that spirals out of control. A LIMS can significantly reduce a facility’s response time by storing clearly defined corrective actions for every potential incident. When and if an incident occurs, the LIMS can provide automated standard operating procedures that walk staff through the actions necessary to contain and resolve the problem.
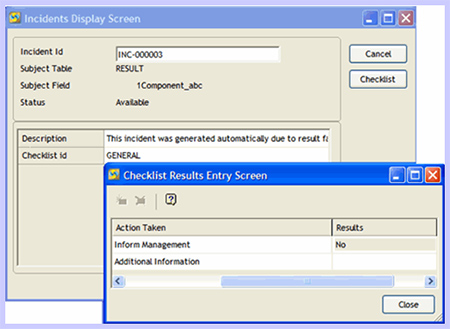 After the incident is contained, records stored in the LIMS can be used to determine the potential extent of the damage caused by the incident. This allows management to make more informed decisions about next steps, up to and including recalls. Once the incident has been fully resolved, detailed incident reports are stored within the LIMS for review by both internal and external parties (Figure 4).
After the incident is contained, records stored in the LIMS can be used to determine the potential extent of the damage caused by the incident. This allows management to make more informed decisions about next steps, up to and including recalls. Once the incident has been fully resolved, detailed incident reports are stored within the LIMS for review by both internal and external parties (Figure 4).
Conclusion
Solid data management systems are the catalysts that transform food safety regulations from burdens into opportunities. By handling as much of the facility monitoring, data collection and compliance reporting processes as possible, a LIMS allows food safety professionals to cut out much of the most time-consuming, error-prone and least value-added work as possible. By focusing on the beneficial consequences of meeting industry regulation, LIMS enables increased process efficiency and helps avoid product safety incidents altogether or minimizes the impact if any recalls do occur.
Colin Thurston is a project director at Thermo Fisher Scientific.



