The foods we eat are extraordinarily complex in terms of texture, granularity and biochemical and microbial composition. At the microscale, our foods provide a bonanza of physical and nutritional niches that can be exploited by various microorganisms, including human pathogens. Physical manipulations commonly used in food preparation, such as slicing, chopping, grating and grinding, both release nutrients and provide additional colonizable surface area.[1] Things get even more complicated when we consider that a typical recipe combines different ingredients from multiple plant and/or animal sources, each with its own capacity to support microbial growth and its own intrinsic microbial content, which could be classed as either “normal” flora or contaminants. This complexity of foods presents challenges to microbiologists, whose job it is not only to control the growth of spoilage and pathogenic microbes but also to screen for and detect specific organisms, should they be present.
Some foods, such as raw poultry, are more likely than others to host bacterial pathogens, including Salmonella and Campylobacter, sometimes at relatively high levels.[2] Other foods may contain only trace levels of these or other pathogens, if at all. The rare and/or sporadic nature of such contamination makes these organisms difficult to detect, a problem that may be further compounded by low infectious doses for some pathogens: as few as 10 CFU for Escherichia coli O157:H7, 500–800 CFU for Campylobacter or 10–100 virions for norovirus.[3] Apart from bulk sample volume, properties of foods that can complicate (or thwart) successful detection of target pathogens, especially at low levels, include the presence of particulate matter, high viscosity, formation of macromolecular networks upon addition of water, opacity or coloration, high fat content, high loads of background microflora, heterogeneous distribution of target organisms and, in some foods (e.g., alfalfa sprouts), biofilms.
Pathogen Detection — From Soup to Nuts
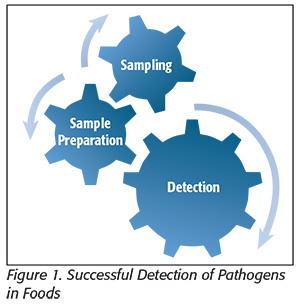 Successful detection of pathogens in foods involves the seamless integration of three interdependent steps: 1) statistically validated sampling, 2) pre-analytical sample preparation (the physical, chemical and/or enzymatic manipulations needed to address the issues and limitations posed by complex food matrices) and 3) detection (Figure 1). Weak links in any of these steps will propagate through the system and degrade the end result and, in a worst-case scenario, lead to false-negative results. “Upstream” methods for sampling have long been established,[4] and the past 25 years or so have seen a revolution in development of the hardware and reagents needed for truly rapid detection of pathogens. However, even the most sophisticated detection methods cannot reach their full potential without next-level advancements in pre-analytical sample preparation, which is the key bottleneck in getting from sample to answer when detecting pathogens in foods.[5]
Successful detection of pathogens in foods involves the seamless integration of three interdependent steps: 1) statistically validated sampling, 2) pre-analytical sample preparation (the physical, chemical and/or enzymatic manipulations needed to address the issues and limitations posed by complex food matrices) and 3) detection (Figure 1). Weak links in any of these steps will propagate through the system and degrade the end result and, in a worst-case scenario, lead to false-negative results. “Upstream” methods for sampling have long been established,[4] and the past 25 years or so have seen a revolution in development of the hardware and reagents needed for truly rapid detection of pathogens. However, even the most sophisticated detection methods cannot reach their full potential without next-level advancements in pre-analytical sample preparation, which is the key bottleneck in getting from sample to answer when detecting pathogens in foods.[5]
Feeding the Need for Funding
With these challenges in mind, the food microbiology research community has focused intently in recent years on unstopping the sample prep bottleneck, spurred on by private funding from the food industry and federal funding from agencies such as the U.S. Department of Agriculture (USDA) and the U.S. Food and Drug Administration (FDA). For example, over the past few years, requests for proposals for USDA’s Agriculture and Food Research Initiative Competitive Grants Program have routinely encouraged submissions focused on development and validation of “novel concentration and purification methods for the rapid, low-cost and efficient isolation or capture of viable infectious human pathogens from foods or environmental samples relevant to food production,” with special preference given for methods applicable to multiple matrices and pathogens. In 2014, FDA posed a nontraditional, $500,000 open challenge to “scientists, academics, entrepreneurs and innovators from all disciplines” to submit concepts aimed, in part, at accelerating or eliminating sample prep for detection of Salmonella in fresh produce.[6] This FDA Food Safety Challenge received submissions from 49 teams, and five finalists were awarded $20,000 each to further their concepts. Ultimately, one university-led team and one private company were selected to share the remaining $400,000 in prize money. These financial incentives and support for development of sample prep methods simultaneously acknowledge the importance of the problem and help facilitate work toward development of solutions that can benefit the food industry and regulatory agencies and, ultimately, improve public health.
