Many analytical methods have been accelerated and improved with the help of innovative laboratory automation. A higher sample throughput is often needed to justify the associated investment in new capital equipment. In addition, established laboratory processes must be adapted to the new operating protocol, and the required technical expertise in the team must be present or has to be built up.
With partial automation of the critical process steps, where the execution and the results depend on the individual working style of employees, the accuracy and reliability of the testing results can be improved, and often at the same time, working processes can be accelerated. In contrast to full automation, a complete redesign of established laboratory processes and significant investment into costly laboratory equipment and highly skilled personnel can be avoided by clever partial automation.
Laboratory trials and proficiency testing have shown that the automated sample dilution provided by the Inlabtec Serial Diluter increases reliability and accuracy of food testing compared to the traditional manual test tube method. The automatic dilution process is completely independent of the different working styles and experience of the operator. Thus, the quality of the testing results obtained is increased, and the use of test tubes eliminated.
Serial 10-fold dilutions of homogenized samples for viable cell count determinations are part of the standard tests of most microbiological control laboratory testing food, beverages, animal feed, and cosmetics. For this purpose, 1 mL of the homogenized sample to be tested is diluted in 9 mL of sterile diluent over several dilution steps, resulting in a dilution factor of 10, followed by inoculation of agar plates with the diluted samples. After the given incubation period, the colony-forming units (CFU) are counted, and the concentration of viable organisms in the original sample can be calculated from the dilution factor. A representative sample and the correct serial dilution are the key factors in obtaining accurate results.
The international standard ISO 6887-1: 2017 defines the technical requirements for this fundamental microbiological technique as well as including some recommendations. For example, it is recommended to mix samples in test tubes with 9 mL of diluent for about 5–10 seconds using a vortex mixer. Lab technicians experienced in undertaking serial dilutions know very well that in addition to the mixing time, the speed of the vortex movement, as well as the angle of how the test tubes are applied on the vortex mixer, influence the degree of homogenization. Careful operation, as well as work experience, is the prerequisite for homogeneous dilutions leading to reliable and repeatable viable cell count determinations.
 In contrast to manual dilution in test tubes, the Serial Diluter automates and standardizes the mixing and dilution of samples in accordance with ISO 6887-1 in a functional and resource-optimized polyethylene bag (Figure 1). Pipetting becomes more convenient and precise as the sample is automatically rinsed out of the pipette tip to fully participate in the dilution process. This makes viable cell count determinations much more efficient and reliable, and the dependence of the findings on the individual way of working is considerably reduced, as the following independent experiments show.
In contrast to manual dilution in test tubes, the Serial Diluter automates and standardizes the mixing and dilution of samples in accordance with ISO 6887-1 in a functional and resource-optimized polyethylene bag (Figure 1). Pipetting becomes more convenient and precise as the sample is automatically rinsed out of the pipette tip to fully participate in the dilution process. This makes viable cell count determinations much more efficient and reliable, and the dependence of the findings on the individual way of working is considerably reduced, as the following independent experiments show.
The sample together with the pipette tip is put into the first bag, and the dispensing nozzle is connected to the tip. At the touch of a button, 9 mL of diluent flush the sample out of the pipette tip into the bag, creating an intense vortex; together with a sterile air blast, this process ensures a perfectly homogeneous mixture in less than 4 seconds. For a demonstration, see our demo videos.
Proficiency Tests Confirm: Automation Increases Reliability
Microbiological testing laboratories accredited to DIN EN ISO/IEC 17025 must regularly take part in proficiency tests. Proficiency testing is the testing of unknown samples sent to the participating laboratory by an approved testing provider. This allows verification of the quality of the current tests, as well as the assessment of the performance and reliability of the laboratory and its staff over time. An important performance parameter of proficiency testing is the z-score, which specifies the distance of a measured value from the mean in the number of standard deviations: z = (X - μ) / σ. Where X is the measured value to be assessed, μ is the mean value (reference value) and σ is the standard deviation of the normally distributed measured values of the laboratories involved in proficiency testing.
The assessment of the z-score is based on the following criteria:
- | z | ≤ 2 acceptable testing result
- 2 <| z | < 3 questionable testing result
- | z | ≥ 3 unacceptable testing result
The statistical basis for this is that normally distributed testing results are within ± 2 standard deviations with a probability of 95 percent and within ± 3 standard deviations with 99.7 percent.
The z-scores of an accredited food testing laboratory are shown in Figure 2. The z-scores obtained by the technicians in 2014 and 2015 using test tubes for manual sample dilution are between –1.65 and 0.65, thus within the acceptable range. Although identical prepared test tubes and testing equipment were used, the maximum difference in z-score for the same sample between two employees was 2.3 standard deviations. The only explanation for this questionable result is individual differences by the technicians in performing pipetting and mixing of the sample.
 The z-scores of the proficiency test 2016 after converting to using the Serial Diluter confirm the expectations regarding precision and reproducibility of the testing results (Figure 2). Using the diluter, all z-scores obtained by the participating technicians were in the range of ± 0.1, which means that all technicians involved—regardless of the level of experience with proficiency tests or individual differences in working style—delivered almost identical test results of highest accuracy.
The z-scores of the proficiency test 2016 after converting to using the Serial Diluter confirm the expectations regarding precision and reproducibility of the testing results (Figure 2). Using the diluter, all z-scores obtained by the participating technicians were in the range of ± 0.1, which means that all technicians involved—regardless of the level of experience with proficiency tests or individual differences in working style—delivered almost identical test results of highest accuracy.
Furthermore, Figure 2 shows that using test tubes and vortexer six z-score values are less than or equal to 0.5 and only one value is greater than 0.5. Negative z-score values indicate that the measured viable cell counts are lower than those actually present in the samples tested. What is the reason for the tendency of negative z-scores with the test tube technique, which is not detected if samples are automatically diluted with the Serial Diluter? Incomplete sample mixing could be the reason as results presented below show.
