The wake of the StarLink episode of last October has seen a confused and contentious landscape emerge regarding genetically modified (GM) foods, one characterized by a knot of discussions, regulatory initiatives, hastily devised monitoring programs and conflicting information. Confronted by this cacophony, consumer opinion waffles.
Despite this, bioengineered products designed to promote human health continue to show up on the radar. The most visible of these is Golden Rice, which carries three genes from the daffodil flower that produce beta-carotene, and is viewed by some as a panacea for combating vitamin deficiency in Third World countries. Raw potatoes have been modified to carry a vaccine against a strain of diarrhea-causing E. coli and, more recently, scientists in the U.K. and The Netherlands have used gene technology to increase levels of flavonols, a class of powerful antioxidants, in tomato peel. The researchers believe the fruit could help ward off heart disease and cancer.
It is these sorts of developments that, during the next three to four years, will drive the planting and consumer acceptance of genetically engineered crops, and bring significant change to the testing strategies and technologies employed throughout the food production community, according to a recently released market report.
Published by Strategic Consulting Inc., a market research firm in Woodstock, VT, the report, titled The World Market for GM-Food Testing concludes that during the years 2000-2005, improved technologies will deliver corn, soy and other hybrids with specific traits desired by food and other industrial customers, creating advanced agricultural products that are superior in quality and performance.[1]
“The impact on GM testing will be dramatic,” says Tom Weschler, president of Strategic Consulting. “End customers will need to ensure that they are getting what they paid for.”
Plantings to Increase in More Diverse Regions
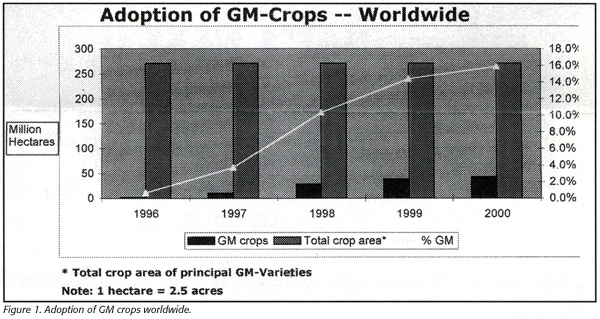 The report is based on surveys and in-depth interviews with representatives from agricultural companies, farms, grain elevators, food processing companies, regulatory agencies, testing laboratories and industry associations. Along with testing of GM foods, the report analyzes trends in plantings of transgenic crops, of which more than 40 varieties have been cleared through the U.S. federal review process. Roughly 43 million hectares of GM crops were planted worldwide in 2000 and the report predicts this will increase to 85 million hectares in 2005 (Figure 1). Soybeans, corn, cotton and canola are the dominant GM crops, and the report says that in 2000, GM varieties accounted for 34% of the 72 million hectares of soy planted globally, 16% of the 34 million hectares of cotton, 11% of the 25 million hectares of canola and 7% of the 140 million hectares of maize.
The report is based on surveys and in-depth interviews with representatives from agricultural companies, farms, grain elevators, food processing companies, regulatory agencies, testing laboratories and industry associations. Along with testing of GM foods, the report analyzes trends in plantings of transgenic crops, of which more than 40 varieties have been cleared through the U.S. federal review process. Roughly 43 million hectares of GM crops were planted worldwide in 2000 and the report predicts this will increase to 85 million hectares in 2005 (Figure 1). Soybeans, corn, cotton and canola are the dominant GM crops, and the report says that in 2000, GM varieties accounted for 34% of the 72 million hectares of soy planted globally, 16% of the 34 million hectares of cotton, 11% of the 25 million hectares of canola and 7% of the 140 million hectares of maize.
Crop plantings are concentrated geographically in three primary areas—U.S., Argentina and Canada— which account currently for 98% of the total area of GM plantings. The report predicts significant opportunity for growth, specifically in developing countries such as Brazil, India and China. It notes that about one quarter of the global transgenic crop area from 1997-2000 was in developing countries, which experienced strong growth in GM plantings compared with that in industrialized countries, where a slower adoption rate was experienced during the period 1999-2000 due to the fact that “farmers in industrial countries are becoming more concerned as consumers in some geographies become more skeptical,” the report says.
