Ready-to-eat (RTE) meat products produced in U.S. Department of Agriculture (USDA)-inspected processing plants must achieve appropriate lethality of the most resistant vegetative bacterial pathogens reasonably likely to occur. Typically, processes are designed to kill 6.5 to 7.0 log CFU/g Salmonella as a food safety objective according to federal regulations. This food safety objective also happens to kill other vegetative bacterial pathogens that can occur in raw meat and meat ingredients, such as Escherichia coli O157:H7 and Listeria monocytogenes. Some processes like dry sausage fermentation and drying are targeted specifically to kill E. coli O157:H7.
Depending on the product, these so-called “lethality” processes could include heating (cooking), fermentation by use of starter cultures, drying or combinations of these processes. For instance, bologna products are heated to internal temperatures of typically greater than or equal to 160 °F and then cooled rapidly to maintain safety and quality. Products like pepperoni and hard salami are seeded with starter cultures, fermented and dried to achieve lethality and then cooled prior to final packaging. Products like prosciutto undergo a weeks-long curing process followed by a drying step that work together to provide the necessary lethality of potentially harmful bacteria.
Finished Product Packaging
At the packaging stage in RTE meat processing plants, most products are placed into plastic bags with certain barrier properties to moisture and air. The primary purpose of modified atmosphere or vacuum packaging is to delay oxidative rancidity of the lipids, and a secondary purpose is to delay microbial growth during shelf life. Packaging film for RTE meats is typically comprised of coextruded plastic materials, including combinations of nylon, polyethylene and polyvinyl dichloride. As mentioned, packaging film is designed to limit transmission of oxygen and moisture. Examples of oxygen transmission rates (OTRs) for RTE meats are the following: less than 18.6 cm²/m²24 h at 23 °C and 0% relative humidity (RH) and 3–5 cm²/24 h at 4.4 °C and 0% RH; moisture vapor transmission rates are commonly 0.4–0.6 g H2O/100 in²/24 h at 38 ºC and 90% RH. These barrier properties can be reduced by the degree to which packaging film is stretched, particularly in the corners of the packages. Once the packaging seal is broken, these barrier properties are lost.
Food Safety During Shelf Life
When RTE meat products reach the packaging environment in plants, they should be free of harmful bacteria. However, the inherent risk of recontamination by L. monocytogenes in meat processing environments has been well documented and must be addressed. As such, risk assessments should be conducted on these products in the finished packaged form to include assessment of their ability to prevent the growth of the pathogen for the duration of the refrigerated shelf life.
Products neither fermented nor dried (e.g., ham, turkey breast and bologna) are frequently formulated with antimicrobial agents, such as sodium or potassium lactate and sodium diacetate. These antimicrobial ingredients are widely used in the industry and have been extensively validated to control the growth of L. monocytogenes on vacuum- or modified atmosphere-packaged RTE meats held at refrigeration temperatures. Some research has also been done showing growth control of the pathogen when RTE meat products are stored exposed to air (i.e., open packages) at refrigeration.
Fermented and dried RTE meat products typically do not support the growth of L. monocytogenes due to acidity, low moisture and competition from the starter culture. In fact, the pathogen decreases (dies off) on many of these products when held at room temperature or under refrigeration. Behavior of L. monocytogenes and other foodborne bacterial pathogens on both types of RTE meats is reviewed below.
Survival and Growth of Foodborne Pathogens
Typical High-moisture, Non-fermented RTE Meats
Researchers from Colorado State University simulated contamination of RTE meats during slicing and handling at retail or at home.[1] Uncured, cooked turkey breast was vacuum-packaged and stored at 4 °C (39.2 °F) for 5, 15, 25 and 50 days before being opened, sliced and inoculated with L. monocytogenes. Inoculated turkey breast was stored aerobically in delicatessen bags at 7 °C (44.6 °F) for 12 days. In this retail/home-contamination simulation, mean growth rates (log CFU/cm²/day) of the pathogen during aerobic storage ranged from 0.14 to 0.16 in product with lactate and diacetate, equating to about a 2-log increase in L. monocytogenes on turkey breast within about 14 days. In similar work performed on cured ham with lactate and diacetate, the growth rates ranged from 0.18 to 0.25 log CFU/cm²/day,[2] equating to a 2-log increase on opened packages of ham within about 10 days.
