Food allergies are serious medical conditions that are responsible for an estimated 30,000 emergency room visits and 150 deaths per year, and it is likely that many more less severe reactions go unreported. Allergies may affect up to 6% of children and up to 4% of the total U.S. population. Because there is no cure for food allergies, allergic consumers and their families must rely on avoidance to prevent reactions. This means that these consumers must be avid food label readers, and they need to know the source of everything that is in the food that they eat.
 To help these consumers and their families, Congress passed and the President signed the Food Allergen Labeling and Consumer Protection Act of 2004 (FALCPA). FALCPA requires that all food product labels declare in plain English the presence of any of eight specific foods/food groups or protein derived from one of these foods or food groups. These labeling provisions took effect in January 2006. The eight foods or food groups identified by FALCPA are believed to account for most allergic reactions, as well as the most severe food allergic reactions, although more than 160 other foods are known to occasionally cause food allergies (Table 1).
To help these consumers and their families, Congress passed and the President signed the Food Allergen Labeling and Consumer Protection Act of 2004 (FALCPA). FALCPA requires that all food product labels declare in plain English the presence of any of eight specific foods/food groups or protein derived from one of these foods or food groups. These labeling provisions took effect in January 2006. The eight foods or food groups identified by FALCPA are believed to account for most allergic reactions, as well as the most severe food allergic reactions, although more than 160 other foods are known to occasionally cause food allergies (Table 1).
FALCPA also allows manufacturers or individuals to obtain exemptions from the labeling requirements for specific ingredients through either a petition or notification process. To obtain an exemption, petitioners must provide FDA with scientific evidence that demonstrates that the ingredient “does not cause an allergic reaction that poses a risk to human health.” Those requesting an exemption through the notification process must provide scientific evidence that demonstrates that an ingredient “does not contain allergenic protein.” FDA’s evaluation of this scientific evidence may require, among other things, consideration of the question of whether allergenic proteins can occur in a food at a level that is too low to cause harm. In other words, these provisions of FALCPA could raise the issue of whether a level of an allergen (i.e., a threshold) exists below which either “does not cause” or “does not contain” would apply and, if so, how to establish this level. Understanding food allergen thresholds and developing a sound scientific framework for establishing such thresholds are therefore important to FDA’s analysis of and response to FALCPA petitions and notifications.
The Threshold Working Group
Early in the process of implementing FALCPA, FDA realized that the methods and data used to answer threshold questions for additives or contaminants in foods are not necessarily applicable to food allergens. This is because: (1) allergic consumers react to normal, naturally-occurring food components that are desirable and nutritious for most of the population; (2) there is a large range of sensitivities and reaction severities within the allergic population for each food allergen; (3) sensitivity and reaction severity can vary over time for an individual, so that it is difficult to predict whether, or how strongly, an individual will respond to each exposure; and (4) there are no validated animal or laboratory test systems that can be used to predict either response sensitivity or severity.
In response to the complexity of these issues, FDA formed an ad hoc interdisciplinary Threshold Working Group to identify approaches that might be used to establish thresholds for food allergens, and the advantages, limitations and data needs of each approach. The role of this group was not to choose or apply any particular approach. Furthermore, one option that is implicit in the report’s discussion of potential approaches is a decision not to establish thresholds at this time.
The Threshold Working Group reviewed scientific information on the mechanism of food allergy, the prevalence of various food allergies, the effects of processing on allergenic proteins in foods, the available methods for detection and characterization of allergenic foods and proteins, and the clinical studies that have been carried out to characterize food allergic responses. A report describing the findings of the Threshold Working Group was released for public comment in June of 2005, and was peer-reviewed by the FDA Food Advisory Committee (FAC) at a public meeting held in July 2005. The report was revised based on both the recommendations of the FAC and on the public comments received in the docket and at the FAC meeting. The revised report, “Approaches to Establish Thresholds for Major Food Allergens and Gluten in Foods,” was issued in May 2006, and is available online at www.cfsan.fda.gov/~dms/whalrgy.html.
The Approaches
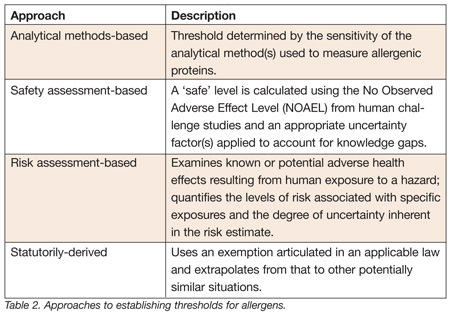 Four approaches were identified that might be used to establish thresholds for food allergens: analytical methods-based, safety-assessment based, risk assessment-based, and statutorily derived. These approaches are summarized in Table 2 and briefly described below.
Four approaches were identified that might be used to establish thresholds for food allergens: analytical methods-based, safety-assessment based, risk assessment-based, and statutorily derived. These approaches are summarized in Table 2 and briefly described below.