However, this skepticism is predicted to abate and pave the way for a greater volume and variety of crops. GM crops, particularly those planted in 2004 and 2005, increasingly will encompass what the report calls Generation 2 and Generation 3 output traits, which will be critical in mitigating consumer backlash against agricultural biotechnology.
Consumer Opinion Malleable
Opinion surveys about GM foods abound and offer varying conclusions, giving the overall sense that consumer opinion in the U.S. is in a state of flux. This was underscored by the recent published Pew Initiative on Food and Biotechnology. The core finding of the survey, according to executive director Mike Rodemeyers, is that “public opinion is ‘up for grabs’ because this new technology has moved faster than the public’s ability to fully understand it and its implications.”
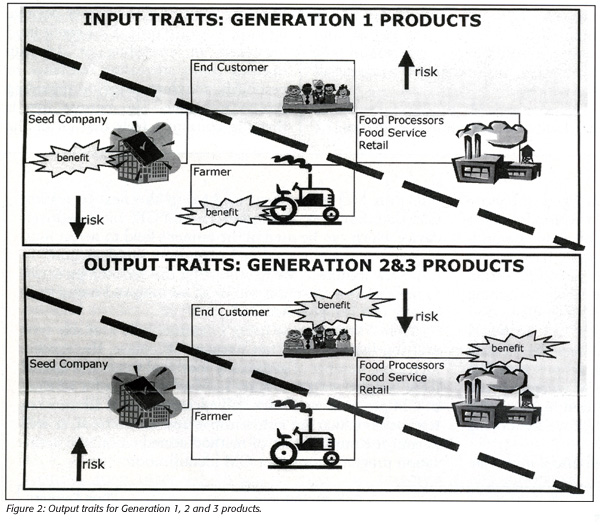 Conducted in January via telephone poll of 1,001 Americans, the Pew survey found that 46% of respondents did not know what to think about the safety of GM foods, and that those with an opinion were evenly split, with 29% believing GM foods are safe and 25% indicating they are not. Safety is just one component of what the Strategic Consulting report identifies as the perceived risk problem (Figure 2). With Generation 1 input traits, the perception is that consumers absorb the perceived risk of GM foods while the food producers accrue all the benefits.
Conducted in January via telephone poll of 1,001 Americans, the Pew survey found that 46% of respondents did not know what to think about the safety of GM foods, and that those with an opinion were evenly split, with 29% believing GM foods are safe and 25% indicating they are not. Safety is just one component of what the Strategic Consulting report identifies as the perceived risk problem (Figure 2). With Generation 1 input traits, the perception is that consumers absorb the perceived risk of GM foods while the food producers accrue all the benefits.
“Today’s GM products, such as StarLink corn, are termed Generation 1,” Weschler explains. “They yield positive input traits which primarily benefit food producers in extending or delaying ripening times, increasing resistance to pesticides and herbicides or providing disease resistance. The bottom line is that the consumer feels that all of the advantage is for the grower while the consumer bears all of the perceived risk for Generation 1 GM products.”
Generation 2 traits also benefit food producers in reducing processing, energy, storage or transportation costs. Examples here include low-fat potatoes, longer shelf-life tomatoes and soy products with high oleic acid expression that mimic the taste and health benefits of olive oil.
Conversely, Generation 3 products yield output traits that directly benefit the consumer, such as edible vaccines, anti-cancer vegetables, cholesterol-reducing grains and crops fortified with micronutrients. It is the continued development of foods yielding these benefits, along with improved education, will prompt better consumer trust of genetic modification. These products will appear with increased frequency in 2004 and 2005.
Testing to Assure Presence and Absence
The emergence of Generation 3 products also will bring about a change in GM testing strategies. Currently, most testing, while mandated by food processors and retailers, is pushed back to their ingredient suppliers and to grain elevators. The third-generation, value-added products “will shift the test burden to food processors, who will become more vertically integrated and institute identity preserved systems for added assurance that their products contain the positive genetic traits,” Weschler says. In other words, there will be a gradual shift from testing to assure the absence of genetically modified material to testing to assure the added value is present.