However, Pal and others from the University of Minnesota[3] did not see growth of L. monocytogenes on cured, sliced ham with lactate and diacetate when stored in air at 8 °C (46.4 °F). In their work, growth rates of the pathogen on uncured sliced turkey breast with lactate and diacetate were not markedly different at 8 °C (46.4 °F) when stored in vacuum-packages versus air. Times to reach a 2-log increase were greater than 50 days.
Incidentally, the behavior of L. monocytogenes in pork chops or ham does not appear to be significantly different when meat is vacuum-packaged versus modified atmosphere-packaged.[4] Packaging atmospheres comprised of 60% O2, 20% CO2 and 20% N2 had only slight differences compared with 100% N2 on growth of total viable bacteria and lactic acid bacteria on cooked turkey breast and pork sausage stored in film with an OTR less than 35 cm²/m².[5]
A survey of retail and deli unpackaged ham in New Zealand found 4.5% of 301 samples contained L. monocytogenes following a 7-day simulated home storage at 5 °C (41 °F).[6] One sample had counts of L. monocytogenes near 3 log CFU/g, and 13 samples contained other Listeria spp. Retail delicatessen ham samples in New Zealand did not contain lactate and diacetate.[7] Surveys of the incidence of L. monocytogenes in retail, foodservice and delicatessen RTE meats indicated a higher prevalence of the pathogen than incidence in manufacturer-packaged RTE meats.[8,9]
Fermented and Dried RTE Meats
Fermented and dried meats are not favorable for the survival of vegetative bacterial pathogens. E. coli O157:H7 declined slightly on fermented and dried sausage during vacuum-packaged storage for 8 weeks at 4 °C (39 °F).[10] A slight decline in E. coli O157:H7 was also observed on semidry cooked summer sausage during vacuum-packaged storage for 7 days at 4 °C (39 °F) and at 25 °C (77 °F).[11] Inoculated L. monocytogenes declined on vacuum-packaged summer sausage during storage over several weeks at 4 °C (39 °F).[12]
On a Norwegian-style fermented dry sausage stored vacuum-packaged at 4 °C (39 °F) and 20 °C (68 °F), E. coli O157:H7, L. monocytogenes and Salmonella all declined by at least 1 log CFU, and after 46 days, some of the pathogens were undetectable.[13] These same three foodborne pathogens were later shown to decline on soudjouk-style fermented semi-dry sausage stored vacuum-packaged at refrigerated, ambient and abusive temperatures.[14] The rates of decline of the pathogens increased with increasing storage temperature. Others confirmed that generic E. coli and L. monocytogenes, as well as Yersinia enterocolitica, declined when fermented sausage was stored in air at 8 °C (46 °F) or 20 °C (68 °F).[15]
The survival of L. monocytogenes on sliced salami stored under air and vacuum at temperatures meant to simulate retail and home storage was investigated.[16] The degree of reduction in L. monocytogenes was greater with higher storage temperature: 25 °C (77 °F) greater than 15 °C (59 °F) greater than 5 °C (41 °F) storage. Additionally, vacuum packaging resulted in slower destruction of the pathogen than air packaging, and this effect increased as storage temperature decreased.
