From a food safety perspective, 2007 wasn’t a very good year for the food industry. Numerous recalls (with food pathogens being a major contributor) occurred, which has again weakened consumer confidence in food. Until early in 2007, the food industry appeared to be successfully rebounding from problems in the 1990s and the early part of this decade.
Food safety wasn’t one of the key fears in the public’s collective concern, having been replaced by bioterrorism, global warming, the economy and several other topics. However, 2007 proved to be a consumer confidence disaster and, once again, food safety now occupies the "front brain" of John Q. Public. Adding credence to the issue, recent CDC data demonstrated that incidence levels for the four key food pathogens have "flat-lined" over the past five years and continue to remain above target levels. As is often the case, changes in regulations follow such notoriety and are actively being debated.
The fundamental objective of any food company is to produce a safe and wholesome product that meets the claims of the product’s label. Recent data suggest that food plants are having trouble consistently achieving this objective. This is in spite of the significant investments made by the food industry over the past decade in plant design, production equipment, process changes, training and many other parameters. There is no question that huge strides have been made, but the data indicate that perhaps additional changes are needed.
HACCP has been a cornerstone of regulatory and industry practices since the 1990s. In fact, companies like Pillsbury have been using HACCP-based plans for over 40 years, which proved to be instrumental in the Apollo moon missions in the 1960s. The fundamental concept of HACCP is to control raw materials and maintain proper control of the production process/environment. If this is accomplished, you are guaranteed that the end product will be under control. HACCP is a preventative approach, rather than reactionary: take steps to prevent the problem from occurring rather than react to a problem after it occurs. When the HACCP parameter is physical or chemical (e.g., temperature, flow, pressure, moisture, foreign objects and certain chemical requirements), today’s measurement technology can provide near-immediate results at the point of sample collection—on the factory floor, in line and at the receiving dock. Given this, it is possible to achieve HACCP control, minimizing end-product issues and helping the food companies to meet their basic objective of producing safe food.
However, when microbiology is one of the HACCP parameters, food companies have great difficulty properly applying HACCP. With microbiology, it just takes too long to get results. Currently, the time-to-actionable results (TTAR), even with the best detection technologies, still take between 1 and 3 days. If assessment of microbial levels in raw materials is critical to a company, how can it be properly controlled? It is generally impractical to hold raw materials until results are received. Food companies do their best to push testing further back in the supply chain and rely on trusted suppliers. However, the testing for microbes in food results in a lengthy TTAR, putting the food industry in a reactive and not HACCP-based preventative mode.
Over the past 20 years, there have been significant advances in detection technologies and major adoption by the food industry of these newer testing methods. In spite of these advances, a significant gap still exists between the needs of food-processing companies and the ability of diagnostic companies to meet their needs. The gap is two-fold:
• TTAR—need information near realtime
• Distance—need analyses to be proximal to sample collection
There is a new report from Strategic Consulting, Inc. (SCI) entitled "Food Micro—2008 to 2013." The information in this report is based on hundreds of interviews with QA/QC managers in food companies worldwide. The report examines the microbiology testing practices at these plants, including the number of samples performed, sample location, which organisms are tested and which methods are used for analysis. In addition, the research also explores nonquantitative issues, including expected changes in testing practices and identification of the biggest needs for testing improvements.
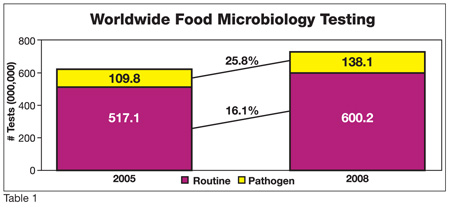 Worldwide food microbiology testing continues to increase. Since 2005, total microbiology testing volume has increased from 629.9 million to 738.3 million tests, which represents an increase of 17.8% over this three-year period (Table 1). Testing for food pathogens has grown even faster at 25.6% and now represents 138.1 million tests. Routine tests account for 81.3% of all food microbiology testing at 600.2 million tests. In 2008, the total market value for food microbiology testing exceeded $2.0 billion.