Weschler and others believe this will precipitate a steady increase in the volume of tests conducted. The Strategic Consulting report predicts that the number of tests conducted for GM food will rise roughly ten-fold from 2000 to 2005. Testing technologies used in the US currently are divided evenly among polymerase chain reaction (PCR) (35%), lateral flow immunoassay (35%) and enzyme-linked immunosorbent assay (ELISA) (30%), according to the report.
“There will be sustained testing the next few years specifically for StarLink, as well as to meet non-GM contract needs,” Weschler says. “Testing needs will change through 2005 with the emergence of Generation 2 and 3 products, shifting from primarily meeting non-GM contract requirements to ensuring that desirable traits are present in the product.”
Bruce Flickinger is a freelance writer and editor specializing in the food and pharmaceutical industries.
Reference
1. Weschler, T. The World Market for GM-Food Testing. Strategic Consulting Inc., 2014 Grassy Lane, Woodstock, VT 05091. www.strategic-consult.com.
GM Testing & Analysis: No One-Stop Method
Respondents to the study, The World Market for GM-Food Testing were unanimous in indicating that currently there is no ideal test for GM foods. Such a test would be versatile enough to detect universal genetic modification, enable users to set customizable thresholds and be validated by a recognized, international organization.
 One thing is certain: The Aventis CropScience StarlinkTM corn recall last October has upped the ante on improving detection and traceability of GM crops and foods. As a result, the U.S. government, scientific trade organizations, food laboratories and test kit and analytical system manufacturers have increased efforts to meet the analytical methodology challenges posed by genetically engineered foods and new demands from food processors who wish to separate GM from non-GM products.
One thing is certain: The Aventis CropScience StarlinkTM corn recall last October has upped the ante on improving detection and traceability of GM crops and foods. As a result, the U.S. government, scientific trade organizations, food laboratories and test kit and analytical system manufacturers have increased efforts to meet the analytical methodology challenges posed by genetically engineered foods and new demands from food processors who wish to separate GM from non-GM products.
“What everyone would like is a simple test that could quickly and accurately differentiate GM from non-GM grain, and ideally further identify approved varieties from non-approved varieties,” says Dean Layton, vice president of marketing and sales at EnviroLogix, Inc., a leading GM test kit manufacturer. “Unfortunately, the ‘magic bullet’ doesn’t yet exist, and given the dynamic nature of international approvals, the feasibility of that ultimate test seems unlikely, as it would be targeting a constantly moving target.
“Practical solutions will likely center on identity preservation (IP) programs that track and document the GMO status of seed and grain throughout the system,” Layton adds. “These IP programs utilize rapid screening tests such as lateral flow strips or enzyme-linked immunosorbent assay (ELISA) microwell plate assays, perhaps followed by polymerase chain reaction (PCR) confirmation.”
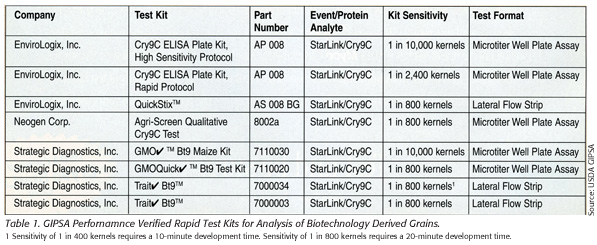
In response to the Starlink biotech event, the U.S. Department of Agriculture’s Grain Inspection Packers and Stockyards Administration (USDA/GIPSA) Federal Grain Inspection Service (FGIS) instituted a performance verification program to evaluate rapid tests for the detection of the presence or absence of biotech products. The program focuses on two of the most used techniques for GM detection: microtiter well ELISA technology and lateral flow strip ELISA technology, both designed to detect the presence of GM grains through the detection of a specific protein produced in the biotech grain. The former provides quantitative and qualitative results using antibodies incorporated into microtiter wells and enzymatic, colorimetric reagents for detection, while the latter typically provide qualitative results using antibodies and color reagents incorporated into a lateral flow strip. These tests usually provide screening results in five to 30 minutes. GIPSA plans to incorporate verification of PCR tests in the future. Table 1 lists the rapid test kits that have received verification from the GIPSA program.
