With an increasing global population, sustainable food production requires the use of effective pest intervention practices. When natural or biological processes are insufficient, remediation may be achieved by application of pesticide chemicals. Pesticides have been designed to deter, attract, inhibit and exterminate the organisms that jeopardize the successful growth, harvest, storage and subsequent production and distribution of many foods. Commodities benefit from a variety of chemicals that target insects, weeds, fungi and other pests applied pre- and postharvest. From the early use of elemental metals to the current use of thousands of synthetic pesticides, the goal has remained the same: prevent or eliminate unwanted intruders destroying valuable agricultural commodities in fields, mills, manufacturing facilities and eating establishments.
Beyond food and agricultural uses, pesticides facilitate disease prevention and reduction. Consider the proliferation of the West Nile virus, responsible for tens of thousands of reported cases and nearly 1,500 deaths across the United States since the U.S. Centers for Disease Control and Prevention initially reported cases in New York in 1999.[1] Localized treatments such as the use of certain predatory fish to destroy mosquito larvae in water offer relief, but these are indiscriminate and affect other indigenous insect populations. Chemical spray treatments control mosquitoes in nonaquatic environments; however, spray drift can deposit on agricultural crops. Additionally, occupational, public and ecological exposures must also be considered.[2]
Regulations on Pesticide Use
Safety considerations pertaining to the use of chemical pesticides for food production and public health require adherence to proper application practices. In the United States, the Federal Insecticide, Fungicide and Rodenticide Act (FIFRA) regulates the use of approved chemical pesticides. The act “mandates that the EPA [Environmental Protection Agency] regulate the use and sale of pesticides to protect human health and preserve the environment.”[3] To meet the legal requirements, pesticide manufacturers register pesticides with EPA based on a particular pesticide’s mode of action upon the targeted pest pertaining to the specific commodity. Registrants conduct toxicological, environmental fate and ecological impact studies of the pesticide based upon the application rates. Registrants often perform these studies for major crops (soybeans and wheat) but may not register minor-use crops (many fruits and vegetables) because the crop “does not provide sufficient economic incentive to support its registration.”[4] To support growers’ needs for registered pesticides, the federal-state Interregional Research Project Number 4 (IR-4) conducts studies and generates data for regulatory agencies of pesticides on minor-use crops.[5] EPA, upon review of registrant or IR-4 studies, conducts human health and environmental risk assessments to ultimately rule on whether the pesticide will be approved for use. Appropriate residue tolerances are issued for the pesticide based upon the risk assessments conducted by EPA and the application rates directed by the registrants. Pesticides not registered through FIFRA for application on a specific commodity are considered not permitted for use on that crop.
Regulations for approved pesticide use apply to both domestic crops and imported foods. As food commodities destined for the U.S. cross the border, they fall under FIFRA, regardless of country of origin. How does this regulation affect international food producers exporting to the United States? If that food producer regularly exports to the U.S., it may choose to follow the EPA tolerances to meet the FIFRA requirements. However, countries often independently establish their own limits, commonly referred to as maximum residue limits (MRLs). These limits are based on the maximum allowable applied chemical pesticides that result in residues that produce no apparent adverse health or ecological effects. Although investigations, evaluations, assessments and final rulings resemble those performed by EPA with regard to pesticide registration, the resulting residue level allowed can vary by country. Regional geographic and growing conditions, native and invasive pest populations and cultural practices contribute to the selection and application rates of pesticides used.[6] Do multiple residue regulatory limits suggest that the safe use of a pesticide is questionable? Hardly. But the context of federal regulations ultimately dictates the acceptance or refusal of any commodity into a country. Furthermore, the U.S. Food and Drug Administration (FDA) also ruled on July 26 of this year that U.S. food companies receiving food ingredients used in products are responsible for ensuring that no unintentional chemical residues (including pesticide residues) are present on their foods as part of the Food Safety Modernization Act (FSMA).[7] These regulations, in tandem with nonharm-onized MRLs, imply a risk of unlawful pesticide residues not only in the U.S. but also globally, thus complicating the import and export of foods.