The survival of E. coli O157:H7 on pepperoni as affected by storage temperature and air, vacuum or modified-atmosphere packaging was studied by the University of Wisconsin-Food Research Institute (UW-FRI).[17] At ambient temperature, the pathogen declined by more than 1 log CFU/g within 7 days, and within 14 days, populations were significantly lower in air-stored samples than in vacuum-packaged samples. Decline in E. coli O157:H7 on refrigerated or frozen pepperoni slices was slight and was not significantly different between air, vacuum-packaged or modified atmosphere- packaged samples. A separate study found that storage of vacuum-packaged pepperoni sticks at 4 or 21 °C (39 or 70 °F) caused Salmonella typhimurium DT104 counts to decline by 4.6 and 6.6 log CFU/g within 56 days.[18] UW-FRI researchers also investigated the survival of E. coli O157:H7 during fermentation, drying and storage of full and reduced fat pepperoni under air or vacuum but found few significant differences.[17] Reductions were greater when pepperoni was stored at room temperature compared with refrigeration. Others found that post-process inoculated L. monocytogenes declined rapidly (> 1.5 log CFU/cm² in 6 days) on pepperoni under refrigeration and even more rapidly at ambient temperature.[19]
Foodborne bacterial pathogens do not survive well on dry-cured pork products either. Slices of dry-cured country-style hams were obtained from six different manufacturers and inoculated with E. coli O157:H7, L. monocytogenes, Salmonella spp. and Staphylococcus aureus, and populations were tracked weekly for 28 days of storage at 2 and 25 °C (35.6 and 77 °F).[20] Although survival varied with different dry-cured ham samples, on average, populations of each pathogen declined at both temperatures. Populations of S. aureus increased within 28 days on three of six dry-cured ham types stored at ambient but not refrigeration temperature. Although research comparing pathogen survival on air- and vacuum-packaged, stored, dry-cured ham appear to be lacking, counts of mesophilic aerobic microorganisms on dry-cured ham stored for 8 weeks in film with an 8.3 cm²/m² OTR were not different between vacuum, 100% N2 and 20% CO2/80% N2 atmospheres.[21]
Government Guidance on Storage of RTE Meats
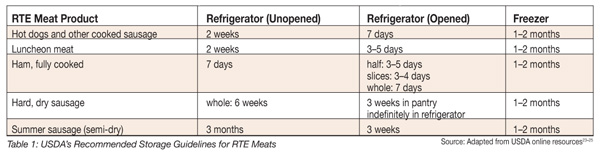 A 2003 quantitative risk assessment comparing the relative risk of listeriosis from various refrigerated RTE foods conducted by the U.S. Food and Drug Administration in collaboration with the USDA’s Food Safety and Inspection Service (FSIS), identified that storing refrigerated foods at or below 40 °F (4.4 °C) and consuming refrigerated RTE foods within less than 8 days could reduce the risk of illness from L. monocytogenes by more than 50%.[22] USDA-FSIS currently recommends that consumers set home refrigerator temperatures less than 5 °C (41 °F) and store opened or unopened packages of frankfurters for no longer than 7 or 14 days, respectively.[23] USDA-FSIS recommended storage conditions for other RTE meats are listed in Table 1.[23-25]
A 2003 quantitative risk assessment comparing the relative risk of listeriosis from various refrigerated RTE foods conducted by the U.S. Food and Drug Administration in collaboration with the USDA’s Food Safety and Inspection Service (FSIS), identified that storing refrigerated foods at or below 40 °F (4.4 °C) and consuming refrigerated RTE foods within less than 8 days could reduce the risk of illness from L. monocytogenes by more than 50%.[22] USDA-FSIS currently recommends that consumers set home refrigerator temperatures less than 5 °C (41 °F) and store opened or unopened packages of frankfurters for no longer than 7 or 14 days, respectively.[23] USDA-FSIS recommended storage conditions for other RTE meats are listed in Table 1.[23-25]
Conclusions
The published scientific literature on the fate of pathogenic bacteria of concern in RTE meat products with respect to storage of unopened and opened packages has been briefly reviewed. These studies clearly show that the pathogens of concern do not grow, and in fact decline, on semi-dry sausage, dry sausage and dry-cured pork products. However, USDA-FSIS-recommended consumer storage times for opened packages are very conservative for semi-dry and dry sausage and do not appear to be based on scientific studies of bacterial pathogen survival rates. Also, studies indicate that non-fermented, high-moisture RTE processed meats (e.g., ham and uncured turkey breast) formulated with lactate and diacetate can reasonably restrict the growth of foodborne bacterial pathogens of concern in opened packages when stored for up to 14 days at or below 4.4 °C (40 °F).[26] However, USDA-FSIS-recommended consumer storage times for opened packages of luncheon meats and ham range from 3 to 5 days. It is possible that these recommendations take into account the surveys showing home refrigerator temperatures tend to be greater than 4.4 °C (> 40 °F)[26] and revealing the mean temperature of deli meat in the U.S. is 7.1 °C ± 3.28 °C (44.8 ± 5.91 °F)[27] as well as the unknown degree of possible contamination that could occur in retail, foodservice and home settings.
