Continuum: A continuous sequence in which adjacent elements are not perceptibly different from each other, although the extremes are quite distinct.[1]
I am approaching the end of my career, and I am very aware that I was the last graduate student of my major professor at the end of a long academic career that started with his own graduate research characterizing microbiological changes in packaged meat in the early 1950s, when I was born. Generations of scientists have made their careers working on this issue, and it absolutely fits the definition of a “continuum;” you may not be able to see differences year to year, but the chicken of today is nothing like the “Chicken of Tomorrow” of 1951 (I recommend that you read the article referenced below):
“On a sunny June day in 1951, 8,000 chicken fans filled the Razorback Stadium at the University of Arkansas in Fayetteville in the culmination of a nationwide effort to create the fowl of the future. As a band played and the crowd cheered, the U.S. vice president Alben Barkley handed a California farmer named Charles Vantress a $5,000 cheque for his winning entry.”[2]
Today’s poultry is much more efficient in terms of genetics than anything previously seen, and a chicken in 2050, when there may be approximately 35 percent more people looking for wholesome and nutritious food, will be significantly different from today’s; it will absolutely need to be. And I hope it will be safer in addition to wholesome and nutritious, but this is where relativity may play a factor. If you think of this term in physics, relativity refers to how things like speed, sound and light appear differently depending on the factor being observed “relative” to the observer. Or this definition:
Relativity: The absence of standards of absolute and universal application,[3] which refers to no clear agreement of good or bad. How is “safety” defined for raw poultry? The concept of safety is relative to the individual. Purchasers of all food, including meat and poultry, expect the product to taste good and meet all quality expectations—without instilling fear of becoming ill; no one purchases food thinking, “I want to eat this, but it might make me or my family sick.” However, because there are billions of servings of any food per day, probability says some people will experience a foodborne illness related to poultry or other foods of animal origin. And it may have nothing to do with the poultry or meat except the food is a vector from the food handler to the consumer. Science tells us that any raw food of animal origin has some level of inherent risk. In other words, it can be either safe or unsafe depending on how it is handled prior to consumption. Pathogens such as Salmonella and Campylobacter are not adulterants of raw meat and poultry; they can be considered as naturally occurring or inherent at some level. This was exactly what Dr. Ron Chapman, director of the California Department of Public Health (CDPH) in 2013 said when explaining why the CDPH was not asking Foster Farms to recall chicken following a U.S. Department of Agriculture Food Safety and Inspection Service (USDA FSIS) public health alert:
“Chicken is a raw animal protein that is expected to have some level of naturally occurring bacteria present. Cooking chicken fully to 165 degrees Fahrenheit will kill the bacteria that are present. Provided that consumers do not cross-contaminate fully cooked chicken with raw chicken juices, it is safe to consume,” said Chapman.[4]
Will 2015 Be Different?
The year 2015 is shaping up to be possibly different in this continuum with several new aspects of food safety affecting poultry that began to be put into place in late 2014 and continues through 2015. We may see a noticeable difference in a relatively short time, but it also may depend on what we are watching to tell us of change.
First is the modernization of poultry slaughter inspection with implementation of the New Poultry Inspection System, or NPIS, in October 2014. As establishments express their desire to be governed under NPIS rules, FSIS is evaluating and ranking establishments into “clusters” to be scheduled for implementation based upon agency resources. All establishments had until February 23, 2015, to communicate to FSIS their intent to operate under NPIS. Establishments that did not choose NPIS by this date will continue under their current inspection system, with the notable exception of the Hazard Analysis and Critical Control Points (HACCP)-Based Inspection Models Project, or HIMP, broiler plants. These broiler plants either chose to go to NPIS with a waiver for the higher line speeds established under HIMP or will revert to the appropriate inspection system other than NPIS.