Regulatory Monitoring Programs
The fast pace of enterprise in the 21st century presents challenges with regard to the regulatory laws pertaining to pesticide residues. For that reason, FDA tests both domestic and incoming foods for pesticide residues as part of its enforcement activities. Risk-based programs such as the Pesticide Monitoring Program (PMP) allow FDA to monitor domestic and foreign food products primarily consumed by young children. Testing performed by FDA laboratories provides the basis for investigations into so-called violative residues. These investigations have resulted in fines to the U.S. suppliers, producers or importers of the products, even if the residues detected originated from pesticide application that was legal outside the United States.[8]
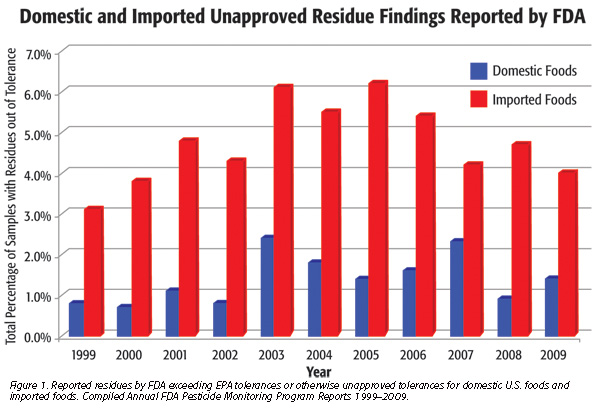 For the 2009 PMP annual report, released in March of this year, FDA found violative residues in 1.4 percent of 1,385 domestic food samples tested, in contrast to finding violations in 4.0 percent of 4,196 foreign food samples. FDA reported similar findings when comparing domestic and imported products spanning the last 10 years (Figure 1). FDA sample selection criteria, rather than being statistically based, “focused towards products and countries of origin that have a history of violations or are suspected of violations based on available intelligence.”
For the 2009 PMP annual report, released in March of this year, FDA found violative residues in 1.4 percent of 1,385 domestic food samples tested, in contrast to finding violations in 4.0 percent of 4,196 foreign food samples. FDA reported similar findings when comparing domestic and imported products spanning the last 10 years (Figure 1). FDA sample selection criteria, rather than being statistically based, “focused towards products and countries of origin that have a history of violations or are suspected of violations based on available intelligence.”
The Pesticide Data Program (PDP), managed by the United States Department of Agriculture (USDA), has reported pesticide residue results statistically representative of those foods consumed most often by children since 1991. The program was designed not for enforcement purposes, but rather to provide data to EPA “as a critical component for dietary assessments of pesticide exposure, a critical step to verify that all sources of exposure to pesticides meet the safety standards set by the 1996 [Food Quality Protection Act].”[9] The PDP, like FDA’s PMP, incorporates a combination of domestic and imported samples, characterizing a young child’s diet. In the 2011 report, 0.27 percent of samples tested exceeded EPA tolerances, with about one-fifth of those domestic in origin. The PDP reported more above-tolerance violations for domestic compared with foreign U.S. samples between 2005 and 2009, with the reverse observed in 2010 and 2011.[10–15] Some violations may have been due partly to different MRLs in the country of origin. However, unintentional use (spray drift or crop rotation) or intentional misuse also may have played a role in violations. Pesticide residue monitoring and surveillance is not unique to the United States. Canada, the European Union (EU), Japan and other developed nations have organized programs to assess and regularly monitor domestic and incoming foods for pesticide residues. This ensures that foods meet necessary regulatory and safety requirements for consumers. Additionally, international agreements such as the Rotterdam Convention were established to “promote shared responsibility and cooperative efforts among Parties in the international trade of certain hazardous chemicals in order to protect human health and the environment from potential harm and to contribute to their environmentally sound use.…”[16] This joint effort applied restrictions to the use of known toxic and hazardous pesticides as listed in Annex III of the document. The list contained 43 chemicals that “… have been banned or severely restricted for health or environmental reasons…,” 32 of which are pesticides.[17] Most of the chemicals on this list are also not approved for use by EPA, as published in the Code of Federal Regulations (40 C.F.R. Part 180).