The information reviewed here should be useful for setting guidelines for the use of opened packages of RTE meats. Some inferences might also be made to other RTE products of similar intrinsic properties. However, the degree of exposure of opened packages of RTE products to foodborne bacterial pathogens of concern is not fully understood and is the subject of ongoing research.
Peter J. Taormina, Ph.D., is Principal Scientist at John Morrell Food Group. He serves on the editorial boards of the Journal of Food Protection and International Journal of Food Microbiology and is the author of over 20 peer-reviewed publications on microbiological food safety.
References
1. Lianou, A., I. Geornaras, P. A. Kendall, J. A. Scanga and J. N. Sofos. 2007. Behavior of Listeria monocytogenes at 7 °C in commercial turkey breast, with or without antimicrobials, after simulated contamination for manufacturing, retail and consumer settings. Food Microbiol 24:433-43.
2. Lianou, A., I. Geornaras, P. A. Kendall, K. E. Belk, J. A. Scanga, G. C. Smith and J. N. Sofos. 2007. Fate of Listeria monocytogenes in commercial ham, formulated with or without antimicrobials, under conditions simulating contamination in the processing or retail environment and during home storage. J Food Prot 70:378-85.
3. Pal, A., T. P. Labuza and F. Diez-Gonzalez. 2008. Shelf life evaluation for ready-to-eat sliced uncured turkey breast and cured ham under probable storage conditions based on Listeria monocytogenes and psychrotroph growth. Int J Food Microbiol 126:49-56.
4. Michaelsen, A. R., J. G. Sebranek and J. S. Dickson. 2006. Effects of microbial inhibitors and modified atmosphere packaging on growth of Listeria monocytogenes and Salmonella enterica typhimurium and on quality attributes of injected pork chops and sliced cured ham. J Food Prot 69:2671-80.
5. Pexara, E. S., J. Metaxopoulos and E. H. Drosinos. 2002. Evaluation of shelf life of cured, cooked, sliced turkey fillets and cooked pork sausages− ‘piroski’−stored under vacuum and modified atmospheres at +4 and +10 °C. Meat Sci 62:33-43.
6. Cornelius, A. J., J. A. Hudson and T. L. Wong. 2008. Enumeration and growth of naturally occurring Listeria spp. in unpackaged ham. Food Microbiol 25:407-12.
7. Hudson, J. A. 2010. Personal Communication.
8. Gombas, D. E., Y. Chen, R. S. Clavero and V. N. Scott. 2003. Survey of Listeria monocytogenes in ready-to-eat foods. J Food Prot 66:559-69.
9. Lianou, A. and J. N. Sofos. 2007. A review of the incidence and transmission of Listeria monocytogenes in ready-to-eat products in retail and food- service environments. J Food Prot 70:2172-98.
10. Glass, K. A., J. M. Loeffelholz, J. P. Ford and M. P. Doyle. 1992. Fate of Escherichia coli O157:H7 as affected by pH or sodium chloride and in fermented, dry sausage. Appl Environ Microbiol 58:2513-6.
11. Calicioglu, M., N. G. Faith, D. R. Buege and J. B. Luchansky. 1997. Viability of Escherichia coli O157:H7 in fermented semidry low-temperature-cooked beef summer sausage. J Food Prot 60:1158-62.
12. Glass, K. A. and M. P. Doyle. 1989. Fate of Listeria monocytogenes in processed meat products during refrigerated storage. Appl Environ Microbiol 55:1565-9.
