Microorganisms are both our friends and our foes in that they can cause deadly illnesses, but on the other hand, life would not be possible without them. With our ever-increasing understanding of microorganisms, we have learned in some instances to harness their beneficial properties to combat pathogens that can be detrimental to human health. Many people today are familiar with the term “probiotic,” especially as it pertains to beneficial microorganisms in yogurt and some dietary supplements. The term “probiotic” is commonly used to describe the use of beneficial bacteria that exhibit human health benefits. The concept of a direct-fed microbial (DFM) is very similar to that of a probiotic and the terms are often used interchangeably. Probiotics have been defined by Fuller[1] as a “live microbial feed supplement which beneficially affects the host animal by improving its intestinal microbial balance.” The U.S. Food and Drug Administration (FDA) uses the term “DFM” to define these products for use in animals.[2] FDA works with the Association of American Feed Control Officials (AAFCO) to regulate animal feed ingredients, including DFM. The AAFCO statement for defining DFM in animal feeds is “Contains a source of live (viable) naturally occurring microorganisms.”[2] Over the last several decades, research into the beneficial effects of DFM for companion animals and livestock has increased much like the information for probiotic benefits for humans. DFM studies have demonstrated improved feed efficiency, milk yields and overall growth and performance in livestock animals. Many DFM studies have also shown a decrease in pathogens carried on the hides and in the feces of livestock. This article will focus on DFMs as food safety interventions for cattle production.
Pathogenic E. coli and Salmonella Presence in Cattle
It is well known that beef cattle harbor foodborne pathogens, such as Salmonella and Shiga-toxin producing Escherichia coli (STEC), in their digestive tracts.[3–6] Studies have reported a significant correlation between fecal and hide contamination with E. coli O157:H7 and subsequent carcass contamination. Results from a study conducted by Barkocy-Gallagher et al.[5] indicated an overall seasonal shedding variation for E. coli O157:H7, non-O157 serotypes and Salmonella in the commercial beef processing plants used in their study. The pathogen prevalence levels were found to be higher on the hides than in the feces. There are many postharvest interventions in place such as organic acid washes and steam pasteurization to reduce pathogen contamination due to fecal material and hides. Nevertheless, it is important to have effective and complementary preharvest interventions to provide added public health protection from farm to fork.
Preharvest Interventions and Food Safety
A wide variety of DFM microorganisms are generally recognized as safe—or GRAS—for use in cattle. In the farm-to-fork dynamic, preharvest interventions such as the use of DFM can improve food safety and reduce public health risks. In addition to this, postharvest interventions may potentially be more effective if the incoming load of foodborne pathogens is decreased before the cattle arrive at processing plants.
A large number of studies have investigated the effect of DFM supplementation on growth and performance and pathogen carriage in beef cattle. As a preharvest food safety application, DFM products may be effective due to several modes of action. The mechanisms are strain dependent and may include the production of antimicrobial compounds such as hydrogen peroxide, organic acids and bacteriocins, competition for colonization sites and immune system stimulation. Rigorous strain selection and in vitro and in vivo testing is needed to develop DFM products that produce the desired results in an animal model.
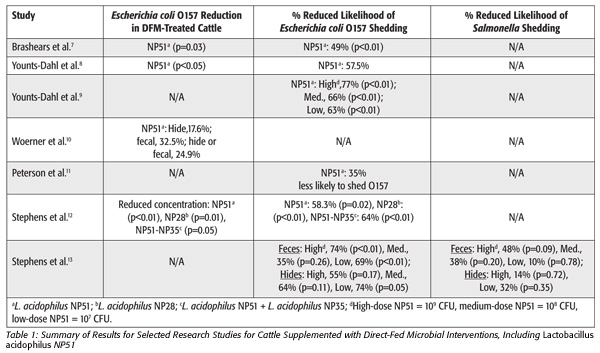 An example of a well-characterized DFM strain that reduces foodborne pathogens in live cattle is Lactobacillus acidophilus NP51, which has been commercially available for many years. This strain has been carefully selected and screened out of a pool of many isolates based on several characteristics in the laboratory and in live animals before a product was developed. More than 12 years of research support the food safety benefits of this DFM. Some of these research results are summarized below and are outlined in Table 1.[7–13]
An example of a well-characterized DFM strain that reduces foodborne pathogens in live cattle is Lactobacillus acidophilus NP51, which has been commercially available for many years. This strain has been carefully selected and screened out of a pool of many isolates based on several characteristics in the laboratory and in live animals before a product was developed. More than 12 years of research support the food safety benefits of this DFM. Some of these research results are summarized below and are outlined in Table 1.[7–13]
Results of Bacterial Interventions
In the initial in vivo studies, Brashears et al.[7] blocked cattle by weight and randomly assigned them to a control group, a treatment group receiving 109/head/day of L. acidophilus NP51 (also known as NPC 747) or a treatment group receiving 109/head/day of L. crispatus NPC 750. The cattle receiving NP51 had significantly less fecal shedding of E. coli O157:H7. The control group was almost twice as likely to have a fecal sample positive for E. coli O157:H7 when compared with the NP51 group. Live weight-gain efficiency was not significantly different among the treatments. There was a trend for carcass-based gain and feed ratios to be improved for animals receiving either one of the DFM treatments. This study highlights the importance of rigorously testing specific strains for the desired activity. While both DFM treatments showed some improvement in carcass-based performance, only NP51 (NPC 747) showed significant reductions in E. coli O157:H7 as a preharvest intervention.