Sampling Practices for Pesticide Testing
The technique used to collect a sample for pesticide testing is undeniably the most critical step in the residue testing process. Proper sampling ensures the sample submitted fully represents the crop, shipment or production lot. Localized events, such as drift of a pesticide from a neighboring crop, can result in an illegal residue, particularly if the neighboring crop is a different commodity. Statistically based sampling ensures all regions of crop identified for testing are represented and that sample test data reflect the entire crop. Localized sampling will contribute a bias to the result; if the crop is later resampled, a subsequent result can conflict with the original number.
The approach for sampling varies based on the product type and composition. Procedures for acceptable sampling techniques are available through regulatory agencies. Grain sampling can be conducted following the USDA Grain Inspection, Packers and Stockyard Administration plan FGIS-PN-12-09;[18] harvested products or shipments may be sampled according to the FDA Operations Manual, Chapter 4,[19] whereas EU imports may be sampled as described in the EU Directive 2002/63/EC.[20] Companies specializing in sampling protocols can conduct sampling if requested or required.
Following the collection of a sample for testing, expedient steps to ensure the integrity of the sample are important to ensure the generation of meaningful sample data. Numerous pesticides are subject to loss due to volatility or degradation at ambient or elevated temperatures. The preferred approach is to send the sample to the laboratory immediately upon collection. However, if this is not feasible, freezing stabilizes the sample by slowing down the potential loss of pesticide residues.
Selecting a Laboratory and Pesticide Screen
Identifying an appropriate laboratory and residue screen ensures proper risk mitigation for all involved. Pesticide residue screens offered by laboratories often include single or specialty residue screens, along with multi-residue screens. Tests targeting single or specific compound classes often are available based on the chemistry of the pesticide(s) and, in some cases, apply only to a small subset of commodities. These screens deliver highly targeted results—often with a short turnaround—when only a single pesticide residue is in question. For example, a supplier may choose to test for a specific residue to verify its absence on a crop. In January 2012, FDA reported an illegal fungicide, carbendazim, in Brazilian orange juice imported into the United States in 2011. While the fungicide was approved for use on oranges in Brazil and Mexico, carbendazim was not registered with the EPA. FDA targeted carbendazim in imported orange juice at all ports of entry.[21] In response to the demand for fast single-residue testing, some laboratories began offering a single pesticide residue test for carbendazim in oranges and orange juice. Single-residue screens offer useful information about specific pesticides that may or may not have been applied; however, they neglect to deliver additional information regarding other potential residues. Multi-residue screens provide a significant advantage over single-residue screens by detecting and quantitating many classes of insecticides, fungicides, herbicides, growth regulators, repellents and synergists, offering an elevated level of security, particularly when unforeseen factors contribute to unapproved residues in crops. Because they cover a wider range of compounds than single or specialty screens, these screens are the most popular.
Multi-residue screens differ widely in breadth and scope. When selecting a screen, consider the number of pesticide residues included, whether they represent those monitored by regulatory agencies and relevance to the commodity. This can be a daunting task, considering not just the hundreds of residues evaluated in various programs, but also the thousands of different pesticides available for use. Requesting lists from labs can cause more confusion than clarification. Therefore, contacting a laboratory directly and requesting what regulatory programs it uses as a basis for establishing its screen size and scope options can provide insight into not only whether a laboratory offers a comprehensive screen to meet the need, but also as to which screen is most appropriate for the commodities or products requiring testing. For example, Medallion Laboratories offers the Basic Screen of pesticides, metabolites and contaminants monitored in the FDA PMP and USDA PDP. Some pesticides are very stable, but some readily degrade and form metabolites by way of environmental factors or by their chemistry in the analysis. Nonpesticide contaminants in the screen are included to the extent they are monitored as part of FDA’s PMP. Coincidental monitoring of such contaminants is valuable in avoiding manufacturing practices that can introduce them into processed foods. For food products destined for broader, international markets, Medallion’s Expanded Screen includes additional compounds reportedly being monitored by the Federal Bureau of Risk Assessment in Germany, a participating Member State of the European Food Safety Authority.[22] Such a screen provides a level of assurance of international significance and heightened awareness to potential residues originating from pesticides used in the EU or for food products that are destined as EU imports. Medallion also offers the Organic Screen, which includes pesticides listed on the USDA National Organic Program (NOP) Prohibited Pesticide List (NOP 2611-1). In 2013, the NOP required organic certifiers to test a minimum of 5 percent of all certified organic products for pesticide residues as “Residue testing plays an important role in organic certification by providing a means for monitoring compliance with the NOP and by discouraging the mislabeling of agricultural products.”[23] Organic farming practices exclude the use of synthetic chemical pesticides; however, ubiquitous pesticides, particularly those that persist in the soil and those used in nearby nonorganic fields, may drift to an organic crop. Pesticide testing offers the most definitive option to show proper organic crop management.