Basic Categories of Sample Prep
Pre-analytical sample prep methods can be broadly organized under several basic categories, reflecting whether they are targeted toward specific organisms and the fundamental mechanism(s) behind their operation. An overarching distinction between different types of sample prep methods is whether the method is non-specific or specific in nature. Non-specific methods can be thought of as “catch-all” or “brute force” in nature and include methods used to capture and/or concentrate a wide range of microbes (and sometimes co-concentrate particulate matter) in a sample. Non-specific methods may be used to make a “first cut” that prepares the sample for additional downstream manipulations. Examples include “pull-down” methods used to precipitate bacteria or viruses from large volumes of liquid, such as the use of flocculents to precipitate bacteria or granular activated carbon for adsorptive recovery of viruses from large volumes of water.[5, 7] Other non-specific approaches include the use of cationic magnetic beads or metal hydroxide particles for charge-based capture of microbial cells, which carry an overall negative surface charge, and centrifugation, the size selectivity of which can be modulated by speed, duration and the use of density-gradient media. The simplest use of centrifugation as a sample prep step is the use of slow, short spins to cause sedimentation of large food particles, such as alfalfa sprout debris, leaving bacterial cells in suspension for analysis.[8] Continuous-flow centrifugation, a variant technique capable of handling larger volumes than typical “batch” centrifuges, has also been used for concentration of Bacillus and Clostridium spores from milk in support of homeland security efforts.[9] Specific methods are those targeting particular cell types. These can still be relatively broad, such as with lectin-based approaches for selective capture of Gram-negative bacteria, where concanavalin A binds to lipopolysaccharide residues in the Gram-negative outer membrane, or similarly, through use of the antibiotic vancomycin as a ligand for selective capture of Gram-positive bacteria. The level of specificity can be narrowed or “tuned” as desired through use of bioaffinity reagents having more restricted recognition spectra, as with the use of antibodies that bind specifically to antigens present only on the surfaces of certain cells, Salmonella spp. or S. Typhimurium, or through the use of phage tail fibers specific for E. coli O157:H7, for example.
Additional levels of distinction between different sample prep approaches can be made on a mechanistic basis—looking at how these methods accomplish their task. We can class methods used to capture and/or concentrate cells from a sample as physical methods if they do so through applying some physical force to the cells. Examples are varied and include methods ranging from filtration to centrifugation to even acoustic, magnetic or dielectrophoretic selection or concentration methods (application of sonic, magnetic or electrokinetic forces). Chemical (or enzymatic) methods are those applied to modify the structure of complex food matrices through chemical or enzymatic digestion. Examples include the “matrix lysis” approach used by Mayrl et al.[10] to chemically disintegrate various food matrices including fish, meat, ice cream and eggs, the use of proteinases for enzymatic digestion of chicken extracts or rinses[11] or use of ionic liquids to solubilize foods for extraction of bacteria.[12]
In practice, many sample prep techniques can be most accurately classified as combination methods, as they combine both physical and chemical or enzymatic approaches. For example, in the matrix lysis technique, chemical treatment is first used to degrade the food matrix and release bacterial cells, which are then collected via physical means. Similarly, Vibbert et al.[11] used enzymatic pretreatment of chicken extracts and rinses to enhance the efficacy of cell collection using microfiltration (a physical method). Other combination approaches include a method for physicochemical extraction of bacteria from homogenized eggs. In this method, egg samples are mixed briefly (30 seconds) with oleic and caprylic acids, which react with and break up the egg matrix, forming water as a byproduct; as with matrix lysis, released bacteria are then harvested via centrifugation.[13] Similar “treat and spin” approaches have been described for milk, using either enzymatic[14] or chemical[15] approaches.