The results of a study by Peterson et al.11 conducted over 2 years also showed reduced E. coli O157:H7 shedding in cattle supplemented with a high dose of NP51. When compared with the control group, the treatment group receiving NP51 was 35 percent less likely to shed E. coli O157:H7. However, the researchers did not find any significant differences between the control and NP51 cattle concerning feed intake and efficiency. There was a trend for higher yield grades for cattle supplemented with NP51.
Varied dose levels and combinations of DFM strains were studied by Younts-Dahl et al.[9] to determine the supplementation needed for maximum efficacy for reduction of E. coli O157:H7. The study results indicated that the high dose of 109 CFU/head/day of NP51 was the most effective at reducing Escherichia coli O157 in fecal and hide samples. In a similar study by Stephens et al.,[12] cattle were randomly allocated to five treatment groups. These included a control group and cattle supplemented with 109 CFU/head/day of strain NP28, 109 CFU/head/day of strain NP51, 109 CFU/head/day of strain NP35 and 108 CFU/head/day of both NP51 and NP35. This study looked at the prevalence as well as the enumeration of E. coli O157 in each treatment group. The prevalence of E. coli O157 was 26.3, 13.0, 11.0, 22.0 and 11.0 percent for the control group and the groups receiving NP51, NP28, NP35 and NP51-NP35, respectively. Statistically lower concentrations of E. coli O157 were observed in the treatment groups receiving NP51, NP28 and NP51-NP35. This study further illustrates the strain-dependent differences in results. On an E. coli O157:H7 concentration basis, the treatment groups receiving NP51, NP28 and NP51-NP35 had significantly lower concentrations when compared with controls.
In an additional study by Stephens et al.,[13] steers were allocated to one of the following treatments: 1) control; 2) low dose: 1 × 107 CFU NP51 + 1 × 109 CFU Propionibacterium freudenreichii NP24; 3) medium dose: 5 × 108 CFU NP51 + 5 × 109 CFU NP24; and 4) high dose: 1 × 109 CFU NP51 + 1 × 109 CFU NP24. Feces and hide swabs were collected from each animal after slaughter and analyzed for E. coli O157 and Salmonella. When compared with controls, the high-, medium- and low-dose groups were 74 percent (p<0.01), 35 percent (p=0.26) and 69 percent (p<0.01) less likely to shed E. coli O157 in feces. For the hide results, E. coli O157 was 55 percent (p=0.17), 64 percent (p=0.11) and 74 percent (p=0.05) less likely to be isolated from the high-, medium- and low-dose treatment groups, respectively. In fecal samples, Salmonella was less likely to be shed by 48 percent (p=0.09), 38 percent (p=0.20) and 10 percent (p=0.78) for the high-, medium- and low-dose groups, respectively. For the high- and low-dose groups, Salmonella was less likely to be isolated from hides by 14 percent (p=0.72) and 32 percent (p=0.35), respectively. However, for the medium-dose group, Salmonella was 110 percent (p=0.83) more likely to be isolated on hides when compared with controls.
Salmonella Presence in Cattle Lymph Nodes
In addition to E. coli O157:H7, Salmonella is also a concern. Several publications have documented Salmonella internalized and isolated from lymph nodes in cattle.[14, 15] Lymph nodes in the chuck and flank regions of a beef carcass often are incorporated into the trimmings used to produce ground beef. If these lymph nodes are contaminated with Salmonella, they could potentially increase the amount of ground beef contaminated with Salmonella. Arthur et al.16 detected Salmonella in the flank lymph nodes of 3.86 percent of cull cattle and 1.05 percent of fed cattle samples. Current postharvest interventions such as organic acid washes and steam pasteurization cabinets will treat only the surface of the carcass and will not reduce internal lymph node contamination with Salmonella.