Selecting a laboratory with proper quality assurance provides added confidence in the reported results. Testing laboratories accredited by ISO standard 17025 participate in regular laboratory performance audits and proficiency testing programs, conduct scheduled ongoing quality control procedures and fully document corrective actions to any deficiencies to ensure full transparency and traceability of results.[24]
Residue Testing Methods
To test for chemical residues, laboratories must first prepare the sample for testing. Homogeneity is a critical step in analytical processes; it ensures that what is detected in the subsampled portion represents the sample submitted. To achieve homogeneous samples, cryo-treating the sample with liquid nitrogen or dry ice prior to grinding helps produce samples of uniform particle size. Cryo-cooling also prevents excessive heating as samples are ground. Finely ground solid or homogeneous liquid samples submitted as finished ingredients, such as flours and juices, require no further treatment prior to analysis.
Screening for residues in foods employs an extraction method to isolate the compounds to be analyzed from the sample. FDA’s Pesticide Analytical Manual (PAM) includes multiple extraction variations, the selection of which depends on the sample and residue. The variations utilize multiple solvents in liquid-liquid extraction processes to transfer the compounds to be analyzed into the solvent of choice for the quantitation procedure. Depending on the variation, additional solvents are used to “wash” the extract, removing unwanted interfering components. Subsequent cleanup procedures further isolate the compounds to be analyzed utilizing various solid-phase extraction techniques such as Florisil, C18 and/or charcoal.[25] The most popular variation in the PAM was developed by Milton A. Luke et al. in 1981.[26] This method simplified the overall procedure by eliminating the labor-intensive Florisil cleanup. Further modifications to the “Luke” method have been developed over the years, many of which focused on various solid-phase extraction techniques.
To test for residues in the extract, gas and liquid chromatography methods separate compounds using either temperature or solvent polarity, respectively. Once the compounds are individually separated, they pass through to a detector that identifies each compound as a peak on a chromatogram. Selective detectors, such as electron capture, flame photometric and fluorescence detectors, can detect specific characteristics in a molecule, such as elements or functional groups. Electron capture detectors, for example, detect regions of high electron density in a molecule, such as halogenated pesticides. Although very sensitive, electron capture detects not only electron-rich organochlorine pesticides but also other sample components in the extract with high electron density.
Mass spectrometry (MS) detection is an attractive alternative to selective detectors for chromatography. MS offers detection and verification of molecules by mass and mass fragmentation following chromatographic separation. Thus, MS adds multiple dimensions of selectivity to the existing chromatographic selectivity. As a molecule passes into the spectrometer, the molecule ionizes and breaks apart into fragments that can be detected. Whereas selective detectors offer a single peak with no additional selectivity, MS discriminates based upon the mass of the compound, its fragments, tracking of fragmentation or combinations thereof. In the last decade, MS has become an affordable technology for laboratories and has become the preferred detection method for pesticide residue analysis.
