The way processors view their responsibilities regarding microbiological testing—particularly pathogens—is changing. Regulatory pressure and a focus on food safety are causing changes in where processors do their testing.
As we’ve mentioned before, there is a clear focus on pathogenic harborage organisms and getting them out of the plant. This is also causing more processors to get pathogens out of the plant laboratory by outsourcing more of this type of testing to commercial food laboratories. This trend is having a dramatic impact on testing markets and will affect the businesses of processors, food commercial laboratories and diagnostic device manufacturers.
To find out more, we conducted a survey of more than 100 food processors in the U.S., Canada and Mexico to learn how they may be changing their testing programs. The survey respondents were evenly represented by companies in the protein, fruit/vegetable, processed foods, dairy and spices/ingredients sectors, at processing companies with from 50 to 5,000 employees and test volumes of a few samples to more than 1,000 samples per week.
We also conducted interviews with senior executives at several leading commercial laboratories to get their responses to what we found and how that may be impacting their businesses as well.
Survey Results
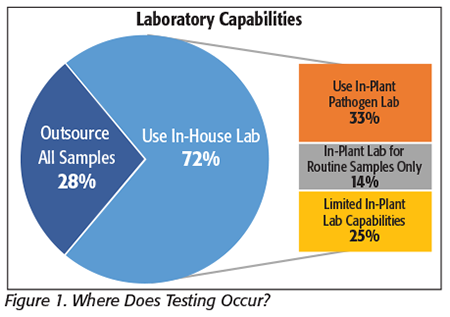 About three-quarters (72%) of the processors in the survey reported that they have an in-plant microbiology laboratory (Figure 1). Thirty-three percent of the respondents reported that their lab is equipped to handle any of the testing that they need, and they typically send out only confirmatory samples; 14 percent report that their lab is equipped for routine (nonpathogen) analysis only and all pathogen tests are sent out; and 25 percent report that they have a lab but with limited capabilities. (We learned from our interviews that this typically means they are equipped for one or two test formats and some screening tests—but their limited capabilities mean that they have to outsource at least some of their pathogen tests and portions of their routine micro samples as well.)
About three-quarters (72%) of the processors in the survey reported that they have an in-plant microbiology laboratory (Figure 1). Thirty-three percent of the respondents reported that their lab is equipped to handle any of the testing that they need, and they typically send out only confirmatory samples; 14 percent report that their lab is equipped for routine (nonpathogen) analysis only and all pathogen tests are sent out; and 25 percent report that they have a lab but with limited capabilities. (We learned from our interviews that this typically means they are equipped for one or two test formats and some screening tests—but their limited capabilities mean that they have to outsource at least some of their pathogen tests and portions of their routine micro samples as well.)
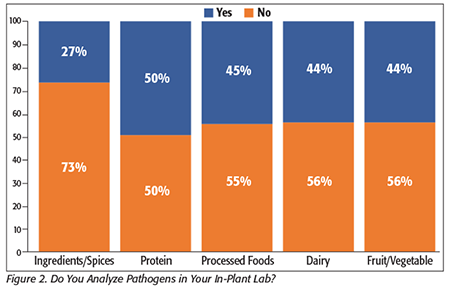 We found that this distribution is representative across different types of processors; Figure 2 shows the distribution of in-house pathogen analysis versus outsourcing across the five processor types in this survey. Consistent with our findings in previous surveys, the one processor type that shows a higher level of outsourcing is ingredients and spices, and in our interviews with microbiologists at these companies, they often report that the technical difficulties in testing dry products and the complexities that accompany testing so many different ingredient and spice matrices make outsourcing to a better-equipped commercial laboratory easy to justify.
We found that this distribution is representative across different types of processors; Figure 2 shows the distribution of in-house pathogen analysis versus outsourcing across the five processor types in this survey. Consistent with our findings in previous surveys, the one processor type that shows a higher level of outsourcing is ingredients and spices, and in our interviews with microbiologists at these companies, they often report that the technical difficulties in testing dry products and the complexities that accompany testing so many different ingredient and spice matrices make outsourcing to a better-equipped commercial laboratory easy to justify.
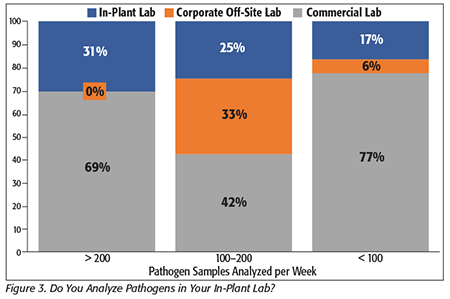 But while outsourcing may be uniform across processor types, there is more diversity across processors by size and numbers of samples collected. As can be seen in Figure 3, processors that analyze more samples tend to use their in-house laboratory more than those with fewer samples; companies with fewer than 100 samples per week used an in-house pathogen lab for 17 percent of total sample volume, whereas those that analyzed more than 200 samples per week used an in-house lab for 31 percent of samples.
