Biofilms are an accumulation of inorganic and organic materials that can attach to most surfaces. Bacteria, both pathogenic and non-pathogenic, are incorporated into a biofilm during a stepwise formation. With time and nutrients, a biofilm and the bacteria within the biofilm will grow and become strongly attached to the surfaces. On occasion, parts of the biofilm slough off into the surrounding environment. This can be hazardous if the location of the biofilm happens to be in a food processing setting; after sloughing, the bacteria incorporated in the biofilm can contaminate other surfaces, as well as food products.
For this reason, removal of biofilms in the food processing environment is critical. Formulations and concentrations of cleaning and sanitizing agents, temperature, time of exposure and mechanical activity all play a role in the removal of biofilms. Additionally, bacteriocidal agents can be absorbed onto surfaces to help prevent initial formation or adhesion of bacteria. Cleaning and sanitizing regimes that incorporate steps to remove biofilms will result in a cleaner, safer processing environment and a safer product that has a longer shelf-life. Understand-ing how biofilms form, as well as how to detect, control and remove them from the food contact surfaces in the production plant, are all critical to the successful sanitation program.
What Are Biofilms and How Do They Form?
In nature, most bacteria do not exist as suspended, or planktonic, cells. Rather, they exist attached to a surface. Bacteria have the capacity to attach and colonize the surface of most natural and man-made materials. Attachment often results in the production of extracellular polysaccharides and changes in cellular morphology and growth rates. Addition-ally, diverse genes are expressed in bacteria that are attached to surfaces as compared to their planktonic counterparts.[1] As a result of these changes, surface-attached bacterial cells display increased resistance to toxic chemicals and biocides.[1,2]
While biocides have been shown to display nearly 100% effectiveness in the destruction of cells suspended in solution, they do not effectively destroy cells attached to surfaces unless mechanical action is employed. One explanation for the increased efficacy of biocides toward suspended cells includes the understanding that suspended cells have larger surface areas exposed to biocidal activity. Only the exposed areas of surface-attached cells are susceptible to biocidal activity.[3] Therefore, it is thought that bacterial attachment to surfaces may have arisen as a means of protection.
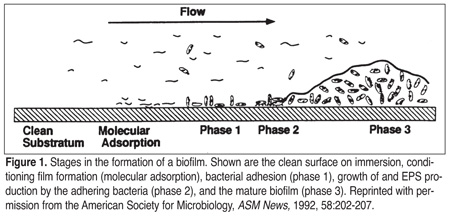 As bacteria attach to a surface and produce extracellular polysaccharides, a mass or biofilm is formed. Biofilm formation takes place in a step-by-step manner (Figure 1). First, inorganic or organic molecules are adsorbed to a surface. This creates a conditioning layer, or bacterial primer, that increases the ability of bacteria to attach to that surface.[4] Proteins often form conditioning layers that aid bacterial adhesion. In food production facilities, biofilm formation is found more frequently when high protein concentrations are present. Whey proteins, which are prevalent in dairy plants, have been shown to cause an increase in bacterial adhesion and selectively increase the adhesion of several milk-associated organisms.[5]
As bacteria attach to a surface and produce extracellular polysaccharides, a mass or biofilm is formed. Biofilm formation takes place in a step-by-step manner (Figure 1). First, inorganic or organic molecules are adsorbed to a surface. This creates a conditioning layer, or bacterial primer, that increases the ability of bacteria to attach to that surface.[4] Proteins often form conditioning layers that aid bacterial adhesion. In food production facilities, biofilm formation is found more frequently when high protein concentrations are present. Whey proteins, which are prevalent in dairy plants, have been shown to cause an increase in bacterial adhesion and selectively increase the adhesion of several milk-associated organisms.[5]
Once a conditioning layer is formed, bacterial adhesion ensues. Processing factors that increase bacterial attachment to surfaces include high or low pH extremes and high contact surface temperature; both will denature proteins, facilitating the formation of a conditioning layer. Further, low fluid flow rates over a biofilm allow increased nutrient contact time. Other factors include nutrient availability, which is ubiquitous in food plants; length of time that the bacteria is in contact with the surface; bacterial growth stage; and surface hydrophobicity.