Analytical methods-based approach. This approach would establish thresholds based on the sensitivity of the analytical methods that measure levels of allergenic protein in specific foods. This type of approach has been used in the past, for example, in relation to levels of sulfiting agents in foods. The major advantages of this approach are that it would be straightforward and easy to implement. The major disadvantages would be that it could only be used in situations in which validated analytical methods are available and that any thresholds established using this approach would not be linked to public health. Further, it is not clear how the development of more sensitive or specific methods would affect the applicability of thresholds established using this approach. Another limitation in applying this approach is that at this time validated commercially available analytical test kits are available for some but not all food allergens.
Safety assessment-based approach. This approach would establish thresholds in a manner similar to that currently used for chemical contaminants and food additives. In the typical chemical safety assessment, an Acceptable Daily Intake (ADI) is derived using the exposure level for which there is no apparent adverse effect in conjunction with one or more uncertainty factor(s). The adverse effect levels may be obtained from controlled animal studies, human clinical studies, or epidemiological studies. The uncertainty factor(s) reflect the quality and quantity of the data available.
For food allergens, No Observed Adverse Effect Levels (NOAEL) and Lowest Observed Adverse Effect Levels (LOAEL) would be determined experimentally or by analysis of the published literature. A threshold would then be calculated by applying an appropriate uncertainty factor(s) to these levels. The advantages of this approach would be that it is relatively straightforward, and that there would be a direct link between any thresholds and public health. The major disadvantage of this approach would be that it relies on data on LOAELs and NOAELs that are often not readily available in the literature. Most published clinical studies only report the observed LOAELs for each patient. This is a problem if the LOAEL corresponds to the lowest dose tested because it is not possible to determine if the patients would have reacted to a lower dose. Further, different clinical studies use different criteria to qualify patients to be tested and to describe the observed responses.
Risk assessment-based approach. Risk assessment is a systematic, scientific examination of adverse health effects resulting from exposure to a hazard. This framework allows for organization of information, definition of uncertainties, and identification of data gaps. In quantitative risk assessment, the risk is expressed as a numerical estimate of the chance of illness or death after exposure to a specific hazard. This estimate represents the probabilities of certain events happening and the uncertainty associated with those events. Additional information on risk assessment including links to quantitative risk assessments and models is available at www.foodrisk.org.
The advantages of this approach would include the fact that it is scientifically rigorous, that it provides insights into both the level of risk and the degree of uncertainty associated with an exposure, and that it is linked to public health. The major disadvantage of this approach would be that it needs a great deal of data, much of which is not currently available in the literature. Further, no consensus has been reached regarding the most appropriate mathematical procedures and models to use for analyzing allergen reaction data.
Statutorily-derived approach. The application of this approach would establish thresholds by extrapolation based on a statutory exemption established by Congress for another purpose. In particular, Congress included a labeling exemption for “highly refined oils” in FALCPA. Therefore, it might be feasible to establish thresholds based on the level of protein found in “highly refined oils.” The advantage of this approach would be that it is derived from a provision of FALCPA. The major disadvantages would be that it is not linked to a scientific evaluation of data or to public health. In addition, to date, there is a lack of reliable methods to measure protein levels in oils.
Data Quality
In addition to identifying approaches that might be used to establish thresholds, the Threshold Working Group described criteria to use in evaluating the data that would be used in each approach. These included both general criteria that apply to all the approaches and criteria specific to each approach. For example, for the analytical methods-based approach, the criteria include evaluation of whether the method has been validated, the sensitivity of the method, whether it has been validated for use with both raw and processed foods, and whether it is practical.
Conclusion
The report summarizes the current state of scientific knowledge regarding food allergy, presents data needed to evaluate various approaches to establish thresholds, and discusses the strengths and limitations of each approach. Although the report acknowledges that the application of any approach may be limited by the availability of appropriate data, the Working Group recognized that the scientific understanding of food allergy is advancing rapidly. Improved methods for detecting and measuring allergens and improved risk assessment tools and modeling techniques are being developed and applied. Clinical studies that will provide more information on food allergen dose-response relationships are in progress or are being planned. Furthermore, the Threshold Working Group expects that any decisions on approaches for establishing thresholds for food allergens would require consideration of additional factors not covered in this report.
Further information and updates on FALCPA and food allergens can be found on the CFSAN home page.
Steven Gendel, Ph.D., served as the Writing Team Lead for the Threshold Report. He has conducted research in allergenicity assessment in FDA’s Center for Food Safety and Applied Nutrition (CFSAN).
Sherri B. Dennis, Ph.D., served as the Associate Lead for the Threshold Working Group. She serves as the Food Safety Risk Analysis Coordinator in FDA’s CFSAN.
Robert Buchanan, Ph.D., served as the Lead for the Threshold Working Group. He is an internationally recognized expert in predictive microbiology and risk assessment and serves as the Senior Science Advisor in FDA’s CFSAN.
Series editor Louise Dickerson serves with the Office of Food Safety, Defense, and Outreach in FDA’s CFSAN.
Acknowledgment
The members of the Threshold Working Group were Robert Buchanan (Lead), Sherri Dennis (Associate Lead), Steven Gendel (Writing Team Lead), David Acheson, Sue Anne Assimon, Nega Beru, Philip Bolger, David Carlson, Ricardo Carvajal, Catherine Copp, Kenneth Falci, Elizabeth Harden, Rhonda Kane, John Kvenberg, Stefano Luccioli, Douglas Park, Richard Raybourne, Terry Troxell, and Katherine Vierk.