Last year was a very unusual year in food safety. This year, however, may be even more unusual and show that 2018 was not an aberration but a harbinger of a change to what some may call a “new normal.”
What exactly am I talking about? In 2018, we saw an unusually high number of foodborne illness outbreaks and recalls. The year started out with a large recall of romaine lettuce, ended the year with another large recall of romaine lettuce, and in between, we saw more recalls of ground turkey, ground beef, pet food, pre-cut melons, vegetable trays, breakfast cereals, and the world’s largest Listeria outbreak (to date) in South Africa.
We have certainly seen foodborne illness outbreaks and food recalls in the past, but those from this past year were different. The recalls listed above were prompted by epidemiological investigations of outbreaks where the causative pathogens were recovered from ill patients, and identified and traced back to their sources through whole-genome sequencing (WGS). None were detected through routine surveillance by food processors. Also, in most of these incidents, the food volume being recalled had most likely already been consumed and caused most of the illnesses it was going to cause. This was the case for the recall of ground beef in the last few months of the year in that regulators acknowledged that much of the product had already been consumed, but the recalls were primarily targeted at those consumers who may have kept some of the product in their freezers. In the past, without WGS, this ground beef outbreak may not have been recognized as a foodborne illness and almost certainly would not have been linked to its source. Now, however, these incidents are being accurately traced to their source, and alerts and recalls are showing up on the screens of everyone’s smartphone and on TV news stations.
WGS Improves Traceability
This change is echoed in official statements by U.S. Food and Drug Administration (FDA) Commissioner Scott Gottlieb. In an FDA statement released on June 28, 2018, referring to the romaine lettuce Escherichia coli outbreak from Yuma, Arizona, the commissioner was quoted as saying:
“…In recent years, the FDA and the [U.S.] Centers for Disease Control and Prevention (CDC) have advanced new tools that make it easier and faster to identify outbreaks of human illness and to link them back to the food source that is the culprit responsible for the illnesses…. While there have been many advances over the past years, one of the most powerful of the new technologies we employ is whole-genome sequencing…. By using this technology, public health investigators can link illnesses in different people and locations to reveal outbreaks with a level of precision never previously available. Simply put, this means we’re better at identifying outbreaks and their source.”
This use of WGS will completely change our understanding and assumptions of what constitutes adequate pathogen surveillance and sanitation in the food industry.
If that’s not enough to get your attention, as of last month, the pace of this change has likely accelerated. In mid-January, CDC accelerated their transition of their PulseNet network to reliance on WGS from the previous primary use of pulse-field gel electrophoresis (PFGE). This change has been coming over the past few years as the number of WGS isolates added to the PulseNet database annually has grown from 0 in 2013 to more than 100,000 by the end of 2018—representing well more than one-half of the new isolates in the database. The use of PFGE ceased on several organisms at the end of 2018, and the priority in 2019 will be to exclusively use WGS on Listeria monocytogenes isolates, followed by Shiga toxin-producing E. coli, and then as many Salmonella samples as capacity allows. CDC had already transitioned Listeria isolates from PFGE to WGS by the end of 2018. But this change in focus on Salmonella should be a warning. Considering that Salmonella causes orders of magnitude more foodborne illness than Listeria, even if not all Salmonella isolates are sequenced and traced back to their source, those that are will represent a dramatic increase in the numbers of new Salmonella cases discovered and reported.
And this considers only the samples collected by CDC through epidemiological investigations and does not include any other sequences added through sampling efforts by the U.S. Department of Agriculture Food Safety and Inspection Service or FDA. The European Centre for Disease Prevention and Control (ECDC) also cooperates with the PulseNet program through its Food- and Waterborne Diseases and Zoonoses Network program, and reports from ECDC have shown that most European countries are currently using WGS and all but one plan to use WGS by the end of 2019.
Expect Dramatic Increase in Outbreaks, Recalls
I had an opportunity to discuss this issue with Dr. Jeff Kornacki of Kornacki Microbiology Solutions and a member of the Food Safety Magazine Editorial Advisory Board. Jeff has previously written about this issue in Food Safety Magazine and predicted exactly what we are now seeing.[1,2]
With the change in the PulseNet database, Jeff projects that we will certainly see more outbreaks identified and a resultant increase in recalls. This has already been the case with more numerous outbreaks related to Listeria being uncovered and reported, with a roughly fivefold increase in listeriosis outbreaks since 2015. He states, “This year, as more Salmonella isolates are added to the database, and considering that Salmonella infections are far more prevalent than Listeria (~1,000 times more Salmonella cases in the U.S.), the number of previously undiscovered Salmonella outbreaks can be expected to increase dramatically.” Jeff also asks, “If the impact of the PulseNet database on Salmonella cases is similar to what it has been with Listeria, can you imagine what a fivefold increase in the roughly 50,000 clinical cases of Salmonella would look like?”
The most likely result will be many more highly publicized outbreaks and far more recalls with consumers facing more and more dramatic headlines (recalls declared “largest,” “longest running,” “most states,” etc.). Yet, even as most of these outbreaks affect fewer people per outbreak, this will not be apparent to consumers—only the frequency of the outbreak headlines. This will dramatically impact consumers’ perception of the safety of their food supply and create a market issue for food processors that they may anticipate, but there may be other ancillary issues that they may not.
“Consumers have seen the news on outbreaks and recalls, and it seems as if there has not been a food group that hasn’t been affected,” says Shawn Stevens of Food Industry Counsel LLC, a law firm that exclusively represents the food industry. “These are not only your customers, but, as an attorney, I recognize that they are also your potential jury in any trial. They have always understood that they must cook their food, but now they are being told that their food is dangerous. This will impact your legal risks.”