In general, increased surface hydrophobicity enhances bacterial attachment. Stainless steel is an example of a hydrophobic surface. Bacillus spores, which also have a hydrophobic surface due to their outer coat proteins, have enhanced attachment capability on hydrophobic surfaces compared to vegetative cells.[6,7]Therefore, spores adhere to stainless steel in greater concentrations than do vegetative cells.[7]
Interestingly, cell viability has limited influence on attachment propensity. Live or dead cells will attach to selected surfaces with similar propensities.[7] Bacterial attachment is mediated by fimbriae, pili, and flagella, all of which are appendages extending outward from the cell surface. Bacterial attachment also is enhanced by extracellular polysaccharides that act to form a bridge between the bacteria and the conditioning layer.[3,5] This bridge actually is a combination of electrostatic, covalent and hydrogen bonding, along with dipole, Van der Waals, and hydrophobic interactions. Initially, the bonds between the bacteria and the conditioning layer may not be strong and can be easily removed by flowing water. However, with time, these bonds are strengthened making attachment irreversible.
Once embedded within a biofilm, injured or small, nutrient-deprived cells have the opportunity to repair, metabolize nutrients contained within the conditioning layer, grow and reproduce.[4] As growth continues, the copious volumes of extracellular polysaccharides that are produced further provide a protective barrier around the cells.[5]Inorganic and organic matter flowing over the biofilm becomes entrapped, increasing biofilm size and providing additional nutrient sources. Biofilms develop rapidly when there is a continuous source of nutrients.[8] Under such conditions, a biofilm may be considered "mature" within 24 hours and may continue to grow to millimeter proportions in a matter of days. Biofilm development can occur within one hour with 10% of the bacterial population irreversibly adhering to the conditioning layer. After an eight-hour production shift, greater than 91% of the bacteria are irreversibly attached.
As the biofilm matures, resistance against various disinfectants increases, which may be due to the increased production of extracellular polysaccharides.[5] Biofilm removal during the nightly sanitation routine becomes a difficult task, because the increased chemical contact time and mechanical activity required taxes both personnel and time constraints. If extended production runs are performed, weekly sanitation routines must be exceedingly rigorous in order to remove mature and recalcitrant biofilms.
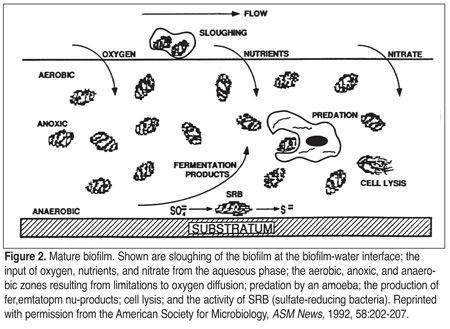 If nutrients are in close proximity to bacterial cells, motility requirements may be reduced, and thus, energy demands. Therefore, biofilms afford a protection that allows for extended bacterial longevity. Periodically, pieces of the biofilm may slough off due to flow rate dynamics, the shearing effects of flowing fluids, chemicals within the fluid, or changing properties of the biofilm bacteria (Figure 2). The released bacteria may be transported to a new location where biofilm formation can start again or the bacteria may remain in the fluid as a contaminant.[5]
If nutrients are in close proximity to bacterial cells, motility requirements may be reduced, and thus, energy demands. Therefore, biofilms afford a protection that allows for extended bacterial longevity. Periodically, pieces of the biofilm may slough off due to flow rate dynamics, the shearing effects of flowing fluids, chemicals within the fluid, or changing properties of the biofilm bacteria (Figure 2). The released bacteria may be transported to a new location where biofilm formation can start again or the bacteria may remain in the fluid as a contaminant.[5]
The Presence of Biofilms in Food Production Facilities
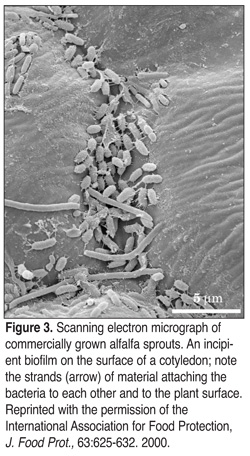 Biofilms have been found not only on food production surfaces but on food products themselves, including sprouts (Figures 3 and 4), spinach, and lettuce.[9-11] Surfaces in food production facilities, such as stainless steel, aluminum, glass, nylon materials, Buna-N, and Teflon seals, can harbor biofilms. Biofilm formation has been associated with environmental surfaces, such as floors, walls, pipes and drains.[12] Environmental surfaces have led to cross-contamination via air, personnel or cleaning.[13] Biofilms also are found on food contact surfaces, such as gaskets, conveyer belts, pasteurizers, and equipment containing crevices or dead spaces.[5,6] These areas often are hard to reach during cleaning and sanitation and thus optimal conditions for the formation and development of biofilms are established. The bacteria are protected from sanitizers while being exposed to a flow of water and nutrients. Surfaces that are pitted, scratched, cracked or corroded trap food particles and provide the bacterial adhesion sites required to begin the stepwise formation of biofilms.[13]
