Food CGMPs, or Current Good Manufacturing Practices, are federal regulations enforced by the U.S. Food and Drug Administration (FDA) that apply to all human foods regulated by the FDA. These regulations, entitled “Current Good Manufacturing Practice in Manufacturing, Packing, or Holding Human Food” are found in Part 110 of Title 21 of the Code of Federal Regulations. Good manufacturing practices are the procedures and actions that are necessary to protect foods from becoming contaminated with filth, harmful substances or microorganisms, and also to prevent foods from becoming unfit for consumption through spoilage or decomposition. These regulations include the most important practices for protecting the food supply from contamination or spoilage during manufacturing, packing and storage.
It is necessary to periodically reexamine these regulations to determine whether or not they should be revised to incorporate control practices based on new knowledge about food safety risks and/or new technology. Recently, the FDA’s Center for Food Safety and Applied Nutrition (CFSAN) began the process of reevaluating the CGMP regulations to identify control practices that should be incorporated into the regulation and to identify current requirements that can be improved. On Nov. 2, 2005, CFSAN published a working group report that identified opportunities for the modernization of food CGMPs.[1]
The Relationship Between CGMPs and HACCP
CGMPs are preventive controls that work with other preventive control programs, such as Hazard Analysis Critical Control Point (HACCP) systems, to assure the safety and integrity of the food supply. CGMP controls differ from HACCP controls in several fundamental ways. CGMPs can be applied to any food manufacturing, packing or storage operation, regardless of the type of food or manufacturing process. In contrast, each HACCP control plan is specifically designed for a particular food or a related group of foods or food processes. CGMP controls address not only food safety but the prevention of contamination of food with filth and food decomposition or spoilage as well, while HACCP controls are focused on food safety. CGMP controls address the general design, construction and operation of food manufacturing and storage facilities, equipment sanitation, and employee hygienic practices and require that foods do not become adulterated as defined in Section 402 of the Federal Food, Drug and Cosmetic Act. In contrast, HACCP systems identify specific food hazards that are unique to a particular food or food manufacturing processes, and then specify critical controls that reduce or eliminate these food safety hazards. Each HACCP critical control requires monitoring, recordkeeping and verification activities that ensure the successful implementation of the critical control.
Both CGMPs and HACCP assure the safety of foods. For those food processes where food safety is controlled by HACCP systems, CGMPs are essential to a successful implementation of HACCP. CGMPs are often referred to as prerequisite programs for HACCP. HACCP preventive controls alone cannot assure the safety and wholesomeness of the food supply.
Why Do the CGMPs Need to be Revised Now?
CFSAN last revised the CGMP regulations in 1986. The primary purpose of the 1986 revision was to establish new, updated, or more detailed provisions concerning food industry personnel; plants and grounds, sanitary facilities, controls, and operations; equipment and utensils, warehousing, and distribution, and natural or unavoidable defect levels.[2] Although these regulations have not been updated in many years, they are broad enough to apply to many situations that could not be envisioned at their inception in 1969. The Center understands the importance of preserving the flexibility of the CGMP regulations, yet believes that there have been significant developments in our understanding of food safety hazards and in the control of these hazards to warrant a reexamination of the CGMP regulations.
Since 1986, new foodborne pathogens and food vehicles for pathogens have been identified. In 1986, Listeria monocytogenes had only recently been recognized as a foodborne pathogen and very little was known about the importance of controlling this organism in the food processing plant environment. Similarly, the significance of pathogens such as Escherichia coli O157:H7, Campylobacter jejuni, Cryptosporidium parvum, Cyclospora cayetanensis and Norovirus were not as well understood in 1986 as they are today. In addition to these new pathogens, familiar pathogens such as Salmonella are causing outbreaks in foods where they were previously not thought to be a problem. For example, Table 1 lists recent outbreaks involving fresh produce items. Modern good manufacturing practices can play a role in reducing the risk of these pathogens.