 The analysis of more complex food matrices requires the third most commonly used technique, PCR, a highly sensitive technique that allows the analyst to extract and isolate genomic DNA of a sample, followed by amplification of the purified DNA. More complex products include foods such as those that contain blends of ingredients, processed foods and finished products range from corn flakes, flour, vegetable oils and fats, to chocolate, cakes and bakery products, honey and meat and sausages. PCR analysis used in for the detection of GM foods can be used as a screening tool or for genetic trait quantification and qualification, for which time to result can range from 20 minutes to two days.
The analysis of more complex food matrices requires the third most commonly used technique, PCR, a highly sensitive technique that allows the analyst to extract and isolate genomic DNA of a sample, followed by amplification of the purified DNA. More complex products include foods such as those that contain blends of ingredients, processed foods and finished products range from corn flakes, flour, vegetable oils and fats, to chocolate, cakes and bakery products, honey and meat and sausages. PCR analysis used in for the detection of GM foods can be used as a screening tool or for genetic trait quantification and qualification, for which time to result can range from 20 minutes to two days.
Mike Russell, president of GeneScan USA, a laboratory specializing in quantitative and qualitative GM food product and animal feed analyses, notes that the company’s worldwide network of laboratories based in Europe, have experienced a remarkable increase in the number of complex food matrix samples sent in for analysis. “We’re seeing everything from foundation seeds to highly processed ingredients and finished products. We continue to develop new analytical procedures as an ongoing process to address the steadily growing number of genetically modified food products, but we primarily use PCR, and immunoassay when appropriate for the sample. With the more complex food matrices that we are seeing, we must use PCR, especially if we are looking for a specific protein.”
Russell adds that GeneScan has entered into a joint venture with Motorola Corp. to develop the next-generation GM detection test: a handheld, non-PCR, biochip-driven device which can be used in the farmer’s field to detect multilayered genetically modified organisms simultaneously in just a few minutes. “We’re estimating this technology is two to three years away, but it will be an exciting and useful technology that is highly needed.”
Table 2 lists some of today’s commercially available tests utilizing the top three test formats. Of course, the steadily increasing number of genetically modified foods and animal feeds coupled with consumer activists’ demands for labeling of GM products worldwide will encourage more advances in test methods that are fast, sensitive and economical, as well as spur the introduction of method accreditation and verification programs for better GM identification.
Traceability to Boost Confidence in U.S. Grain Exports
Following the discovery of Starlink corn in consumer food products, world confidence in U.S. grain integrity has declined. According to U.S. Department of Agriculture (USDA) projections released in March, corn exports will slip by at least 50 million bushels. During the Food Safety Summit, held in Washington, DC, in April, online grain auditing pioneer CropVerifeye.com LLC, which has built a field-based, Web-enabled traceability program, addressed these issues, presenting traceability as a solution to the threat of decreasing exports and product recalls.
“Biotechnology isn’t a ‘GMO or non-GMO’ issue. It’s actually a consumer- driven issue, but without global consensus about what makes food safe,” said Jim Mock, Ph.D., CropVerifeye co-founder and Summit speaker. “We need to show the world that the U.S. agricultural industry is capable of helping food companies offer grain and vegetable choices to meet consumer demand—wherever those consumers might be and whatever their preferences.”
According to CropVerifeye, the best way to deliver grain that meets grain processors’ expectations is through a traceability system that begins in the “The idea of grain traceability is not really that different from the ‘seed to cellophane’ approach of the meat industry,” Mock said. “Adopting a similar system for grain would make Starlink a non-issue.”
Because crop traceability systems are fairly new to the marketplace, industry adoption is in its infancy. But food processors and exporters “have got to stop looking at traceability as something that’s ‘nice to have,” said Mock. “With shrinking export markets and new biotech varieties for wheat and rice on the horizon, traceable crops are an absolute necessity. And the cost is nominal, especially when you think about the $100 million to $1 billion that any food company could accumulate in potential liabilities. And these figures don’t include losses in brand equity. Food processors today can’t afford to live in a commodity age.”
Mock believes consumers will continue to drive the demand for grain tracking. “Today’s consumers are more sophisticated, but we just don’t know how interested they’ll be in the minute production details of the food they buy. Will they have the time or desire to read about the origination of the wheat in their favorite bread? It’s possible. Or maybe brand assurance is all they need.
“What is clear, however, is that food safety will be a top consumer concern. Producers, processors and others in the food chain will need a data trail to back up that brand promise, whether consumers ever see it or not.”