Biofilms have been found not only on food production surfaces but on food products themselves, including sprouts (Figures 3 and 4), spinach, and lettuce.[9-11] Surfaces in food production facilities, such as stainless steel, aluminum, glass, nylon materials, Buna-N, and Teflon seals, can harbor biofilms. Biofilm formation has been associated with environmental surfaces, such as floors, walls, pipes and drains.[12] Environmental surfaces have led to cross-contamination via air, personnel or cleaning.[13] Biofilms also are found on food contact surfaces, such as gaskets, conveyer belts, pasteurizers, and equipment containing crevices or dead spaces.[5,6] These areas often are hard to reach during cleaning and sanitation and thus optimal conditions for the formation and development of biofilms are established. The bacteria are protected from sanitizers while being exposed to a flow of water and nutrients. Surfaces that are pitted, scratched, cracked or corroded trap food particles and provide the bacterial adhesion sites required to begin the stepwise formation of biofilms.[13]

Biofilms contain diverse bacterial populations. In food production facilities, biofilms have been found to contain Listeria, Pseudomonas, Campylobacter, E. coli and Salmonella.[12] Non-starter lactic acid bacteria and thermoduric species often are found in dairy plant biofilms.[5,14]Thermoduric species are those that have increased heat resistance, grow at high temperatures and can survive pasteurization. Dairy plant biofilms often are predominated by a single bacterial species as the result of pasteurization, which eliminates most gram-negative species, allowing thermoduric species to grow without competition for nutrients.[5]
Biofilm Detection
The presence of a biofilm often can explain sporadic colony counts that are observed on the plates of swabs taken from environmental and food contact surfaces. A high total plate count or "marginal/reject" ATP bioluminescence value one day and low total plate count coupled with an "accept" ATP bioluminescence value the next day often indicates the presence of biofilms. This is due to the fact that the protective extracellular polysaccharide coating often inhibits bacterial removal with cotton-tipped swabs. Using sponges, where more mechanical action is involved during sampling in the form of rubbing, is one way to overcome this obstacle. Surface swabbing/sponging techniques often recover only a small proportion of the total biofilm bacterial populations. This is due to residual chemical sanitizers on the swab or sponge, recovery media used and aerobic growth conditions that limit complete bacterial detection.[1,5] Conduct-ing a total plate count along with a total anaerobic plate count may give a better indication of the bacterial population contained within a biofilm.
Additionally, tryptic soy agar with lecithin and polysorbate 80 can be used to neutralize chemical sanitizers remaining on the sponge or swab. Many food processing facilities use ATP bioluminescence luminometers to assess the effectiveness of cleaning and sanitation. For most brands of luminometers, 1.0 x 10[3] colony forming units per gram (CFU/g) are required for ATP detection. This level of detection has been determined using actively growing cells that are known to have increased levels of ATP. In cases where bacterial cells are stressed due to cleaning and sanitation procedures, the CFU/g needed for detection is even greater.[6] For embedded bacteria, where growth and activity are depressed, less energy in the form of ATP is required. This further increases the CFU/g needed for ATP detection. Despite these drawbacks, environmental monitoring via surface sponge or swabbing techniques and ATP readings still remain the most economical and effective means, to date, for biofilm detection. However, it is important to understand the limitations of these techniques.
Removal of Biofilms
Removal of biofilms is achieved by a combination of four factors: 1) formulations and concentrations of cleaning and sanitizing agents; 2) exposure time; 3) temperature; and 4) mechanical activity.[13] Removal of a mature biofilm most often will require extensive mechanical action, such as scrubbing or scraping in conjunction with the use of cleaning and sanitizing agents. Passing sanitizers over the surface removes the top layer and exposes the subsequent layers to nutrients; this hastens the growth and development of biofilms. Repeated sanitizer applications tend to favor the growth of bacteria directly under the surface. These bacteria then produce large amounts of extracellular polysaccharides that protect the cells from further sanitizer applications.[4] The goal of cleaning is to break the bonds of the extracellular polysaccharide conditioning layer. Once a bacterial cell is released from the protection of a biofilm, it is much less resistant to subsequent bacteriocidal sanitizers used in the cleaning/sanitizing regime.