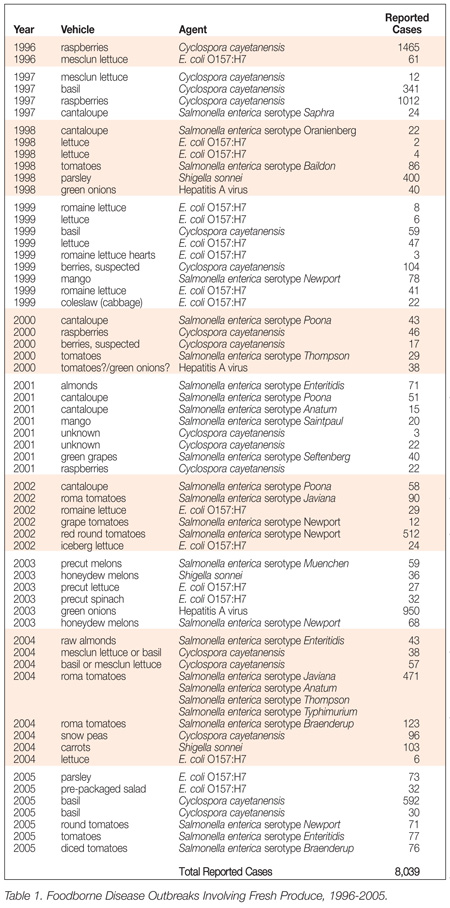 Table 1 Source: Food and Drug Administration, Center for Food Safety and Applied Nutrition, Office of Compliance, Emergency Coordination and Response Team
Table 1 Source: Food and Drug Administration, Center for Food Safety and Applied Nutrition, Office of Compliance, Emergency Coordination and Response Team
In 1986, the problem of food allergens was not appreciated to the extent that it is today. In a recent report, H.A. Samson writes that “Recent epidemiologic studies suggest that nearly 4% of Americans are afflicted with food allergies, a prevalence much higher than appreciated in the past.”[3] An analysis of food recalls reported to CFSAN between 1999 and 2003 showed that 34% of food recalls involved undeclared allergenic ingredients (e.g., milk, eggs, fish, crustacean shellfish, tree nuts, peanuts, wheat, or soybeans).[4] The prevention of cross-contact of food products with a food allergen and the prevention of errors in product labeling depend on current good manufacturing practices at the food processing establishment. Many of these recalls could have been avoided through the use of CGMP-type preventive controls.
The CGMP Modernization Effort is Risk–Based
A risk-based approach to food safety regulation is one in which regulatory requirements are matched to food safety outcomes. Ideally, risk-based regulations should give regulated establishments maximum flexibility to adapt the required controls to their unique situation. A modernized CGMP regulation should preserve the flexibility of the current regulation while requiring the implementation of controls that will significantly enhance food safety.
Through research and public comment, CFSAN’s Food CGMP Modernization Working Group identified current good manufacturing practice requirements that can effectively reduce the risk of foodborne illness and better support process-specific food safety control programs such as HACCP. The working group believes that these requirements can be implemented in a regulation that targets the food products and processes where they will be most effective. In its report on CGMP modernization, the working group identified opportunities for modernization of the regulations. Each of these opportunities and the practical implications are discussed below.
Training. The current regulations do not require that food establishment employees be trained in the principles of food safety and good manufacturing practices. Yet, proper training in these areas is likely to have a positive impact on food safety principles, for example, an increased awareness of food allergen control, proper equipment sanitation, personal hygiene, and compliance with temperature control requirements. In its report, the CGMP Modernization Working Group recommended that an amended CGMP regulation should include a requirement for appropriate training for supervisors and workers to ensure that they have the necessary knowledge and expertise in food hygiene, food protection, employee health and personal hygiene to produce safe food products. The working group stated that this training must be delivered in a manner easily understood by the worker and food processors must be able to show that each employee has been appropriately trained.
CFSAN has sought comments on this modernization opportunity. Those commenting on the need for training have been supportive and suggested that the training program needs to have a flexible curriculum that can be tailored to the specific needs of each establishment. Generally, comments do not support the need for an accreditation program for this training, preferring to use an “alliance” approach similar to what has been done with HACCP training.
