This past summer, Food Safety Insights conducted a survey of approximately 200 food processors throughout the U.S. on their concerns about their sanitation programs under the Food Safety Modernization Act (FSMA). We wanted to hear about their most pressing problems and how they are dealing with them. Because we learned that food processors have some anxiety about upcoming U.S. Food and Drug Administration (FDA) inspections and FSMA compliance, we also asked whether they had had a FSMA inspection and, if so, what was their experience.
The processors in this survey were located throughout 36 states and included processors in every major category, with 25 percent of respondents in processed packaged foods and with nearly equal representation from processors in meat/poultry/seafood, fruit/vegetables, spices and baked goods categories.
What about Sanitation Programs?
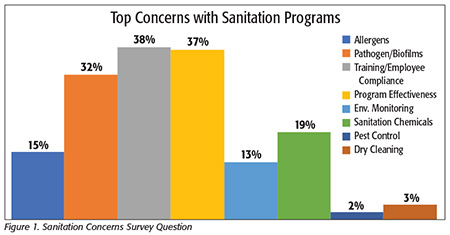 We first asked what processors felt were their biggest concerns with their sanitation programs (Figure 1). This was an open-response question where processors could indicate any concern in any category. The most frequent answer, cited by 38 percent of respondents, was employee training and compliance. Second was a general concern over whether their program was indeed effective and meeting all the requirements. The third-most common answer, cited by 32 percent, was control of pathogens and biofilms, with Listeria the most common species mentioned (specifically called out by 50% of those who cited pathogen control as a concern).
We first asked what processors felt were their biggest concerns with their sanitation programs (Figure 1). This was an open-response question where processors could indicate any concern in any category. The most frequent answer, cited by 38 percent of respondents, was employee training and compliance. Second was a general concern over whether their program was indeed effective and meeting all the requirements. The third-most common answer, cited by 32 percent, was control of pathogens and biofilms, with Listeria the most common species mentioned (specifically called out by 50% of those who cited pathogen control as a concern).
Processors are clearly signaling that major concerns with their sanitation programs are related to employee issues. Many of the comments we heard dealt with finding and retaining good people to work in the plant. When asked in a separate question about employee turnover in their sanitation staff, 59 percent of respondents agreed it was a “major issue,” with only about one-half of those indicating that it is a “manageable” problem and 5 percent indicating that the problem was severe, with turnover and position vacancies at least once having interfered with their ability to produce product. Only 41 percent indicated that they have “very low turnover” and do not consider this to be a major issue. When asked if they had seen any change in the turnover rate in the past 2 or 5 years, 55 percent said they had seen no change, with the remaining 45 percent equally split between reporting higher and lower turnover. Yet surprisingly, when asked if they offer retention incentives to sanitation staff, 80 percent indicated that they do not, with most commenting that any retention and incentive programs apply equally across all departments and not preferentially to sanitation or food safety staff.
These responses, however, do not seem to tell the whole story; it became clear from our follow-up interviews that this problem has a significant regional component and is far worse in some states than in others. Several of the individuals we spoke with who manage multiple facilities in different geographical areas confirmed that the issues are not uniform. In some states, the companies see no problems; yet in others, their issues with turnover may be critical. One processor reported, for instance, that their company operates facilities in three states, and while employee retention is somewhat of an issue at all of them, it was a particularly serious issue in one of their facilities in a state where the local unemployment rate is less than 4 percent, and they are seeing many new employers coming into the area. These new employers are not in the food processing business, and the situation was described as “…we are competing with offers of a first-shift position in a 72-degree warehouse versus a third-shift sanitation position in a cold food processing facility. That’s tough for us to compete with, and we are finding that we have positions that we cannot keep filled, and it is not uncommon to have a shift where we have 30–40 percent of the positions open.” Other processors reported similar problems, and it is clear from our survey that any national trends will not likely be as impactful on processors as the regional dynamics near their plants.
The respondents also indicated a general concern over knowing—or confirming—whether their program was effective. Comments such as “Are we finding everything?” “Do we have the right validations?” and “How do we know?” were common. Many of the concerns voiced were similar to what we have reported in the past in that processors are generally confident that their programs are effective, but they have some uncertainty about how their programs will be evaluated by FDA inspectors. Others expressed uncertainty over the measurement tools available to them, whether the tools are scientifically valid, if they work as they should and, specifically, if they “find everything.” It was clear from our interviews that at least some of these processors are hoping to find better analytical tools. One, for example, reported that they see “…a gap in the expectations of the FDA for validation of cleaning processes and the available validations and performances of the analytical tests available, especially in the multiple food matrices we work with.” Some of these analytical concerns were clearly related to issues around pathogen and biofilm detection, but processors also reported similar concerns about better tools for allergen, protein and chemical (sanitizer) residue detection.