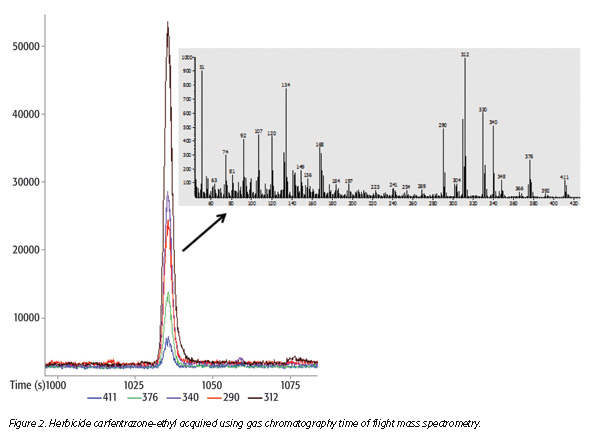 Advances in technologies for MS allow for simpler extraction methods that reduce the overall time and materials required for successful analysis. The Quick, Easy, Cheap, Effective, Rugged and Safe (QuEChERS) method, developed by Anastassiades et al.,[27] applies a simple acetonitrile sample extraction to high-moisture produce followed by the addition of magnesium sulfate and salts that forces residues into the acetonitrile and facilitates the separation of the organic and sample/aqueous layers. An aliquot of the acetonitrile extract is then treated with additional magnesium sulfate and a primary/secondary amine using a dispersive solid-phase extraction technique. The resulting extract is chromatographed using either gas or liquid chromatography, coupled with MS. This method is safer and more cost effective than the Luke method and appeals to many laboratories. The QuEChERS approach has been modified many times to accommodate the wide variety of sample types (Figures 2 and 3). For many laboratories, QuEChERS is either the primary or only technique for pesticide residue testing.
Advances in technologies for MS allow for simpler extraction methods that reduce the overall time and materials required for successful analysis. The Quick, Easy, Cheap, Effective, Rugged and Safe (QuEChERS) method, developed by Anastassiades et al.,[27] applies a simple acetonitrile sample extraction to high-moisture produce followed by the addition of magnesium sulfate and salts that forces residues into the acetonitrile and facilitates the separation of the organic and sample/aqueous layers. An aliquot of the acetonitrile extract is then treated with additional magnesium sulfate and a primary/secondary amine using a dispersive solid-phase extraction technique. The resulting extract is chromatographed using either gas or liquid chromatography, coupled with MS. This method is safer and more cost effective than the Luke method and appeals to many laboratories. The QuEChERS approach has been modified many times to accommodate the wide variety of sample types (Figures 2 and 3). For many laboratories, QuEChERS is either the primary or only technique for pesticide residue testing.
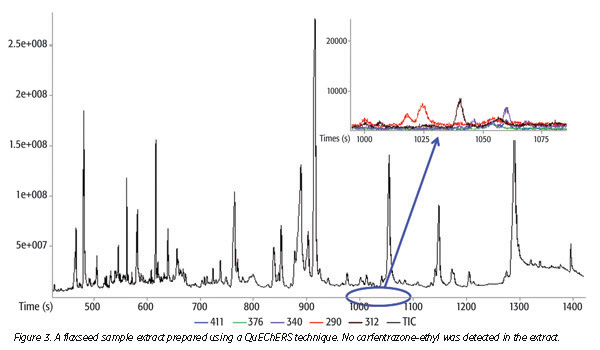
Some residues present challenges in chromatographic analysis, even with the use of QuEChERS extracts and chromatography with MS. As sample extraction procedures are simplified, more opportunities for interferences cause complications in sample analyses. Extracts containing interfering components (the number and nature of which vary greatly, depending upon the type of food tested) “mask” a residue by essentially sharing the same mass as the desired compound, suppress the response of a compound or may not enter the ionization chamber of the instrument due to poor sample introduction. These considerations require carefully developed methods that maximize the detection of as many compounds in as many sample types as possible.
Summary
Regardless of the technique used for analysis, trace-level chemical residue testing requires due diligence in verifying the detection and subsequent quantification of a residue. The initial result obtained from an analysis should be carefully evaluated and replicated with a second analysis, including a spike recovery determination, a step that is particularly important for illegal residues or those exceeding MRLs. Presumptive positive results from the initial analysis may be a direct result of artifacts of the analysis—improper sample preparation, laboratory contamination, misidentification during analysis, etc. Laboratories performing further verification of a presumptive result deliver a more defensible result, very important when that result demands regulatory action.
The regulations and science of pesticide residues permeate every facet of the food industry, from the farm to the consumer. Comprehensive understanding of the regulatory climate, both domestically and internationally, ensures that the proper precautions are taken to mitigate regulatory and safety risks associated with pesticide use. Furthermore, evaluating pesticide testing screens and capabilities offered by laboratories encourages meaningful results that represent ingredients used for food products at home and abroad.