Worldwide food microbiology testing continues to increase. Since 2005, total microbiology testing volume has increased from 629.9 million to 738.3 million tests, which represents an increase of 17.8% over this three-year period (Table 1). Testing for food pathogens has grown even faster at 25.6% and now represents 138.1 million tests. Routine tests account for 81.3% of all food microbiology testing at 600.2 million tests. In 2008, the total market value for food microbiology testing exceeded $2.0 billion.
It is clear that the food industry has recognized the benefits provided by faster and better testing methods since, during the past 10 years, there has been a significant shift to newer, rapid methods. In 2005, rapid micro tests totaled 224.3 million. In 2008, rapid tests now total 306.8 million, which is an increase of 82.5 million over the past three years or 36.8%. In fact, the increased usage of rapid methods accounted for 74.1% of the increase in total microbiology testing over the past 3 years.
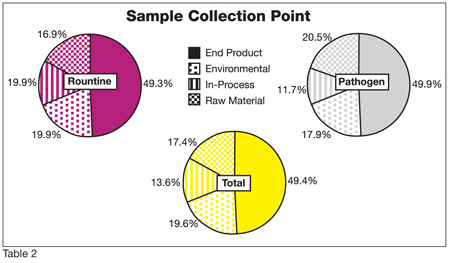 Under "ideal" HACCP conditions (real-time micro results available at the sampling point), theoretically there would be no need for end-product testing. Preventative measures and testing would guarantee control over the product produced, eliminating the need for end-product screening. However, endproduct testing currently accounts for almost 50% of all micro tests done in the food industry (Table 2). Why? Given the current TTAR for microbiology, food companies can’t guarantee prevention because of the time delays. Given this reality, when interviewed, 63% of plant QA/QC managers indicated that their company holds inventory while awaiting micro results, which is up from 52% in 2005. This practice is very expensive, especially when you consider the need to refrigerate many of the products, and is not, fundamentally, the best approach.
Under "ideal" HACCP conditions (real-time micro results available at the sampling point), theoretically there would be no need for end-product testing. Preventative measures and testing would guarantee control over the product produced, eliminating the need for end-product screening. However, endproduct testing currently accounts for almost 50% of all micro tests done in the food industry (Table 2). Why? Given the current TTAR for microbiology, food companies can’t guarantee prevention because of the time delays. Given this reality, when interviewed, 63% of plant QA/QC managers indicated that their company holds inventory while awaiting micro results, which is up from 52% in 2005. This practice is very expensive, especially when you consider the need to refrigerate many of the products, and is not, fundamentally, the best approach.
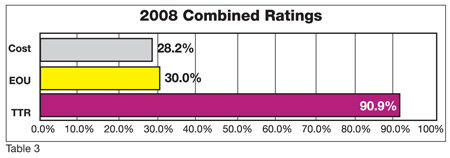 These QA/QC managers recognize the shortcomings of current micro testing methods that do not enable them to easily accomplish their jobs. When asked for their most desired improvement in micro testing, they overwhelmingly requested more rapid results. Over 90% of managers cited this as their biggest "wish." Ease of use and lower cost were also mentioned but less frequently. (Note: the numbers in the chart total more than 100% because many times it was difficult to limit the QA/QC managers to only one "wish"; Table 3.)
These QA/QC managers recognize the shortcomings of current micro testing methods that do not enable them to easily accomplish their jobs. When asked for their most desired improvement in micro testing, they overwhelmingly requested more rapid results. Over 90% of managers cited this as their biggest "wish." Ease of use and lower cost were also mentioned but less frequently. (Note: the numbers in the chart total more than 100% because many times it was difficult to limit the QA/QC managers to only one "wish"; Table 3.)
There has to be a better way. Perhaps some of the recent changes underway in the control of hospital-acquired infections (HAI) could represent a ray of hope and a path forward for the food industry (see sidebar). If such a food micro testing capability existed (a rapid, "all in one" testing system), it could be taken to the factory floor and provide microbiology results in near real-time. With such a test, microbiology HACCP parameters could be controlled before wasted value is added and/or potential problems could be proactively addressed. Raw materials above acceptable limits could be restricted from entering the factory. In-process testing could immediately flag when/where the plant is out of spec. Given such increased capabilities in control, there would be higher assurances that a company’s end product would satisfy expectations.