Although the matrix of the biofilm will affect removal, there are a number of cleaning/sanitization combinations that have been successfully utilized. One such product, SU727 Trippel produced by Suomen Unilever, contains anionic active tensides, organic complex formers, alkali, and hypochlorite at a working pH of 12.5. This product has been proven to remove 90% of the bacterial load contained within biofilms along with the extracellular polysaccharide matrix.[1] Additionally, preparing a stock solution of 23% hydrogen peroxide and 4% peracetic acid, and then mixing the stock to a working concentration of 1-2%, combined with a contact time of 5 minutes at 25C has been found to effectively reduce the survival of Pseudomonas, Escherichia coli, Salmonella, Bacillus, Staphyloccus and Listeria.[2] Similarly, a 50% and 0.05% concentration of hydrogen peroxide and peracetic acid, respectively, mixed to a working concentration of 1-2% with a contact time of 5 minutes at 25C was shown to be effective at reducing survival of the above organisms contained within a biofilm. Hydrogen peroxide powder, mixed in 3-6% concentrations, also has been noted to be an effective biofilm removal agent. The oxidative activity of these solutions is thought to be responsible for the bacteriocidal mode of action.[2]
It should be noted that, in general, the greater the contact time, the more effective the bacteriocidal action. Additionally, any chemical treatment combined with mechanical action will remove biofilms more efficiently. Therefore, circulating water for clean-out-of-place (COP) tanks, floor scrubbers, or good old fashioned elbow grease with brushes or scrapers are highly recommended. However, care should be taken because some brushes and scrapers may be abrasive and leave scratches on stainless steel surfaces, further promoting biofilm formation. In many dairy applications, years and years of cleaning with green scrubbies has left its mark(s). In these situations, a strict, routine sanitation protocol is recommended.
An example protocol for COP parts is as follows: First, add parts to a chlorinated alkaline detergent (0.5 oz/gal, pH 11-12) in circulating water at 160F for 20 minutes. Second, rinse with potable water and place parts in circulating 160F water with phosphoric acid (1 oz/gal) for 20 minutes. Then, rinse with potable water and place parts in chlorine solution (0.3 oz/gal) for 15 minutes. Finally, rinse with potable water. This regime is most effective for detaching biofilms and has been found to be extremely effective on biofilms containing organisms such as Staphylococcus aureus. This may be due, in part, to the composition of the extracellular polysaccharide produced by this organism. The composition of the extracellular polysaccharide varies based on species. Depending on the composition of the biofilm, a combination of detergents may be required to remove it from the surface. Cations, in particular calcium, are thought to play a role in bacterial adhesion. The absence of cations often results in the detachment of bacteria. Therefore, chelators included in detergents may be effective in bacterial detachment and subsequent removal of biofilms.[13]
Cleaning by brushing, scrubbing and scraping surfaces or the use of circulating water often are necessary to detach the extracellular polysaccharide layer. The use of high-pressure spray hoses at distances greater than 250 mm from the surface is not recommended, because this will increase the generation of aerosols and will disperse bacteria over a wide area. Interestingly, high-pressure hoses used above 17.2 bar (250 psi) have not been shown to enhance biofilm removal or to significantly increase the removal of biofilms containing S. aureus.[13] For comparison, household water pressure ranges between 4 and 4.5 bar (60-65 psi); this pressure is too low to contribute significantly to the removal of biofilms.
Acid cleaners can be used to remove inorganic soil or material such as rust. Using softened water during cleaning often increases the effectiveness of these cleaning chemicals. When cleaning, water from the hose should be no less than 130F because the temperature drops 8 F to 10F as it flows from the nozzle and contacts equipment. Sanitizing using hot water (180F) instead of chemical sanitizers is permitted by the U.S. Department of Agriculture (USDA). However, this practice is not advisable because it aids in the formation of the conditioning layer by denaturing proteins and increasing the adhesion properties of equipment.
Nisin also has been employed as an anti-biofilm agent. Nisin absorbs to surfaces and acts as a bacteriocidal agent for adhering bacteria.[15] Nisin is a Generally Recognized As Safe (GRAS) substance. It is an extracellular protein excreted by some strains of Lactococcus lactis. Nisin has a mode of action that results in the formation of pores in the cell membrane of the bacteria. Pore formation leads to cell lysis and death. The bacteriocidal activity of nisin has been shown to target other gram-positive bacteria closely related to L. lactis and some gram-positive pathogens, such as Listeria monocytogenes.
The Best Defense
The best defense against biofilms in the food production facility is a good offense developed as part of the sanitation program. Since some bacteria within a biofilm may be pathogenic, strategies to reduce cross-contamination and to control or prevent the formation of a biofilm on food contact surfaces are of high priority and therefore, should be incorporated by food companies. Removal of biofilms in the food processing environment can be accomplished by paying attention to formulations and concentrations of cleaning and sanitizing agents, temperature, time of exposure and mechanical activity, and by using bacteriocidal agents on surfaces to help prevent adhesion of bacteria. Cleaning and sanitizing regimes that incorporate steps to remove biofilms will result in a more sanitary processing environment and a safer product that has a longer shelf-life.