Especially with commercial sample prep products that are available as kits, there may be a tendency for end-users to adopt a “kittology” mindset, where an approach is blithely applied as a formulaic “black box.”[16] Categorization of sample prep methods as described above can be a useful exercise for focusing the end-user’s mind on the processes behind each technique and on considering what stresses and forces may be exerted on food microbiota. This, in turn, can help reveal potential limitations or pitfalls of a given method and shed light on how these might affect the result, or whether it is even appropriate for a given food-pathogen pairing.
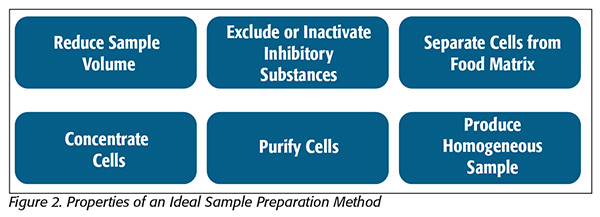 Finally, microbiologists must decide whether they are interested in capturing, concentrating and analyzing whole cells (or virions) or cellular components such as nucleic acids, which can be extracted directly from complex food matrices for culture-independent analyses. Because the broadest range of analytical possibility is available when starting with intact and viable cells, this article will focus primarily on cell-focused sample prep methods (Figure 2[5]).
Finally, microbiologists must decide whether they are interested in capturing, concentrating and analyzing whole cells (or virions) or cellular components such as nucleic acids, which can be extracted directly from complex food matrices for culture-independent analyses. Because the broadest range of analytical possibility is available when starting with intact and viable cells, this article will focus primarily on cell-focused sample prep methods (Figure 2[5]).
Dedicated Sample Preparation Equipment
Standard equipment used in food microbiology labs has evolved over the years, shaped and driven by the need for increased convenience, throughput and efficacy. It is fitting that some of the first sample prep equipment used in food analysis was adapted directly from the kitchen—the mortar and pestle, the blender, the sieve and cheesecloth, as prime examples. The expense of Waring blenders (currently about $2,000 to $3,000 for a scientific-grade blender) and the need for sterilization between experiments place substantial limitations on throughput and convenience, hence, the wide popularity of commercial instruments for sample homogenization that utilize presterilized and disposable plastic bags. Also, the ingenuity of individual researchers knows no bounds, resulting in clever do-it-yourself solutions engineered to assist with manually difficult tasks, such as the paint shaker-like instrument reported by Dickens et al.[17] for shaking turkey carcasses in support of rinse sampling.
Although some of these instruments have been around for so long that their use seems almost reflexive, as with any element in the sample-to-answer workflow, microbiologists still must give careful consideration to their appropriate use. For example, when enumerating surface bacteria on acidic fruits such as strawberries or tomatoes, methods that don’t decompartmentalize tissues and release acid may result in higher plate counts for acid-sensitive organisms. Additionally, high-speed blending is not only a recognized method for nonlethal, shear-based removal of flagella from organisms such as Salmonella,[18] but it can also lead to higher cell counts for bacteria having a streptococcal morphology, due to breakage of cell chains into smaller units.[19] Therefore, blending might not be appropriate for procedures that will subsequently rely on the presence of flagella or when analyzing samples where Streptococcus, Enterococcus or Leuconostoc spp. are expected. “Legacy” sample prep methods should not be discarded because they are older or simpler than newer approaches; Bai et al.20 found that sonication-based homogenization of orange juice samples reduced the yield of bacterium Candidatus Liberibacter asiaticus DNA by 86 percent compared with a mortar and pestle. This example highlights the fact that careful consideration of the purpose of an experimental procedure and of the cellular analyte(s) to be detected can often help guide the choice of the most appropriate sample prep techniques.