Commonly Used Sanitation Methods
 With this concern about sanitation effectiveness and verification, we also wanted to know which methods the processors were using (Figure 2). Not surprisingly, the most common answer was “visual inspections.” In terms of test products, however, it was also not surprising, especially considering the concerns surrounding pathogens and biofilms, that the most frequently mentioned products were ATP swabs (65%) and growth-based microbiology testing (growth media and/or test swabs) for total plate count (63%). Approximately 47 percent cited using tests specific for allergens, whereas 16 percent cited the use of tests specific for proteins. No other technology was mentioned by more than 10 percent of the respondents, although respondents did mention using techniques and tests such as UV light, pH strips and tests for sanitizer chemical concentration.
With this concern about sanitation effectiveness and verification, we also wanted to know which methods the processors were using (Figure 2). Not surprisingly, the most common answer was “visual inspections.” In terms of test products, however, it was also not surprising, especially considering the concerns surrounding pathogens and biofilms, that the most frequently mentioned products were ATP swabs (65%) and growth-based microbiology testing (growth media and/or test swabs) for total plate count (63%). Approximately 47 percent cited using tests specific for allergens, whereas 16 percent cited the use of tests specific for proteins. No other technology was mentioned by more than 10 percent of the respondents, although respondents did mention using techniques and tests such as UV light, pH strips and tests for sanitizer chemical concentration.
We also asked whether they rotate the sanitizers that are used. In this sample, approximately one-third of respondents indicated that they do rotate the sanitizers, and the remaining two-thirds reported that they do not. This distribution did not vary significantly by type of processor, geography or any other parameter. We spoke with several processors who indicated that they do rotate sanitizers. They reported that they are following this practice not out of a concern for microbial resistance that may develop but more for a concern about the effective coverage of any single product. The rationale is that any single sanitizer may be less effective against a particular species or class of organisms, and by periodically changing the product used, these gaps in effectiveness are minimized by the overlap in efficacy of multiple chemicals.
Anxiety about FDA Inspections
Sanitation is a key component of FDA inspections, and many processors have been anxiously awaiting their first FDA inspection under FSMA, unsure about what they would face. Now that the inspections are underway, we wanted to find out more about how they are going and what the processors’ experiences have been. In this survey, we asked those facilities that are subject to FDA inspection (i.e., covered by FSMA and beyond their compliance deadline) if they had yet had an FDA inspection, with 22 percent indicating that they had.
At first, we did not find any correlation between the location (state) or type of processor and the likelihood that they had been inspected. This perhaps indicates that FDA is not focusing inspections on any one particular subset of food processing or in any one geographical area. In previous articles, we reported that FDA indicated that it would be focusing early inspections on facilities that had previous violations, but we did not ask any qualifying questions, so we are not able to report any trends in that area.
We did ask individuals at inspected facilities for their opinions about how the inspections went, and the clear majority indicated that their inspection “went well,” was “routine, with minor findings” and, as one individual at a dairy processing facility mentioned, “It was not as intrusive as we originally expected.” Another respondent, a dry grain processor, mentioned that their inspection “…went very well; it was more of a learning experience for both parties. The inspector acted like she was trying to learn about where we were with FSMA and gave us pointers on what we need to do going forward.”
Of the facilities that had been inspected, at approximately one-half of those inspections, FDA collected swab samples from the facility. As with the likelihood of inspection, we found no correlation between the plant location or types of foods being processed and the likelihood that the inspector collected swabs.
FDA has mentioned that it is planning to “educate before they regulate,” and the early indication from this survey seems to be that perhaps the agency is doing just that. It also seems that a processor is just as likely as not to have an inspector collect samples in their facility. With this understanding, processors can take the appropriate steps to prepare. We will continue to ask this question in future columns and keep you up to date.
Up Next
In our next column, we will take a closer look at how changing demand for testing is changing food safety analytical markets. Increased demand to test food for pathogens and other parameters is changing how food processors test their products, and this will have a significant impact on the business models of both diagnostic system providers and food commercial laboratories, potentially presenting significant opportunities for food processors.
Bob Ferguson is the managing director of Strategic Consulting Inc. and can be reached at
foodsafetyinsights@gmail.com or on Twitter at@SCI_Ferguson.
The New Face of Sanitation Programs: New Rules, New Challenges