Food Allergen Control Program. The current regulation does not specifically address the risk of major food allergens or require major food allergen control programs by food processing establishments that use major food allergens in their products. Major food allergens include milk, egg, fish, crustacean shellfish, tree nuts, wheat, peanuts and soybeans or any food that contains proteins derived from these foods. Clearly, food processing establishments that produce foods containing major food allergens need to have appropriate control programs, yet they should have the flexibility to adapt those programs to their unique circumstances. The Center’s CGMP Modernization Working Group stated that 21 CFR Part 110 should be amended to require food processors that handle any of these major food allergens to develop and adopt allergen control practices within their facilities. The working group stated that this control program must address six areas of control:
• Training of processing and super-visory personnel
• Segregation of food allergens during storage and handling
• Validated cleaning procedures for food contact equipment
• Prevention of cross-contact during processing through measures such as scheduling of production runs; control or rework; and use of dedicated production lines
• Product label review and label usage and control; and
• Supplier control program for ingredients and labels
Comments on this modernization opportunity were generally supportive of a requirement for a major food allergen control program. Commenters recommended that an amended regulation not require specific methods or controls, but focus on final outcomes. Commenters did not support a requirement for dedicated production lines, believing that cross-contact could be adequately prevented by other means.
Listeria Control Program for Ready-to-Eat Foods that Support Growth. During the past 20 years, our experience with L. monocytogenes has taught us that the production of certain high risk foods, in particular ready-to-eat (RTE) foods that support the growth of Listeria, requires additional care. This bacterium is especially difficult to control in the plant environment, and it is necessary to microbiologically monitor the food processing plant environment as both an assessment of the ongoing effectiveness of cleaning and sanitation operations and to identify harborages for this organism in the plant environment. For example, microbiological monitoring could include testing for Listeria on food contact surfaces and potential harborage areas on or near the food processing line.
It is particularly important to conduct such environmental monitoring in food processing establishments that produce RTE foods, because certain of these foods may allow the organism to grow to high numbers before the food is consumed. The current CGMP regulation requires that “All reasonable precautions shall be taken to ensure that production procedures do not contribute contamination from any source. Chemical, microbial, or extraneous material testing procedures shall be used where necessary to identify sanitation failures or possible food contamination.”[5] This provision clearly requires appropriate testing and controls to prevent harmful microorganisms such as L. monocytogenes from contaminating foods, but it does not specifically mention L. monocytogenes. CFSAN’s CGMP Modernization Working Group stated that 21 CFR Part 110 should be modified to specifically require a written environmental pathogen control program for food processors that produce RTE foods that support the growth of L. monocytogenes.
Comments on this modernization opportunity were generally supportive of a requirement for a written environmental monitoring program for processors of RTE foods that support the growth of L. monocytogenes. Commenters stressed that such a program must be flexible enough to accommodate the wide variety of processing conditions that exist in the industry. Commenters recommended that foods that do not support the growth of L. monocytogenes, frozen foods, and foods that are adequately pasteurized in the final package be excluded from this requirement. Commenters suggested that a monitoring program should include: (1) a statement of the monitoring objective or goal; (2) a description of the test organism, sampling plan, procedures and frequency, and the testing method; (3) recordkeeping and review procedures; and (4) corrective actions to be taken when a positive result is found.
Written Sanitation SOPs for Food Contact Surfaces. The current regulation does not require written cleaning and sanitation procedures for food contact equipment. However, proper cleaning and sanitation of food contact equipment is critical to preventing RTE foods from being systematically contaminated by harmful bacteria that might become established on unclean equipment surfaces. Harmful bacteria such as Salmonella and Listeria monocytogenes are known to contaminate RTE foods through unsanitary equipment surfaces. Also, written cleaning and sanitation procedures facilitate effective training and supervision of employees responsible for these tasks.
The Center sought comments on this modernization opportunity. Those commenting on the need for written sanitation standard operating procedures (SSOPs) for food contact surfaces have been supportive and suggested that SSOPs need to be general in nature to accommodate the variation in establishment “size, complexity and scope of operation.” Commenters recommended that SSOPs be required for all “food contact surfaces, including utensils” and “should include: (1) scope; (2) sanitation objective; (3) cleaning and sanitizing procedures; (4) cleaning and sanitizing schedules; (5) cleaning and sanitation monitoring activities; (6) corrective actions; [and] (7) records.”
Application of CGMP Requirements to Certain Agricultural Operations. 21 CFR Part 110.19(a) excludes “establishments engaged solely in the harvesting, storage, or distribution of one or more ‘raw agricultural commodities,’ as defined in section 201(r) of the Food, Drug and Cosmetic Act, which are ordinarily cleaned, prepared, treated, or otherwise processed before being marketed to the consuming public” from compliance with the regulations in Part 110. Part 110.19(b) states: “FDA, however, will issue special regulations if it is necessary to cover these excluded operations.”