It is worth noting that over a decade ago, ATP surface hygiene testing was introduced to the food industry. While not a direct indicator of microbial contamination, ATP testing does provide immediate feedback to the sanitation crew as to the effectiveness of their cleaning efforts. This is the only quasimicro testing approach that does provide the food industry with at line, realtime information. During the past 10 years, the food industry has made ATP testing the norm; presently over 37% of all food plants worldwide have ATP systems and are performing more than 30 million tests annually. Similar capabilities for food micro testing of both raw materials and in-process conditions are needed.
With such testing capabilities, current micro practices could be rethought. Would companies still want/need to send their samples outside for analysis? Could they justify the extra cost/time associated with outside testing? Wouldn’t it make more sense to test at the plant where the sample is collected? Is this mere science fiction given current microbial detection capabilities? Maybe not, given the experiences of hospitals.
Clearly, microbial testing technologies continue to advance. Given the success in hospitals, hopefully these technologies will migrate to food testing and help to "bridge the gap" that currently exists. Maybe this type of technology won’t immediately provide the detection levels needed in the food market and perhaps there will need to be additional sample concentration efforts to aid the detection challenge. Maybe there will be a need to switch to the detection of indicator organisms that are present in higher levels and, therefore, are more easily detected. Regardless, I believe, eventually such efforts will "bridge the gap" and come to the aid of food-processing companies. When they do, we will all benefit through the improvements in food safety.
All charts are reproduced from the report, Food Micro—2008 to 2013, by Strategic Consulting, Inc.
Tom Weschler is president and founder of Strategic Consulting, Inc. He can be reached at 802.457.9933 or weschler@strategic-consult.com.
Is Rapid Micro in Hospitals at the "Tipping Point?"
Each year, hospital-acquired infections (HAIs) result in nearly 100,000 deaths and an additional $6.5 billion in health-care expenditures. Many of these infections are caused by antimicrobial-resistant organisms; methicillin-resistant Staphylococcus aureus (MRSA) ranks among the most prevalent pathogens in hospitals worldwide. As a result, hospitals are quickly moving to mandatory screening of incoming patients for MRSA. In just two hours, hospitals get MRSA results from a nasal swab taken from each incoming patient. With this information, the hospital is able to take the actions required to minimize HAIs.
How much different is a hospital from that of a food-processing plant? (Please grant me a few liberties with this analogy.) A hospital and a food-processing plant both have similar objectives: to meet label claims and achieve this purpose without contamination/infection. Both face huge economics loss and risk should they fail. Hospitals are beginning to recognize that to succeed, they must adopt HACCP-like practices, including near real-time control on their "raw materials". Thus, they are increasingly screening patients for MRSA using advanced detection equipment that can provide results in approximately 2 hours. With this information, they can sequester patients, as necessary, reducing the possibility of cross-contamination and infection and, thereby, of HAI. It has been shown that such testing yields significant savings/ benefits to hospitals that result from reductions in infection rates. Clearly, the reduction in loss of lives is even more fundamentally important.
Several diagnostic companies (e.g., Cepheid, BD, 3M and others) are benefiting directly from this new testing opportunity. The HAI market has generated significant notoriety for these companies and resulted in significant increases in the company’s stock market valuations. In one case alone, Cepheid’s market cap more than doubled in just the last 12 months.
Of critical importance is the design of some of these detection systems. They are "all in one" and "closed" molecular-based systems, which require limited sample prep and/or operator interaction. Microbial testing is getting to the point of "add sample and then get results." With a 2-hour TTAR and the easy-to-use format, testing can move out of the labs and much closer to the sample-collection point. It is my belief that such a success represents a "tipping point" in microbiology testing. Aren’t hospitals doing exactly what foodprocessing plants would love to be able to do—namely, apply HACCP in near real-time. With these rapid MRSA tests, hospitals are screening their incoming raw materials (patients) and have the ability to screen in-process factory conditions. Maybe successes like this represent the "next phase" in terms of changes needed to "bridge the gap" in food microbe testing. Such changes could represent dramatic changes in today’s current testing approach and landscape.