Virginia Deibel, Ph.D., is CEO of Brain Wave Technologies, Inc., a food microbiology research and consulting firm where she utilizes her 15 years of experience in the food microbiology industry to provide clients with technical expertise analyzing, streamlining and improving all aspects of food production. Deibel and her team of scientists work out of a microbiology laboratory in Madison, WI, providing services that include customized food research projects, food safety seminars, HACCP, GMO testing, plant inspections and recall prevention and management planning. Recently elected to serve on the executive board of the Wisconsin Association of Milk and Food Sanitarians, Deibel also is a member of the International Association of Food Protection, American Society of Microbiology, Institute of Food Technologists (IFT), the Wisconsin IFT, where she serves on the board as treasurer, and the American Dairy Science Association.
Jean Schoeni, Ph.D., a food microbiologist with more than 22 years of experience in foodborne pathogen research, is Director of Research at Brain Wave Technologies. Schoeni's work with Salmonella, Escherichia coli O157:H7, Listeria monocytogenes, Staphylococcus aureus, Campylobacter jejuni, Yersinia enterocolitica, Clostridium botulinum, and Bacillus cereus has resulted in 15 publications and one patent. Schoeni recently served on the executive committee of the Institute of Food Technologists (IFT) Biotechnology Division and is a member of Wisconsin IFT and the American Society of Microbiology.
References
1. Bredholt, S., et al. Microbial methods for assessment of cleaning and disinfection of food-processing surfaces cleaned in a low-pressure system. Eur. Food Res. Tech., 209:145-152. 1999.
2. Wirtanen, G., et al. Efficacy testing of commercial disinfectants against foodborne pathogenic and spoilage microbes in biofilm-constructs. Eur. Food Res. Tech., Published online. 2001.
3. Bower, C.K. and M.A. Daeschel. Resistance responses of microorganisms in food environments. Intl. J. Food Microbiol. 50:33-44. 1999.
4. Kumar, C. and S.K. Anand. Significance of microbial biofilms in food industry: a review. Intl. J. Food Microbiol., 42:9-27. 1998.
5. Flint, S.H., et al. Biofilms in dairy manufacturing plantdescription, current concerns and methods of control. Biofouling, 11:81-97. 1997.
6. Storgards, E., H. Simola, A.M. Sjoberg and G. Wirtanen. Hygiene of gasket materials used in food processing equipment part 2: aged materials. Inst. Chem. Engin., 77:146-155. 1999.
7. Parkar, S.G., S.H. Flint, J.S. Palmer and J.D. Brooks. Factors influencing attachment of thermophilic bacilli to stainless steel. J. Appl. Microbiol., 90:901-908. 2001.
8. Marshall, K. Biofilms: an overview of bacterial adhesion, activity, and control at surfaces. ASM News, 58:202-207. 1992.
9. Fett, W. Naturally occurring biofilms on alfalfa and other types of sprouts. J. Food Prot., 63:625-632. 2000.
10. Morris, C.E, J.-M. Monier, and M.-A. Jacques. Methods for observing mibcroial biofilms directly on leaf surfaces and recovering them for isolation of culturable microorganisms. Appl. Environ. Microbiol., 63:1570-1576. 1997.
11. Seo, K.H. and J.F. Frank. Attachment of Escherichia coli O157:H7 to lettuce leaf surface and bacterial viability in response to chlorine treatment as demonstrated by using confocal scanning laser microscopy. J. Food. Prot., 62:3-9. 1999.
12. Joseph, B., et al. Biofilm formation by Salmonella spp. on food contact surfaces and their sensitivity to sanitizers. Intl. J. Food Microbiol., 64:367-372. 2001.
13. Gibson, H., et al. Effectiveness of cleaning techniques used in the food industry in terms of the removal of bacterial biofilms. J. Appl. Microbiol., 87:41-48. 1999.
14. Somers, E.B., et al. Biofilm formation and contamination of cheese by nonstarter lactic acid bacteria in the dairy environment. J. Dairy Sci., 84:1926-1936. 2001.
15. Bower, C.K., et al. Protein antimicrobial barriers to bacterial adhesion. J. Dairy Sci., 81:2771-2778. 1998.
Biofilms: Forming a Defense Strategy for the Food Plant