Specialized sample prep instrumentation available commercially or in development includes automatic nucleic acid preparation instruments, vacuum-based surface samplers and instruments for capture and concentration of select pathogens from recirculated food suspensions and to electrophoretically concentrate DNA from complex samples, including those containing large amounts of PCR (polymerase chain reaction) inhibitors, such as humic acids.[21] Weimer et al.[22] (finalists in the 2014 FDA Food Safety Challenge) developed a system that uses a fluidized bed of antibody-functionalized glass beads to capture select organisms in liquified food or environmental samples at processing rates exceeding 100 mL/minute. The dead-end filtration system described by Lim et al.[23] has been applied for collection and concentration of total coliforms and spiked E. coli O157:H7 from 50-L produce wash samples, with subsequent detection via electrochemiluminescent immunoassay. Continued development and commercialization of such specialty sample prep equipment will provide industry with microbial testing tools that are commensurate with their needs for high-throughput testing of large-volume food and environmental samples.
Better Reagents, Better Results
Although we typically approach the idea of sample preparation from the perspective of the upstream physical and chemical manipulations needed to extract, separate and concentrate target cells or their components from foods, we can expand the use of the term to include the reagent choices made immediately prior to the detection step, which can effectively be considered as “last minute” or “just in time” sample prep. One example is in molecular diagnostics such as PCR, which is susceptible to biochemical inhibitors or may suffer from limitations due to degradation of target DNA. In addition to sample preparation actions for inhibitor removal that may be performed during template isolation (sequestration of polyphenolic compounds with polyvinylpyrrolidone, binding of inhibitory cell components with ion-exchange resins, etc.), PCR-optimizing choices can also be made at the level of reaction-mix composition: adjustment of magnesium concentration, addition of PCR enhancers such as betaine, dimethyl sulfoxide or formamide, or use of polymerases that are less prone to biochemical inhibition.[24] In many applications of PCR, the sample preparation mantra of “dilution is the solution” applies, meaning that if sufficient template DNA is present, inhibitors can simply be diluted out.[25]
However, in cases involving low template concentrations or degraded DNA, this is not a tenable approach. In criminal forensics, DNA evidence is sought from blood or other bodily fluids to make an arrest or prosecute a legal case,[24] but these samples may contain inhibitors or be degraded (the latter being especially true if the sample has been exposed to the environment for some time, or for archival samples collected and stored prior to the advent of molecular diagnostics). In microbial forensics, where the “criminals” are the pathogens that we seek to track for epidemiological purposes (to trace the source and the route from environmental contamination to disease), similar issues of low template presence and/or DNA degradation may also be in play.[26] In such situations, it may be possible to borrow lessons learned from parallel fields, such as criminal forensics or the study of ancient DNA. These may take the form of data handling and analysis procedures or novel reagent choices (e.g., alternate DNA polymerases).[24, 25, 27, 28] In this latter category, several naturally occurring Y-family polymerases or chimeric enzymes were examined for their abilities to amplify damaged DNA, with three found to have acceptable processivity and abilities to replicate past lesions typical of damaged DNA, including abasic sites and various dimers and adducts.[28] Alternatively, a combinatorial approach using molecular breeding (directed molecular evolution through gene shuffling) has been used to generate novel polymerases based on genes from various Thermus spp. that can replicate past DNA lesions and outperformed standard Taq polymerase by up to 150 percent with 47,000–60,000-year-old cave bear DNA and also yielded product at dilutions where Taq did not.[27] These approaches highlight the potential for making experiment-optimizing “last minute” or “just in time” sample preparation choices at points as late as the reagent choice step: immediately prior to obtaining a diagnostic output. As described here, such choices may include the use of novel polymerases capable of replicating past DNA lesions commonly encountered in ancient DNA, or in contemporary but damaged DNA exposed to the vagaries of the environment. Lessons learned from the allied fields of ancient molecular diagnostics or criminal forensics might therefore have direct applications to improving diagnostic results in food microbiology.