In recent research, the use of the DFM NP51 was investigated as a potential way to reduce Salmonella harbored within cattle lymph nodes. In two separate studies, very favorable results were observed for decreasing Salmonella in the lymph nodes of cattle at slaughter. This research will soon be published and will be an important resource for the food industry to combat Salmonella contamination of ground beef.
Conclusion
In today’s world, food safety has gained more and more recognition due to tragic foodborne illness outbreaks. The food industry as a whole has implemented many innovative food safety interventions to protect public health. The use of DFM as a preharvest food safety intervention is one of those innovative tools to aid in the reduction of beef product contamination and to thereby decrease foodborne illness.
Mindy M. Brashears, Ph.D., is a professor of food safety and public health and the director of the International Center for Food Industry Excellence at Texas Tech University.
Lacey M. Guillen, Ph.D., is a postdoctoral research associate in the animal and food sciences department at Texas Tech University.
References
1. Fuller, R. 1989. Probiotics in man and animals. J Appl Bacteriol 66:365–378.
2. www.fda.gov/iceci/compliancemanuals/compliancepolicyguidancemanual/ucm074707.htm.
3. Van Donkersgoed, J., T. Graham and V. Gannon. 1999. The prevalence of verotoxins, Escherichia coli O157:H7, and Salmonella in the feces and rumen of cattle at processing. Can Vet J 40:332–338.
4. Barham, A.R., et al. 2002. Effects of the transportation of beef cattle from the feedyard to the packing plant on prevalence levels of Escherichia coli O157 and Salmonella spp. J Food Prot 65:280–283.
5. Barkocy-Gallagher, G.A., et al. 2003. Seasonal prevalence of shiga-toxin-producing Escherichia coli, including O157:H7 and non-O157 serotypes, and Salmonella in commercial beef processing plants. J Food Prot 66:1978–1986.
6. Fluckey, W.M., et al. 2007. Escherichia coli isolates from cattle feces, hides, and carcasses. J Food Prot 70:551–556.
7. Brashears, M.M., et al. 2003. Prevalence of Escherichia coli O157:H7 and performance by beef feedlot cattle given Lactobacillus direct-fed microbials. J Food Prot 66:748–754.
8. Younts-Dahl, S.M., et al. 2004. Dietary supplementation with Lactobacillus- and Propionibacterium-based direct-fed microbials and prevalence of Escherichia coli O157 in beef feedlot cattle and on hides at harvest. J Food Prot 67:889–893.
9. Younts-Dahl, S.M., et al. 2005. Reduction of Escherichia coli O157 in finishing beef cattle by various doses of Lactobacillus acidophilus in direct-fed microbials. J Food Prot 68:6–10.
10. Woerner, D.R., et al. 2006. Preharvest processes for microbial control in cattle. Food Prot Trends 26:393–400.
11. Peterson, R.E., et al. 2007. Effect of Lactobacillus acidophilus strain NP51 on Escherichia coli O157:H7 fecal shedding and finishing performance in beef feedlot cattle. J Food Prot 70:287–291.
12. Stephens, T.P., et al. 2007. Prevalence and enumeration of Escherichia coli O157 in steers receiving various strains of Lactobacillus-based direct-fed microbials. J Food Prot 70:1252–1255.
13. Stephens, T.P., et al. 2007. Reduction of Escherichia coli O157 and Salmonella in feces and on hides of feedlot cattle using various doses of a direct-fed microbial. J Food Prot 70:2386–2391.
14. Paulin, S.M., et al. 2002. Analysis of Salmonella enterica serotype-host specificity in calves: Avirulence of S. enterica serotype gallinarum correlates with bacterial dissemination from mesenteric lymph nodes and persistence in vivo. Infection and Immunity 70:6788–6797.
15. Costa, L.F., et al. 2012. Salmonellosis in cattle: Advantages of being an experimental model. Res Vet Sci 93:1–6.
16. Arthur, T.M., et al. 2008. Prevalence and characterization of Salmonella in bovine lymph nodes potentially destined for use in ground beef. J Food Prot 71:1685–1688.
Direct-Fed Microbials as Preharvest Interventions in Cattle Production