A New Testing Environment
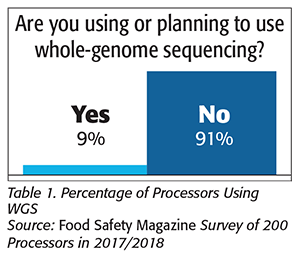 The shift to WGS may also serve to catch processors technically unprepared to respond to this new environment. Over the past few years of surveys for our Food Safety Insights column, we have asked processors about their current and anticipated use of WGS. In those surveys conducted in 2017 and 2018, we asked nearly 200 unique processors whether they have used WGS; 91 percent (Table 1) said that they have not used, nor have considered using, WGS for any of their samples. With this year’s coming changes, this potentially represents a significant “data gap” between processors and regulators.
The shift to WGS may also serve to catch processors technically unprepared to respond to this new environment. Over the past few years of surveys for our Food Safety Insights column, we have asked processors about their current and anticipated use of WGS. In those surveys conducted in 2017 and 2018, we asked nearly 200 unique processors whether they have used WGS; 91 percent (Table 1) said that they have not used, nor have considered using, WGS for any of their samples. With this year’s coming changes, this potentially represents a significant “data gap” between processors and regulators.
When we ask processors why they are not using WGS, the most common answers we hear touch on issues such as regulatory concerns, long turnaround time, limited operational applicability, and high cost. All of these are perhaps legitimate concerns currently, but with the coming changes in the management and use of the PulseNet database, the drivers for the use of WGS may quickly change.
The oft-heard excuse related to company approaches to food safety systems of ‘This is the way that we have always done it and we’ve never had a problem’ will no longer work. “You may have previously thought that no one was getting sick from your products, but if this has been happening, they will discover that now,” adds Jeff. “You may have been going along with a less sensitive environmental surveillance program, thinking that it is OK, but that will no longer be the case. We are going to have to better learn how to clean a plant.”
Shawn agrees with this recommendation. “Processors are going to have to increase surveillance: not only in their own plants but also at the farm level, to ingredients, to every step in their supply chain. You’ll never get the control you are going to need if you keep bringing in pathogens into your plant from the farm or in your ingredients.”
Jeff also makes the point that while all sectors of food processing will be affected by this change, some sectors may be at more risk than others. “The difficulty of tracing back incidents in the past may have been because the source of their foodborne illness was one that a consumer was unlikely to be able to identify, such as a spice or ingredient. Yet, it is a rare occurrence that I look in a plant producing low-water-activity foods and not find Salmonella. These infections have probably always been occurring undetected in foods such as nuts, spices, dry milk, chocolate, and other similar products, but they will increasingly be found.”
So, the response to this situation requires the industry to be proactive. The current system of testing, certification, and auditing is good and has provided processors with the foundation to produce safe food, but these alone will quickly no longer be adequate. Continuing a “business as usual” stance knowing that these outbreaks have probably been occurring—but have been unseen—will be a mistake. “Processors will have to develop a more rigorous environmental monitoring program (EMP), and specifically a ‘Pathogen EMP,’ which allows them to get into the plant and better understand the microbial environment and eradicate the source of pathogen contamination,” Jeff adds. The program should include “…a system to look at positive environmental samples as an ‘investigation’ and not just a routine vectoring program and follow-up process. Processors need to ask why the positive was found and not just follow the routine response steps. Processors should include ‘robust Zone 1 and Zone 2’ indicator testing for pathogen ecology as well as robust end-product testing.” Jeff also believes that the days of only accepting certificates of analyses from suppliers should also be over with more acceptance testing, perhaps based upon appropriate indicator organisms like HTEB and HQA for Salmonella and Listeria, respectively, and better use of validated microbiological control treatments for ingredients.
This will also require a change in the way food safety professionals communicate the issues internally.
Shawn mentions that he is busier these days speaking to corporate leadership to explain the coming changes that will undoubtedly impact their businesses. “We as food safety professionals have understood that pathogens are like gravity—they’re everywhere and if we test for them, we are going to find them. And we have been communicating that message to corporate leadership as we find ‘routine’ positive samples and follow ‘routine’ responses. But you’ll have to change your message. If you are seeing positives in your plant, even if they are only in areas like drains, they can get into your product. This has likely been happening intermittently for many processors, but because of WGS, we are only now able to see it. The situation is like turning on a light in a dark basement—you may not have been able to see the problems before, but now they’ll be clearly revealed.”
Shawn continues, saying, “First, food processors are going to have to make a much greater effort than they have in the past to get pathogens out of their plant. Second, food safety staff are also going to have to get corporate leadership to understand this, especially if they have been told in the past that positive samples are ‘routine,’ and third, companies will need to act with much more urgency than they have in the past.”
Conclusion
Diligence in food safety has always been a moving target. Food safety professionals recognize that continuous improvement is essential, and until foodborne illnesses no longer occur, there is always more that needs to be done. With the increased use of WGS, which will one day be recognized as a pivotal moment in food safety surveillance, that pace of change just got a lot faster.
Bob Ferguson is president of Strategic Consulting Inc. and can be reached at insights@food-safety.com or on Twitter at @SCI_Ferguson.
References
1. www.food-safety.com/magazine-archive1/december-2016january-2017/the-coming-storm-in-the-spice-industry/.
2. www.food-safety.com/magazine-archive1/februarymarch-2017/the-coming-storm-in-the-spice-industry-part-ii-what-the-industry-can-do/.