Unanswered Questions
Low-moisture foods once thought to be intrinsically safe, such as peanut butter and flour, are now recognized as potential vectors for foodborne disease. There is intense interest in understanding the physiological stresses resulting from exposure to such hostile environments and in the adaptations that enable pathogens such as Salmonella and E. coli to survive in these foods and cause disease. Additional work is needed to determine the practical impact of food or food processing environments on our abilities to capture and concentrate pathogens using current sample prep approaches. Gross changes in the physiology and corresponding biomolecular content of cells that have been exposed to environmental stressors could potentially render current sample prep methods less effective. For example, expression of bacterial flagella can be modulated by nutritional or environmental conditions such as the presence or absence of certain amino acids or temperature. If prevailing environmental conditions result in a down-shift in expression of flagella, then how effective will a sample prep technique be under these conditions if that approach relies on capture of target bacteria using antiflagellar antibodies? If cells respond to stress by forming filaments, how will this impact our ability to make effective use of filters with pore sizes targeted toward capture of smaller cells? Could environmental conditions induce physiological responses that impact microbial surface properties such as hydrophobicity and surface charge? If so, how would such changes impact our ability to extract these cells using ionic solvents, two-phase aqueous polymer solutions or adsorptive processes such as cationic beads or hydroxyapatite? In the past, the practice of relying solely on lab strains grown under standard conditions for development and validation of detection methods was considered both a necessity and a liability; standard conditions are needed to ensure experimental and interlaboratory reproducibility, but questions remain regarding how these methods may perform on bacteria living under “natural” or nonstandard conditions. We face an identical dilemma in the development of sample prep approaches, necessitating the evaluation of new or established methods using cells whose physiological condition more closely resembles that of naturally occurring contaminants, as opposed to happy, healthy and domesticated lab strains.
This article was intended to provide a broad overview of concepts and concerns regarding the burgeoning field of pre-analytical sample preparation in food microbiology. Although space precludes a comprehensive review of the topic, hopefully readers will come away with some new information or food for thought regarding this topic that is so fundamentally important to our work as food safety professionals.
Byron Brehm-Stecher, Ph.D., heads the Brehm-Stecher Rapid Microbial Detection and Control Laboratory in the department of food science and human nutrition at Iowa State University.
References
1. Jay, J. 2005. Part III: Microorganisms in Foods, 7th ed. (New York: Springer).
2. McEntire, J et al. 2014. “The Public Health Value of Reducing Salmonella Levels in Raw Meat and Poultry.” Food Prot Trends 34:386–392.
3. Grieg, JD et al. 2010. “Infective Doses and Pathogen Carriage,” Food Safety Education Conference, Atlanta, GA.
4. ICMSF. 1986. Microorganisms in Foods 2, Sampling for Microbiological Analysis: Principles and Specific Applications, 2nd ed. (Toronto: University of Toronto Press).
5. Brehm-Stecher, B et al. 2009. “Sample Preparation: The Forgotten Beginning.” J Food Prot 72:1774–1789.
6. www.gpo.gov/fdsys/pkg/FR-2014-09-24/pdf/2014-22682.pdf.
7. Cormier, J et al. 2014. “Concentration of Enteric Virus Indicator from Seawater Using Granular Activated Carbon.” J Virol Methods 196:212–218.
8. Johnston, LM et al. 2005. “A Simple Method for the Direct Detection of Salmonella and Escherichia coli O157:H7 from Raw Alfalfa Sprouts and Spent Irrigation Water using PCR.” J Food Prot 68:2256–2263.
