Chemical contaminants may occur in our food from various sources. They typically pose a health concern, resulting in strict regulations of their levels by national governments and internationally by the Codex Alimentarius Commission. Therefore, analysis of relevant chemical contaminants is an essential part of food safety testing programs to ensure consumer safety and compliance with regulatory limits. Modern analytical techniques can determine known chemical contaminants in complex food matrices at very low concentration levels. Moreover, they can also help discover and identify new or unexpected chemical contaminants.
Sources of Chemical Contaminants in Food
Chemical contaminants can be present in foods mainly as a result of the use of agrochemicals, such as residues of pesticides and veterinary drugs, contamination from environmental sources (water, air or soil pollution), cross-contamination or formation during food processing, migration from food packaging materials, presence or contamination by natural toxins or use of unapproved food additives and adulterants.
Pesticide Residues
The use of pesticides, such as insecticides, fungicides or herbicides, has become an integral part of modern agriculture to increase crop yields and quality by controlling various pests, diseases and weeds. Registration of new pesticides is a strictly regulated process that evaluates their toxicity and environmental fate, and sets maximum residue limits (tolerances) in raw and processed commodities. There are over 1,400 known pesticides. Some of them should no longer be used but may still be present in the environment. Older pesticides are being reevaluated based on currently available scientific data.
Approved uses of pesticides following Good Agricultural Practices should result in pesticide residues below maximum residue limits established in a given country. However, global sourcing of raw commodities and global distribution of food products complicate the situation because pesticide registrations, uses and limits can be and are different in different countries. Consequently, an approved use in one country may result in an illegal pesticide residue in a food imported into another country, such as the recent case of the fungicide carbendazim in orange juice imported into the United States from Brazil. Furthermore, pesticides can be misused or present in food due to contamination during application (spray drift), storage or transportation or from environmental sources, such as contaminated water or soil.
Veterinary Drug Residues
Similar to pesticides, veterinary drugs are agrochemicals that undergo a thorough registration process, resulting in setting of their maximum residue limits/tolerances in animal-derived foods. The major classes of veterinary drugs include antibiotics, anthelmintics, coccidiostats, nonsteroidal anti-inflammatory drugs, sedatives, corticosteroids, beta-agonists and anabolic hormones. These drugs, which are administered to live animals, can remain as residues in animal tissues. Liver and kidney are highly susceptible to residues given their biological function.
Certain antibiotics, such as penicillin, can cause severe allergic reactions in sensitive individuals, which is an important reason for enforcing their residue limits in foods of animal origin. Another important justification for limiting antibiotic usage in food-producing animals is to reduce the risk of pathogenic microorganisms becoming resistant to antibiotics. Most veterinary drugs are not of acute toxicological concern, but some substances, such as nitrofurans, chloramphenicol, clenbuterol and diethylstilbestrol, have been banned in most countries due to their carcinogenicity. Concern about endocrine-disrupting effects has become another reason for regulation of certain veterinary drugs, such as beta-agonists and hormones.
Environmental Contaminants
Environmental contaminants can be man-made or naturally occurring substances present in air, water or soil. They can enter the food chain and even bioaccumulate. Some can pose an acute health risk if present at higher concentrations, but the major concern related to the presence of environmental contaminants in foods is their potential endocrine disruption, developmental, carcinogenic and other chronic effects.
Examples of environmental contaminants that enter the food chain include heavy metals, polychlorinated biphenyls (PCBs), “dioxins” (polychlorinated dibenzodioxins and dibenzofurans), persistent chlorinated pesticides (e.g., DDT, aldrin, dieldrin, heptachlor, mirex, chlordane), brominated flame retardants (mainly polybrominated diphenyl ethers), polyfluorinated compounds, polycyclic aromatic hydrocarbons (PAHs), perchlorate, pharmaceutical and personal care products or haloacetic acids and other water disinfection byproducts.
The manufacture and use of PCBs and other persistent organic pollutants (POPs) have been banned for years, but they remain in the environment due to their high stability. PAHs can be found in the environment as a result of industrial pollution or can originate from oil spills; thus, they were of concern in seafood after the oil spill accident in the Gulf of Mexico in 2010.
Food Processing Contaminants
Certain toxic or undesirable compounds can be formed in foods during their processing, such as during heating, baking, roasting, grilling, canning, hydrolysis or fermentation. Precursors of these contaminants can occur naturally in the food matrix, such as in the case of acrylamide being formed during the Maillard reaction between the amino acid asparagine and a reducing sugar (especially in potato- and cereal-based, heat-treated products). Alternatively, certain processing contaminants, such as nitrosamines, can be formed by interaction of natural food components with food additives. Carcinogenic and genotoxic chlorpropanols, such as 3-monochloropropane-1,2 diol (3-MCPD), are formed during the acid hydrolysis of wheat, soya and other vegetable protein products.
Examples of other processing contaminants include PAHs (in grilled and smoked products), ethyl carbamate (in yeast-fermented alcoholic beverages and other products) or furan (in a variety of heat-treated foods, especially coffee and canned/jarred food).