One of the reasons for considering a modernization of the CGMP regulations is the significant increase in our understanding of foodborne illness. With respect to fresh produce, the Centers for Disease Control and Prevention (CDC) have reported that “…the mean number of reported outbreaks associated with fruits and vegetables more than doubled from the period 1973 to 1987 (4.3 per year) to the period 1988 to 1991 (9.75 per year). The mean number of persons affected by reported produce-associated outbreaks more than doubled (242 per year in the first period compared with 614 per year in the second period).”[6] Recent investigations of outbreaks linked to fresh produce have identified contamination during production and harvest, initial processing and packing, distribution, and final processing as the likely source of product contamination.[7] Data collected by the FDA supports the observation that foodborne disease outbreaks are being linked to fresh produce items more frequently than in previous years.
The data presented in Table 1 includes only those outbreaks that CFSAN was able to clearly link to a fresh produce item(s). Many more foodborne disease outbreaks involved a suspected link to fresh produce, but such a link could not be proven. Fresh produce is an important part of a healthy diet and we must not allow the safety of fresh produce to come into question. The agency has aggressively sought to promote good agricultural practices (GAPs) as a means of reducing the risk of foodborne illness from fresh produce. In 1998, the agency published its Guide to Minimize Microbial Food Safety Hazards for Fresh Fruits and Vegetables as a means of informing raw agricultural product producers and packers of important food safety controls.[8] Representatives of the melon industry (under the auspices of the Produce Marketing Association and the United Fresh Fruit and Vegetable Association) developed a guidance document entitled “Commodity Specific Food Safety Guidelines for the Melon Supply Chain” and the agency added a link to its website for this document in November of 2005.[9] Similarly, representatives of the fresh produce industry (the International Fresh-cut Produce Association, Produce Marketing Association, United Fresh Fruit and Vegetable Association, and Western Growers) developed produce-specific food safety guidelines to which the agency also included a link on its website in April of 2006.[10] Recently, the agency published a draft of it guidance document for fresh-cut produce, “Guide to Minimize Microbial Food Safety Hazards of Fresh-cut Fruits and Vegetables.”[11]
Although the vast majority of fresh produce items are consumed without any adverse health risk, the recent history of foodborne disease outbreaks linked to these foods demands implementation of controls that will eliminate or reduce contamination of fresh produce items. In light of the recent investigational findings described above, the agency’s CGMP Modernization Working Group recommended that the agency consider removing the exclusion from CGMP compliance in 21 CFR Part 110.19 for establishments engaged solely in the harvesting, storage, or distribution of raw agricultural commodities. However, fresh produce items are fundamentally different from many processed foods. Clearly, not all of the current provisions of 21 CFR Part 110 are applicable to fresh produce items. The question becomes: Which provisions should be applied to producers of fresh produce items and at what stage of food production should these provisions apply? For example, should farms be required to meet certain food CGMP requirements or should CGMP requirements be limited to packing houses? Clearly, there is a pressing need for additional research into the modes of pathogen contamination of fresh produce and the controls that can effectively prevent, reduce or eliminate this contamination. It is very likely that controls unique to fresh produce products will be needed. Is it appropriate to include scientifically validated control requirements in 21 CFR Part 110? If not, how can these controls be effectively implemented for all producers of fresh produce items sold in the United States?
Comments received in response to this modernization opportunity did not directly address the issue of the nature of required controls. Commenters suggested that CFSAN already has sufficient authority to assure the safety of fresh produce items and that it is not appropriate to apply the provisions of Part 110 to raw agricultural commodities. Commenters stated that the agency should continue to focus on the “refinement and promotion of GAPs guidance” as a means of assuring produce safety.