13. Nissen, H. and A. Holck. 1998. Survival of Escherichia coli O157:H7, Listeria monocytogenes and Salmonella kentucky in Norwegian fermented, dry sausage. Food Microbiol 15:273-9.
14. Porto-Fett, A. C., C. A. Hwang, J. E. Call, V. K. Juneja, S. C. Ingham, B. H. Ingham and J. B. Luchansky. 2008. Viability of multi-strain mixtures of Listeria monocytogenes, Salmonella typhimurium, or Escherichia coli O157:H7 inoculated into the batter or onto the surface of a soudjouk-style fermented semi-dry sausage. Food Microbiol 25:793-801.
15. Lindqvist, R. and M. Lindblad. 2009. Inactivation of Escherichia coli, Listeria monocytogenes and Yersinia enterocolitica in fermented sausages during maturation/storage. Int J Food Microbiol 129:59-67.
16. Gounadaki, A. S., P. N. Skandamis, E. H. Drosinos and G.-J. E. Nychas. 2007. Effect of packaging and storage temperature on the survival of Listeria monocytogenes inoculated postprocessing on sliced salami. J Food Prot 70:2313-20.
17. Faith, N. G., N. Parniere, T. Larson, T. D. Lorang and J. B. Luchansky. 1997. Viability of Escherichia coli O157:H7 in pepperoni during the manufacture of sticks and the subsequent storage of slices at 21, 4 and -20°C under air, vacuum and CO[2]. Int J Food Microbiol 37:47-54.
18. Ihnot, A. M., A. M. Roering, R. K. Wierzba, N. G. Faith and J. B. Luchansky. 1998. Behavior of Salmonella typhimurium DT104 during the manufacture and storage of pepperoni. Int J Food Microbiol 40:117-21.
19. Byelashov, O. A., B. A. Carlson, I. Geornaras, P. A. Kendall, J. A. Scanga and J. N. Sofos. 2009. Fate of post-processing inoculated Listeria monocytogenes on vacuum-packaged pepperoni stored at 4, 12 or 25 °C. Food Microbiol 26:77-81.
20. Ng, W. F., B.E. Langlois, B.E. and W.G. Moody. Fate of selected pathogens in vacuum-packaged dry-cured (Country-style) ham slices stored at 2 and 25 oC. J Food Prot 60:1541-7.
21. García-Esteban, M., D. Ansorena and I. Astiasarán. 2004. Comparison of modified atmosphere packaging and vacuum packaging for long period storage of dry-cured ham: effects on color, texture and microbiological quality. Meat Sci 67:57-63.
22. U.S. Food and Drug Administration Center for Food Safety and Applied Nutrition, U. S. Department of Agriculture-Food Safety Inspection Service, and Centers for Disease Control and Prevention. 2003. Quantitative assessment of relative risk to public health from foodborne Listeria monocytogenes among selected categories of ready-to-eat foods.
23. United States Department of Agriculture-Food Safety and Inspection Service. Date, September 8, 2006, Basics for Handling Food Safely. Available at: http://origin-www.fsis.usda.gov/Fact_Sheets/Basics_for_Handling_Food_Safely/index.asp. Accessed March 11, 2010.
24. United States Department of Agriculture-Food Safety and Inspection Service. Date, February 8, 2007, Food Product Dating. Available at: http://origin-www.fsis.usda.gov/Fact_Sheets/Food_Product_Dating/index.asp. Accessed
March 11, 2010.
25. United States Department of Agriculture-Food Safety and Inspection Service. Date, February 13, 2009, Focus on Sausages. Available at: http://www.fsis.usda.gov/fact_sheets/sausage_and_food_safety/index.asp. Accessed March 11, 2010.
26. Pouillot, R., M. B. Lubran, S. C. Cates and S. Dennis. Estimating parametric distributions of storage time and temperature of ready-to-eat foods for U.S. households. J Food Prot 73:312-21.
27. Audits International. 1999. Audits/FDA temperature databases. Available at: http://www.foodrisk.umd.edu/exclusives/audits/index.cfm. Accessed March 11, 2010, 2010.