But while outsourcing may be uniform across processor types, there is more diversity across processors by size and numbers of samples collected. As can be seen in Figure 3, processors that analyze more samples tend to use their in-house laboratory more than those with fewer samples; companies with fewer than 100 samples per week used an in-house pathogen lab for 17 percent of total sample volume, whereas those that analyzed more than 200 samples per week used an in-house lab for 31 percent of samples.
What the Experts Are Saying
This shift is being noticed in the laboratory business as well. In a conversation with Cabot Earle, CEO of Microbac Laboratories, he mentions, “It is clear that FSMA [the Food Safety Modernization Act] is driving better awareness of the importance of accurate sampling and analysis. This is especially clear in smaller and midsized companies as they become more aware of risks to their brand. We are seeing more environmental monitoring, especially with a focus on Listeria, as a proactive approach to better food safety.”
Some of these changes, of course, are driven by economics—it is difficult to justify the capital expense for a laboratory to analyze a small number of samples. Companies that have a higher number of samples have not only a better economic justification for lab capital investments but also more resources to maintain qualified lab staffing and accreditations. In previous surveys, we reported that approximately two-thirds of processors by location will primarily outsource their pathogen analysis and that one-third will use their in-house lab. However, because the larger processors are the ones with more samples, the total volume of outsourced pathogen samples (rather than processor locations) is 50 percent of the total number of samples analyzed. In this survey, an expected 60 percent of the processing locations reported sending their pathogen samples out to a commercial lab, but when measuring the volume of samples outsourced, 75 percent of the samples in this survey were sent out.
We spoke with Kurt Westmorland, senior vice president at Mérieux NutriSciences, who adds, “Demand for outsourcing testing is clearly growing. Larger processors have had established testing programs for a longer time, but we see this outsourcing decision being made in smaller and midsize companies as they pay closer attention to their programs. But all of the processors are seeing that lab accreditation requirements—either from FSMA or other audit requirements—are harder to meet and the difficulty of in-house testing is harder to overlook.”
We also asked processors how long they have been outsourcing pathogen samples. Their answers were also very indicative of the ongoing shift, with one in five respondents reporting that they have changed their processes and started outsourcing their pathogen samples only within the past 2–3 years.
Sean Murray, president of U.S. food testing for Eurofins Scientific, says, “We are seeing the same growth of outsourcing, more in some segments than others; we expect that this demand is backloaded, and we expect to continue to see growth as the FSMA compliance dates affect the programs at midsize and smaller processors.” Gary Smith, director of food safety systems for Eurofins, adds, “Looking at the total cost of in-house pathogen analysis—not just cost of the diagnostic but the training, systems, hiring and requirements from third parties such as BRC [British Retail Consortium] or SQF [Safe Quality Food]—outsourcing pathogens [testing] becomes a less expensive proposition.”
As noticed by these lab executives, every year, commercial food labs account for a larger share of the testing market. In this (and other) surveys that Strategic Consulting Inc. has produced in the past few years, the total volume of microbiology tests worldwide is growing annually at approximately 5–6 percent. The study data, however, show that pathogen testing at food contract labs is growing at nearly twice that rate, roughly 10–12 percent annually.
Cost Analysis
With this shift in testing, we also wanted to find out more about commercial lab pricing. This higher demand seems to be affecting laboratory pricing, as 33 percent of the respondents reported that they are paying higher per sample prices than they were in the past 12–18 months, with average increases of between 5 and 10 percent. Two-thirds reported that they are paying “about the same,” and only one of the processors reported paying less, telling us that they were the beneficiary of a bidding war between two local laboratories.
This last comment also illustrates that the commercial lab market is not a national market but very much a regional market. We have previously reported that 75 percent of processors say that for pathogen analysis they use a commercial lab located within 100 miles of their plant. This proximity is one of the key criteria for selecting a lab, as it is linked with faster turnaround time and easier transport of samples to the lab. So, it is within this radius that the most price competition will occur, and in regions where processors have multiple local labs to choose from, they will probably see better pricing. The laboratories know that the basis of competition should not only be location and price but also the overall value that they can offer during this shift.
Conclusions
One final comment on pathogen outsourcing: We conducted not just electronic surveys but also a series of telephone interviews with many of the respondents. We thus asked a lot of questions about outsourcing of pathogens and the reasons why. As expected, many people reported that they were getting pathogens out of the plant for food safety reasons. But we spoke with several who, while they certainly understood the arguments, seemed to be unmoved by the urgency to outsource and conducted all their pathogen tests within their in-house lab. They told us they have had no adverse incidents; we heard comments like, “If it is done correctly …” and “If properly handled…,” pathogens can be analyzed in-plant without risk. One microbiologist said that having in-plant capabilities gave her greater control and faster turnaround times than using an outside lab. Another added, “If you are overly concerned with pathogen enrichment or working with positive controls at an in-plant lab, you have the wrong installation or the wrong people running it.”
We found these comments very surprising until we looked at one additional data point. It turns out that each of these individuals had been food safety microbiologists for more than 25 years. One was in her 35th year.
Here’s to experience.
Bob Ferguson is the managing director of Strategic Consulting Inc. and can be reached at foodsafetyinsights@gmail.com or on Twitter at @SCI_Ferguson.
Outsourcing: Pathogen Testing under the Microscope