HIMP was introduced in 1997 and became the model for modernizing poultry inspection. HIMP established the concept of having company sorters on the evisceration lines identify and eliminate those defects, primarily quality related, that were handled by FSIS line inspection personnel previous to HIMP and now NPIS. In NPIS, each carcass is still inspected by a federal carcass inspector for wholesomeness as well as by an off-line verification inspector conducting verification tasks and sample collections important to public health tasking. However, the two to four FSIS line inspectors currently in traditional poultry plants are replaced by establishment personnel to sort, trim and take care of any reprocessing necessary prior to inspection by the single online FSIS carcass inspector. FSIS clearly states that HIMP plants demonstrated that establishment sorters were able to perform the online function comparably to federal online inspection personnel. With removal of the ability to have higher line speeds with NPIS, the government has also removed one of the incentives for choosing NPIS over traditional inspection. As is the case with most things, the line-speed provisions, as well as changing responsibilities on the processing line with the modernization of inspection, are not without controversy.
HIMP Brings Change
One of the major changes that occurred with HIMP was the ability to increase line speeds. Maximum line speeds in poultry plants were historically set by agreement with the bargaining unit of FSIS for how many birds the online inspector was to look at per minute. HIMP took those inspectors from the line, and the plants could run faster as long as they could demonstrate control of the evisceration process based on multiple criteria. In 2011, FSIS reported calendar year 2010-calculated average line speeds of 131 birds per minute for the HIMP plants with a line limit of 180 birds per minute, and 115 birds per minute for the non-HIMP plants with line speeds limited to 140 birds per minute.[5] All indications at the time of the first discussions of the Modernization of Poultry Inspection initiative were that the higher HIMP line speeds would become the standard and that conversion to the new system would not be voluntary.
As NPIS emerged from the Office of Management and Budget, only the current HIMP broiler plants will be able to transition to NPIS with their current line-speed maximums by also requesting a line-speed waiver. The years already spent with plants at a higher speed have not generated conclusive data on assessing whether the speed of the processing line is a benefit or a detriment to the consumer. The NPIS rule for turkeys is at the same line speed as HIMP, so a line-speed waiver is not necessary for turkeys.
The other variable to be measured would be the public health effect of the off-line verification inspector position and changes in tasking with NPIS. The final rule gives several tables of estimates of cost savings based on fewer people becoming ill from Salmonella and Campylobacter from poultry as a result of the improved regulatory oversight under NPIS over a 5-year phase-in period with varying levels of industry participation. Some of these estimates are substantial, but all are based on a change in illness rates attributable to poultry because of improved inspection.
Private Sector Initiatives
In addition to the changes made by USDA, there is a private-sector poultry safety initiative that was announced in December 2014 by Walmart/Sam’s Club in Bentonville, AR.[6] This initiative establishes new requirements and expectations that are broader than what is contained in the Modernization of Poultry Inspection because it establishes the expectations of the wholesale buyer for the farm-level operations of the poultry integrators that are not covered in FSIS regulations. The program also specifies standards of process control separate from USDA’s; processors must demonstrate a 4-log reduction in Salmonella from pre-scald to postchill and implement further interventions to achieve an additional 1-log reduction in Salmonella on parts.7 Unlike FSIS, the Walmart standard is not based on percentage positive for Salmonella, but on reducing quantity of Salmonella. Walmart is also not specifying standards relative to Campylobacter but may achieve some collateral benefit by Salmonella reduction. This is in addition to the current and continuing requirement that all suppliers meet Global Food Safety Initiative-approved third-party audit requirements.
One reason the Walmart poultry initiative came out in 2014 may have been due to partnering with the U.S. Centers for Disease Control and Prevention (CDC) to implement reductions in Salmonella. In a 2013 document, CDC talked about success in reducing Escherichia coli O157:H[7], and Walmart’s beef initiative in 2010 may have been a part of that success. Their goal and the title of the infographic is “Targeting Salmonella – A stubborn foodborne infection.”[8] Walmart may have also felt the time was right for a different approach to Salmonella control. To Walmart’s credit, some parts of the poultry industry were involved and informed as the Walmart poultry initiative was developed and were not surprised by the 2014 announcement.