Food processing may also be a source of cross-contamination, such as contamination of nonallergenic foods with known food allergens.
Migrants from Packaging Materials
Direct contact of foods with packaging materials can result in chemical contamination caused by migration of certain substances into foods. Examples of migrants of health concern may include bisphenol A or phthalates from plastic materials, 4-methylbenzophenone and 2-isopropylthioxanthone from inks, mineral oil from recycled fibers or semicarbazide from a foaming agent in the plastic gaskets that are used to seal metal lids to glass packaging.
Toxins
Toxins are naturally occurring substances that are produced by various organisms, with mycotoxins and marine biotoxins typically representing the major concerns in foods. Other examples of toxins in foods may include bacterial toxins (e.g., staphylococcal toxins) or certain plant toxins, such as pyrrolizidine alkaloids that can be found in honey, milk or eggs. While the bacterial/fungal contamination can be eliminated with heat treatment, the toxins can remain in the food product as contaminants.
Mycotoxins are toxic secondary metabolites produced by fungi (molds) that can colonize various crops. They are of concern mainly in cereals, nuts, infant formula, milk, dried fruit, baby food, coffee, fruit juice and wine. There are many mycotoxins, but only a few are currently regulated, with the European Union having a more comprehensive list than most other countries, which includes aflatoxins, ochratoxin A, patulin, deoxynivalenol, zearalenone, fumonisins and T-2/HT-2 toxins. Different mycotoxins are prevalent in different climates and in various growing and storage conditions.
Marine biotoxins, such as saxitoxin, domoic acid, okadaic acid or ciguatoxin, are highly toxic compounds produced by phytoplankton. During so-called harmful algal bloom events, they can accumulate in fish or shellfish, such as clams, mussels, scallops or oysters, to levels that can pose serious health risks or even be lethal to humans.
Unapproved Food Additives and Adulterants
Food adulteration can happen accidentally when unapproved additives are introduced to the food, or the wrong additive is introduced through formulation error. This results in mislabeled food. Perhaps a larger health issue is when foods are adulterated intentionally for economic reasons to sell a low-value food or material for more or to mask food spoilage. Some adulteration may just mislead or cheat consumers, such as adding high fructose corn syrup to honey, but some may be harmful to them. The most notorious example from recent years is the addition of melamine to whey and other protein concentrates to increase their apparent protein content analyzed as total nitrogen. Other examples include the use of toxic Sudan dyes in adulterated chili powders or adulteration of virgin olive oil with hazelnut oil, which can cause unexpected allergic reactions in sensitive individuals.
Analysis of Known Chemical Contaminants in Food
Most known chemical contaminants in foods are small organic molecules. Except for high-level adulterants, they are typically present in foods at low concentrations (parts per trillion to parts per million); thus, their analyses in complex food matrices are often quite challenging. The basic analytical approach involves an extraction using a suitable solvent, cleanup to remove interfering matrix components, a chromatographic separation and a selective detection.
It is not an exaggeration to say that the implementation of mass spectrometry (MS) as a detection technique has truly revolutionized the analysis of chemical contaminants in foods. As opposed to element-selective or nonselective detectors, MS can detect a wide range of compounds independent of their elemental composition and provide simultaneous quantitation and structural identification of detected analytes. It also adds another degree of separation/selectivity on top of chromatographic separations. These unique features have made MS the number one choice for detection and identification/confirmation of trace-level organic chemical contaminants in modern testing laboratories.
First, the combination of MS with gas chromatography (GC-MS) has become popular for the analysis of volatile and semivolatile compounds, including many pesticide residues, PAHs, PCBs and other less-polar POPs. More polar, thermolabile and less volatile analytes were difficult to analyze until the more recent introduction of atmospheric ionization techniques, such as electrospray, for liquid chromatography-mass spectrometry (LC-MS). LC-MS has opened the door to the direct analysis of many more polar contaminants, including modern, new-generation pesticides, and the majority of veterinary drugs and toxins, such as mycotoxins. Many of the emerging and recently identified contaminants, including acrylamide, melamine or Sudan dyes, are analyzed preferably by LC-MS.
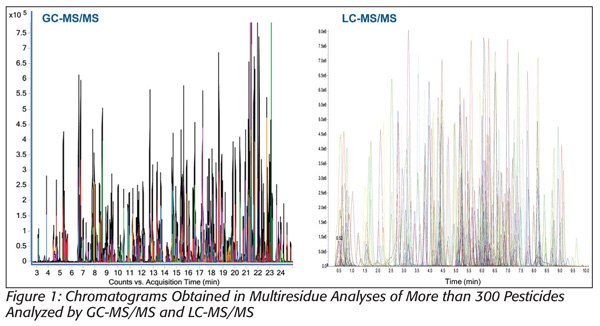 Thus, modern food contaminant testing laboratories utilize both GC-MS and LC-MS to cover the wide polarity range of possible organic chemical contaminants. Tandem MS (MS/MS) is typically employed to provide an increased selectivity (especially in LC-MS) that helps further distinguish target compounds from potential matrix interference. Figure 1 shows an example of chromatograms obtained in a multiresidue analysis of more than 300 pesticides analyzed by GC-MS/MS and LC-MS/MS, demonstrating the speed and selectivity of state-of-the-art instruments that enable the simultaneous and highly sensitive analysis of many compounds.