Incomplete Vortexing Increases Risk of Incorrect Results
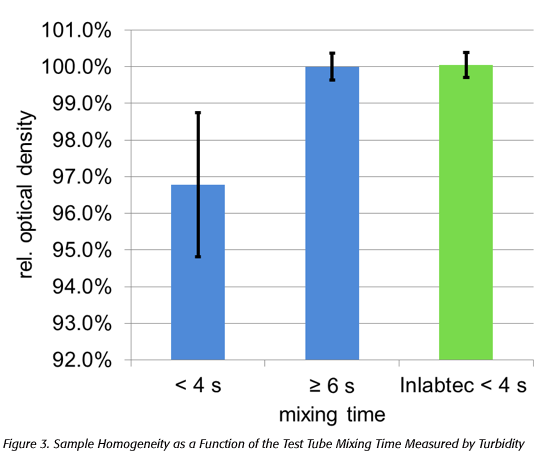 Experiments on the influence of the mixing time on the homogeneity of sample dilution showed that systematically lower bacterial counts can result after incomplete mixing of the samples in test tubes (Figure 3), and thus a potential risk for incorrect results exists.
Experiments on the influence of the mixing time on the homogeneity of sample dilution showed that systematically lower bacterial counts can result after incomplete mixing of the samples in test tubes (Figure 3), and thus a potential risk for incorrect results exists.
A homogenous dilution of 1 mL ultra-high temperature milk in 9 mL water in test tubes is achieved with at least 6 seconds mixing time [relative optical density (OD) at 820 nm = 100%]. Test tube mixing times of less than 4 seconds give rise to systematically low OD values with increased standard deviations (black bars). Using the diluter, the standardized process leads to complete sample mixing and homogenization in less than 4 seconds.
The systematic, negative deviation from 100 percent homogeneity due to mixing times of less than 4 seconds is due to the addition of the sample to the diluent in the test tube and the mixing process itself. Experiments showed that when adding 1 mL sample with a pipette, the aqueous sample is conveyed as a liquid jet into the lower part of the test tube. During mixing (vortexing), the sample is distributed in the filled test tube from the bottom to the top. The subsequent sampling of the diluted sample with a pipette takes place just below the liquid surface if good pipetting practices are applied to ensure accurate sample volumes. Thus, extinction values less than 100 percent means that the sample could not be distributed completely homogeneously in the diluent from the bottom to the top of the test tube (relative OD of water at 820 nm = 0%) and the mixing time was too short for the homogeneous dilution of the sample. Hence, a serial dilution over four steps with 95 percent homogeneity would lead to an underestimation of bacterial counts by about 20 percent.
Manual mixing with a vortexer is time-consuming and can lead to discomfort for the user. Sample mixing as short as possible is therefore understandable. In addition, there is no reliable visual evaluation of what is sufficient mixing. The ISO standard prescribes a mixing time of 5–10 seconds, which is not guaranteed automatically with a vortex mixer. In terms of food safety assessment, the manual test tube technique is not optimal, because typically many lab technicians are under time pressure and systematically low viable cell counts can result.
In contrast, automation of the dilution and the effective mixing process in the serial dilution bags deliver reliable results for every sample under all circumstances (see Figure 2). In addition, the process using the Serial Diluter is significantly more convenient for technicians than the test tube method. Eliminating manual mixing, long periods of handling of test tubes and caps, as well as simplifying pipetting reduce the risk of repetitive strain injuries. Washed is not necessarily clean.
To verify the diluter for routine use in an accredited food testing laboratory, viable cell count determinations of various food samples were carried out using in parallel self-prepared test tubes. Surprisingly, nearly up to five times higher total bacterial counts were detected with the Serial Diluter than when using the previously used self-prepared test tubes (Table 1). However, in the case of the parallel determination of Enterobacteriaceae in the same samples, no significant difference was observed between the two dilution techniques. The average difference in CFU was only 0.017 log CFU/g. An operator error with the Serial Diluter could thus be ruled out, and the diluent and agar plates used were identical.
 Based on these results, verification was repeated with purchased one-way prefilled dilution tubes from an external supplier. For total cell counts as well as for Enterobacteriaceae, no significant difference using both dilution methods was found afterward as might be expected (Table 1).
Based on these results, verification was repeated with purchased one-way prefilled dilution tubes from an external supplier. For total cell counts as well as for Enterobacteriaceae, no significant difference using both dilution methods was found afterward as might be expected (Table 1).
These results confirm the reservations of many laboratories to the multiple use of laboratory glassware, as growth inhibiting substances (e.g., detergent residues, secondary metabolites, etc.) can never be completely ruled out on cleaned glassware, which can influence the growth of sensitive cells. For this reason, ISO7218: 2007 recommends regular checking of the cleaned glassware for the absence of inhibitory substances.
The Inlabtec Serial Diluter uses disposable Serial Dilution Bags in polyethylene, which are highly pure and sterile, and reliably exclude the presence of inhibitors, eliminating the need for periodic inspection of glassware.
Partial automation using the Serial Diluter offers a cost-effective way to consolidate the quality of testing results at the highest possible level while also increasing the productivity of a testing lab.
Martin Jörg is laboratory manager quality testing of Microbact AG, an independent Swiss testing laboratory which is Good Manufacturing Practice-certified by Swissmedic and accredited by the Swiss Accreditation Service SAS. He can be reached at martin.joerg@microbact.ch.
Dr. Ernst Freydl is CEO of iNLABTEC AG. He can be reached at ernst.freydl@inlabtec.com.