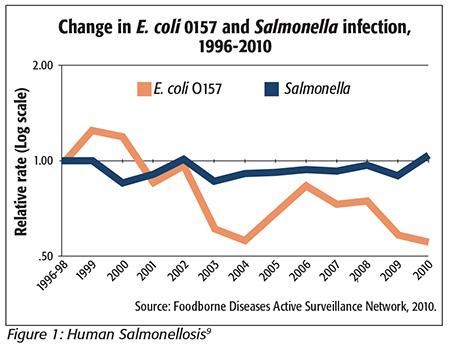 However, some people familiar with the poultry industry look at that same CDC graph of human salmonellosis (Figure 1[9]) and ask, “Since the industry is doing better with Salmonella reduction, why is the human illness line so flat over a 14-year period?”
However, some people familiar with the poultry industry look at that same CDC graph of human salmonellosis (Figure 1[9]) and ask, “Since the industry is doing better with Salmonella reduction, why is the human illness line so flat over a 14-year period?”
Human campylobacteriosis over that same period dropped fairly rapidly from just under 25 illnesses per 100,000 population in 1997–1999 with the implementation of “zero tolerance” and HACCP/pathogen reduction, and at a much lower rate of decline into 2003, where it has been very stable ever since, just above the Healthy People 2010 target of 12.4 illnesses per 100,000 population. It continues to run in the upper 13 to lower 14 per 100,000 population range, even though the Healthy People 2020 target was reduced to 8.5 illnesses per 100,000 population. The very fact of this stability calls into question the supposed benefits of off-line inspection that will somehow dramatically cut those numbers of illnesses.
Many, but not all, of the Walmart requirements were already in place or would be as a result of the Modernization of Poultry Inspection. One of the unique requirements is for poultry companies to vaccinate parent flocks against Salmonella serotypes known to be associated with human illness if found at the farm. This is in addition to encouraging and expecting their poultry suppliers to support changes in the National Poultry Improvement Program (NPIP), which also may be expanded to include serotypes of human health concern in addition to the poultry health program of today. Part of my concern with this approach revolves around unintended consequences. There is evidence that when the NPIP and other programs were so successful in eliminating the poultry-specific species of Salmonella pullorum and S. gallinarum, that a human pathogen, Salmonella Enteritidis, filled that void. When Israel reduced infections caused by Salmonella Enteritidis and Typhimurium from poultry, it experienced an increase in infections caused by a pathogenic strain of S. infantis in poultry.[10] With the multitude of Salmonella species and CDC compiling a list of the top 30 strains of human importance each year, what happens when you eliminate one over another? If it works, great, but only time will tell.
Timely Testing
Another interesting endeavor has been taking shape since the 2011 ground turkey recall conducted by Cargill. Immediately after the recall, FSIS wanted to prevent the outbreak strain from being in the product, and there was a second small recall soon after the ground turkey operation started back into production simply because the outbreak strain was detected, although there were no illnesses. Much of the attention at the time was on gauging how many samples of ground turkey were positive, a qualitative test. Cargill and other companies were also interested in knowing what the level of Salmonella was in a positive sample, a quantitative test. At that time, the only way to do that was by doing nine Salmonella tests using most-probable-number methodology. A result in 8 hours, using a cost-effective and reliable method of quantification and also compatible with Cargill’s needs, did not exist. Cargill worked with other companies and advisors to look at developing the concept of testing on a “semiquantitative” basis using PCR and transcription-mediated amplification technologies to determine if it could use “detection time” as a means of making risk-management decisions.[11] I recently got to hear a presentation from Cargill on how this is progressing within its system, and it is working on a peer-reviewed publication with its development partners. Cargill is comfortable with its approach of diverting product that may have a higher quantitative level of Salmonella to cooked product only and keeping it out of raw product retail distribution. Keep in mind, this is a raw product that would be acceptable on any regulatory basis, but the company is making its own decisions based on a human health-risk basis.