Thus, modern food contaminant testing laboratories utilize both GC-MS and LC-MS to cover the wide polarity range of possible organic chemical contaminants. Tandem MS (MS/MS) is typically employed to provide an increased selectivity (especially in LC-MS) that helps further distinguish target compounds from potential matrix interference. Figure 1 shows an example of chromatograms obtained in a multiresidue analysis of more than 300 pesticides analyzed by GC-MS/MS and LC-MS/MS, demonstrating the speed and selectivity of state-of-the-art instruments that enable the simultaneous and highly sensitive analysis of many compounds.
Identification of Unknown Chemical Contaminants in Food
Detection and identification of unknown contaminants is not an easy task, especially if they are present at low concentration levels. It requires expertise and a good analytical strategy that is based on all gathered information about the sample and potential sources of contamination. Any clues, such as changes in smell, taste or texture, as well as a description of potential poisoning symptoms may be important in this respect. Concurrent analysis of control (“good”) samples with suspect samples is often essential to find differences and eliminate potential false positives.
If a certain compound or a group of compounds is suspected, then a targeted sample preparation and instrumental method(s) can be employed (see sidebar “The Importance of Moisture in Samples Prior to Chemical Analyses”). For a truly unknown analysis, different extraction and separation approaches should be used to isolate compounds with a wide range of physicochemical properties (polarity, solubility, volatility, etc.). Nontargeted analysis should be performed, such as MS with full-spectra acquisition. Statistical analysis of the acquired chromatographic and MS data of contaminated and noncontaminated samples may help identify differences and reduce the number of components that have to be examined. The acquired MS spectra of suspected contaminants can be compared with MS spectral libraries and compound databases. In LC-MS, high-resolution/accurate-mass measurements, using time-of-flight (TOF) or orbitrap MS instruments, should be used for added selectivity. In addition, tandem MS should be employed to help elucidate the structure of unknown contaminants. In the end, strong knowledge and expertise in both analytical and food chemistry are typically required to succeed in this task.
Current and Future Trends in Chemical Contaminant Analysis in Food
The current and future trends in the analysis of chemical contaminants are and will be strongly affected by developments in analytical instrumentation. The speed, sensitivity and selectivity of state-of-the-art MS instruments enable analysis of many compounds in one analytical run. Consequently, streamlined sample preparation approaches, such as QuEChERS (Quick, Easy, Cheap, Effective, Rugged and Safe), can be used that require minimum extract cleanup without any preconcentration steps—thus, they can be miniaturized and automated.
The advancements in high-resolution/accurate MS instruments and development of related software tools show great potential to bring this technology from the research environment into testing laboratories, where it could be employed for nontargeted testing of known and unknown chemical contaminants.
 Katerina Mastovska, Ph.D., is a lead staff scientist at Covance Laboratories. She is actively involved in the AOAC International as a co-chair of the AOAC Chemical Contaminants and Residues in Food community.
Katerina Mastovska, Ph.D., is a lead staff scientist at Covance Laboratories. She is actively involved in the AOAC International as a co-chair of the AOAC Chemical Contaminants and Residues in Food community.
Sidebar
The Importance of Moisture in Samples Prior to Chemical Analyses
Moisture content is an important consideration during sampling procedures, in part because it affects the extent of sample heterogeneity. It may be necessary to determine the moisture content through sample drying to express analytical results on a uniform scale.
Precautions must be considered when drying foods at elevated temperatures, since chemical reactions such as hydrolysis can occur and these reactions can be accelerated. Moisture determinations can be erroneous if hydrolysis has occurred, since the water of hydrolysis has not been released from the sample. A general rule of thumb for sample drying is that it should be as rapid and at as low a temperature as possible. Vacuum methods that can used to dry a sample include vacuum ovens and lyophilization, or freeze-drying. Another method is microwave drying. Unlike external heating devices that operate through the sample vessel, microwaves rapidly heat the sample, keeping temperature gradients to a minimum.
For certain chemical testing applications, such as the presence of metals in food as well as the extraction of crude fat and nutrients from food, microwave digestion is the preparation method of choice. The ability of microwave sample prep to dissolve almost any matrix, leaving target species behind, provides preparative capabilities unavailable through other methods.
The inherent variability in the composition of raw materials, basic ingredients and processed foods requires the use of proper sampling and sample pretreatment techniques, in addition to statistical methods for obtaining representative and replicate samples. Using the proper sample preparation methodology can reduce analytical error and costly detection mistakes that could jeopardize the safety of the food produced as well as lead to an even more costly food safety-related recall.
Modern Analysis of Chemical Contaminants in Food