Temperature Controls. During the course of its public outreach efforts on CGMP modernization, the agency received several comments about the temperature requirements in 21 CFR 110.80(b)(3)(i) and 21 CFR 110.80(b)(3)(iii). Commenters stated that these temperature requirements should either be deleted or made consistent with Food Code 2001.[12] In its report, the CGMP Modernization Working Group noted that specific temperature requirements in this regulation may be problematic, given that pathogens such as L. monocytogenes are capable of growth at temperatures well below those specified for refrigerated foods. The working group recommended that the agency request further comments and suggestions on the use of time-temperature relationships, and on microbial growth models, and how these can be incorporated into regulations or guidance to ensure proper refrigerated storage or hot holding.
Comments on this modernization opportunity suggested that the agency take an approach similar to one suggested by the Institute of Food Technologists (IFT) in its Task Order No. 4 Report to FDA entitled Evaluation and Definition of Potentially Hazardous Foods.[13] Commenters suggested that references to specific temperatures should be deleted and replaced with general language requiring foods to be “held at temperatures that will prevent them from becoming adulterated, including [during] storage and distribution.” Commenters also suggested that the agency develop a guidance document to support the requirements of Part 110.
Conclusion
The CGMP regulations have served the agency and the public well during the almost 40 years since they were first published. The challenge is to retain in these regulations the provisions that have proven value, and to amend the regulation in a way that accounts for new knowledge about food safety risks and controls, while allowing the industry to comply with the regulations in the most efficient manner. To this end, proactive stakeholder input is critical.
Thus far, comments on the CGMP Modernization initiative have been very useful in shaping the agency’s thinking. As the FDA proceeds to develop proposed rules, there will be further opportunity for comments. 21 CFR Part 110 is important to the preservation of a safe and wholesome food supply and it should be continuously updated to reflect the most effective food production practices.
Don L. Zink, Ph.D., is Senior Food Scientist with the U.S. Food & Drug Administration, Center for Food Safety and Applied Nutrition, Office of Plant and Dairy Foods and Beverages. He is Lead Scientist for food processing and microbiological issues related to plant and dairy foods, beverages and infant formula, and is currently involved in policy development related to GMP modernization and as project officer for the National Center for Food Safety and Technology. Zink is a member and subcommittee chairman of the National Advisory Committee on Microbiological Criteria for Foods, and previously served as vice-chairman, Committee on Food Microbiology, International Life Sciences Institute, from 1999 to 2002. He is a member of Food Safety Magazine’s Editorial Advisory Board and recipient of the 2006 Food Safety Magazine Distinguished Service Award.
References
1. Food CGMP Modernization Working Group. 2005. Food CGMP Modernization—A Focus on Food Safety. cfsan.fda.gov/~dms/cgmps3.html.
2. 51 Federal Register 22458 (June 19, 1986).
3. Sampson, H.A. 2004. Update on Food Allergy. J. Allergy Clin. Immunol. 113(5):805-819.
4. U.S. FDA. 2004. Food GMP Modernization Working Group: Report Summarizing Food Recalls, 1999-2003. cfsan.fda.gov/~dms/cgmps2.html.
5. 21 CFR Part 110.80, Subpart E – Production and Process Controls.
6. Tauxe, R., et al. 1997. Microbial Hazards and Emerging Issues Associated with Produce A Preliminary Report to the National Advisory Committee on Microbiologic Criteria for Foods. J. Food Protect. 60(11)-:1400-1408.
7. Tauxe, R. E. 1997. Emerging foodborne diseases: an evolving public health challenge. Emerging Infect. Dis. 3(4):425-434.
8. U.S. FDA. 1998. Guide to Minimize Microbial Food Safety Hazards for Fresh Fruits and Vegetables. cfsan.fda.gov/~dms/prodguid.html.
9. Commodity Specific Food Safety Guidelines for the Melon Supply Chain. 2005. cfsan.fda.gov/~dms/melonsup.html.
10. Commodity Specific Food Safety Guidelines for the Lettuce and Leafy Greens Supply Chain. 2006. cfsan.fda.gov/~dms/lettsup.html.
11. U.S.FDA. 2006. Draft Guide to Minimize Microbial Food Safety Hazards of Fresh-cut Fruits and Vegetables. cfsan.fda.gov/~dms/prodgui2.html
12. U.S. FDA. Food Code 2001. Washington, DC.
13. IFT. 2001. Evaluation and Definition of Potentially Hazardous Foods; IFT/FDA Contract No. 223-98-2333. cfsan.fda.gov/~comm/ift4-toc.html.