There is also increased attention to sanitary dressing/process control with the regulatory changes made with NPIS. The new regulations no longer mandate testing for E. coli biotype I, but do stipulate that broiler plants will test at a frequency based on volume and will test at both prechill and postchill locations. The establishments must develop and implement written procedures to prevent contamination by enteric pathogens and fecal contamination throughout slaughter and evisceration. They are also required to include microbial sampling to monitor their ability to maintain process control.
Which comes first—the chicken or the contamination? This is actually easy—they are linked as part of being an animal. Part of the terminology of “preventing contamination by enteric pathogens and fecal contamination throughout slaughter and evisceration” doesn’t make sense to me because the enteric pathogens and fecal matter are part of their life on the farm and are present on many if not all of the animals as they come to the processing plant. The slaughter and evisceration functions do have steps and processes that reduce that level of pathogenic microbes, but no process in the slaughter and evisceration will eliminate that to a safe-to-eat level; only through proper handling and cooking will foods of animal origin be safe. The 4-log Salmonella reduction in evisceration as required by Walmart is presumably predicated on the concept that contamination is already present and the goal is to get Salmonella to a lower and acceptable level that will equate to fewer foodborne illnesses in people. If it also works for Campylobacter, that will help as well.
I am hopeful that all of these programs signal change that will bring about a constructive dialogue on the issue of how safe is safe enough. I am also curious to know whether the Walmart initiative will change food safety to a competitive business issue. I always expect that the continuum will improve, but I don’t expect dramatic or maybe even noticeable changes in human salmonellosis rates anytime soon. I am also hopeful that I am wrong.
 John A. Marcy, Ph.D., is professor and poultry processing specialist in the Center of Excellence for Poultry Science at the University of Arkansas, Division of Agriculture, Research & Extension. Dr. Marcy’s role is to be an educational resource for the poultry processors of Arkansas through the annual Arkansas Poultry Processors Workshop and the Food Safety Conference in Branson, MO. Since 1994, he has been part of the Conference of Food Protection, which advises FDA on issues related to the FDA Model Food Code. The Institute of Food Technologists (IFT) recognized his many contributions by awarding him the designation of IFT Fellow in 2009. Dr. Marcy is currently in his second 2-year appointment to the National Advisory Committee on Meat and Poultry Inspection.
John A. Marcy, Ph.D., is professor and poultry processing specialist in the Center of Excellence for Poultry Science at the University of Arkansas, Division of Agriculture, Research & Extension. Dr. Marcy’s role is to be an educational resource for the poultry processors of Arkansas through the annual Arkansas Poultry Processors Workshop and the Food Safety Conference in Branson, MO. Since 1994, he has been part of the Conference of Food Protection, which advises FDA on issues related to the FDA Model Food Code. The Institute of Food Technologists (IFT) recognized his many contributions by awarding him the designation of IFT Fellow in 2009. Dr. Marcy is currently in his second 2-year appointment to the National Advisory Committee on Meat and Poultry Inspection.
References
1. www.oxforddictionaries.com/us/definition/american_english/continuum.
2. aeon.co/magazine/society/why-did-the-chicken-cross-the-world.
3. www.oxforddictionaries.com/us/definition/american_english/relativity.
4. www.cdph.ca.gov/Pages/NR13-047.aspx.
5. www.fsis.usda.gov/shared/PDF/Evaluation_HACCP_HIMP.pdf.
6. news.walmart.com/news-archive/2014/12/18/walmart-and-sams-club-to-enhance-poultry-safety-measures.
7. Walmart Stores. 2014. Poultry safety letter. December 17.
8. www.cdc.gov/ncezid/2011-2012-report/.
9. www.cdc.gov/ncezid/pdf/annual-report.pdf#page=76.
10. Mead, G et al. 2010. Scientific and technical factors affecting the setting of Salmonella criteria for raw poultry: A global perspective. J Food Protect 73(8):1566–1590.
11. www.foodprotection.org/files/food-protection-trends/NovDec-14-McEntire.pdf.
Raw Poultry: How Safe Is Safe Enough?