The author would like to express sincere thanks to Jonathan DeVries, Ph.D., for his contributions to this article.
 Kelly Dorweiler, M.Sc., is a senior scientist with General Mills and Medallion Laboratories. She received an M.Sc. in environmental health from the University of Minnesota School of Public Health. She can be reached at kelly.dorweiler@genmills.com.
Kelly Dorweiler, M.Sc., is a senior scientist with General Mills and Medallion Laboratories. She received an M.Sc. in environmental health from the University of Minnesota School of Public Health. She can be reached at kelly.dorweiler@genmills.com.
References
1. www.cdc.gov/westnile/statsMaps/finalMapsData/index.html.
2. wwwnc.cdc.gov/eid/article/7/1/pdfs/70-0017.pdf.
3. www.epa.gov/agriculture/lfra.html#Summary of the Federal Insecticide, Fungicide, and Rodenticide Act.
4. www.epa.gov/pesticides/minoruse/minor_use_rpt.pdf.
5. ir4.rutgers.edu/Other/AnnualReports/2012%20Annual%20Report.pdf.
6. Dharam, P. and A.U. Shankar. 2012. Integrated pest management: Principles and practice. Oxfordshire, UK: CAB International.
7. www.fda.gov/ForConsumers/ConsumerUpdates/ucm362462.htm.
8. www.fda.gov/downloads/Food/FoodborneIllnessContaminants/Pesticides/UCM352872.pdf.
9. www.ams.usda.gov/AMSv1.0/getfile?dDocName=stelprdc5102692.
10. www.ams.usda.gov/AMSv1.0/getfile?dDocName=STELDEV3003674.
11. www.ams.usda.gov/AMSv1.0/getfile?dDocName=STELPRDC5064786.
12. www.ams.usda.gov/AMSv1.0/getfile?dDocName=STELPRDC5074338.
13. www.ams.usda.gov/AMSv1.0/getfile?dDocName=STELPRDC5081750.
14. www.ams.usda.gov/AMSv1.0/getfile?dDocName=STELPRDC5091055.
15. www.ams.usda.gov/AMSv1.0/getfile?dDocName=stelprdc5098550.
16. www.pic.int/TheConvention/Overview/tabid/1044/language/en-US/Default.aspx.
17. www.pic.int/TheConvention/Chemicals/AnnexIIIChemicals/tabid/1132/language/en-US/Default.aspx.
18. www.gipsa.usda.gov/lawsandregs/bulletins/pn12-09.pdf.
19. www.fda.gov/ICECI/Inspections/IOM/ucm123267.htm.
20. www.fsvps.ru/fsvps-docs/ru/usefulinf/files/es2002-63.pdf.
21. www.fda.gov/Food/FoodborneIllnessContaminants/Pesticides/ucm288004.htm.
22. www.bfr.bund.de/de/parameters_for_the_determination_of_pesticide_residues-5832.html.
23. www.ecfr.gov/cgi-bin/text-idx?c=ecfr&sid=ec23ffb6553dbaff8d29d76ffe3b829b&rgn=div8&view=text&node=7:3.1.1.9.32.7.358.27&idno=7.
24. AOAC INTERNATIONAL Analytical Laboratory Accreditation Committee. 2010. Guidelines for laboratories performing microbiological and chemical analyses of food and pharmaceuticals; An aid to interpretation of ISO/IEC 17025:2005. AOAC INTERNATIONAL.
25. www.fda.gov/downloads/Food/FoodScienceResearch/ucm111508.pdf.
26. Luke, M.A., J.E. Froberg, G.M. Doose and H.T. Masumoto. 1981. Improved multiresidue gas chromatographic determination of organophosphorus, organonitrogen, and organohalogen pesticides in produce, using flame photometric and electrolytic conductivity detectors. J Assoc Off Anal Chem 64(5):1187–1195.
27. Anastassiades, M., S.J. Lehotay, D. Stajnbaher and F.J. Schenck. 2003. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and dispersive solid-phase extraction for the determination of pesticide residues in produce. J AOAC Int 86(2):412–431.