9. Agoston, R et al. 2009. “Rapid Concentration of Bacillus and Clostridium Spores from Large Volumes of Milk, using Continuous Flow Centrifugation.” J Food Prot 72:666–668.
10. Mayrl, E et al. 2009. “Broad Range Evaluation of the Matrix Solubilization (Matrix Lysis) Strategy for Direct Enumeration of Foodborne Pathogens by Nucleic Acids Technologies.” J Food Prot 72:1225–1233.
11. Vibbert, HB et al. 2015. “Accelerating Sample Preparation through Enzyme-Assisted Microfiltration of Salmonella in Chicken Extract.” Biotechnol Prog 31:1551–1562.
12. Mester, P et al. 2010. “Use of Ionic Liquid-Based Extraction for Recovery of Salmonella Typhimurium and Listeria monocytogenes from Food Matrices.” J Food Prot 73:680–687.
13. Seo, KH et al. 2003. “Simple and Rapid Methods for Detecting Salmonella Enteritidis in Raw Eggs.” Int J Food Microbiol 87:139–144.
14. Gunasekera, TS et al. 2000. “A Flow Cytometry Method for Rapid Detection and Enumeration of Total Bacteria in Milk.” Appl Environ Microbiol 66:1228–1232.
15. Pahuski, EE et al. Methods and kits for separation, concentration and analysis of cells. U.S. Patent 5700645 filed June 7, 1995, issued December 23, 1997.
16. Kevan, PG and JT Trevors. 2001. “Formula Thinking, Formula Science and Kits.” J Microbiol Methods 46:85–86.
17. Dickens, JA et al. 1986. “Evaluation of a Mechanical Shaker for Microbiological Rinse Sampling of Turkey Carcasses.” Poultry Sci 65:1100–1102.
18. Stocker, BA and JC Campbell. 1959. “The Effect of Non-Lethal Deflagellation on Bacterial Motility and Observations on Flagellar Regeneration.” J Gen Microbiol 20:670–685.
19. Shapiro, SK and WB Sarles. 1949. “Microorganisms in the Intestinal Tract of Normal Chickens.” J Bacteriol 58:531–544.
20. Bai, J et al. 2013. “Extraction of DNA from Orange Juice, and Detection of Bacterium Candidatus Liberibacter asiaticus by Real-Time PCR.” J Agric Food Chem 61:9339–9346.
21. Brehm-Stecher, B. 2014. “Sample Collection and Preparation for Sensor-Based Detection of Pathogens in Food,” in High Throughput Screening for Food Safety Assessment, eds. Bhunia, AK, CR Taitt and MS Kim (Cambridge, UK: Woodhead Publishing), 11–22.
22. Weimer, BC et al. 2001. “Solid-Phase Capture of Proteins, Spores, and Bacteria.” Appl Environ Microbiol 67:1300–1307.
23. Magaña, S et al. 2013. “Automated Dead-End Ultrafiltration for Concentration and Recovery of Total Coliform Bacteria and Laboratory-Spiked Escherichia coli O157:H7 from 50-Liter Produce Washes to Enhance Detection by an Electrochemiluminescence Immunoassay.” J Food Prot 76:1152–1160.
24. Hedman, J et al. 2009. “Improved Forensic DNA Analysis through the Use of Alternative DNA Polymerases and Statistical Modeling of DNA Profiles.” Biotechniques 47:951–958.
25. King, C et al. 2009. “A Quantitative Approach to Detect and Overcome PCR Inhibition in Ancient DNA Extracts. Biotechniques 47:941–949.
26. Foley, SL et al. 2009. “Molecular Typing Methodologies for Microbial Source Tracking and Epidemiological Investigations of Gram-Negative Bacterial Foodborne Pathogens.” Infect Genet Evol 9:430–440.
27. d’Abbadie, M et al. 2007. “Molecular Breeding of Polymerases for Amplification of Ancient DNA.” Nat Biotechnol 25:939–943.
28. McDonald, JP et al. 2006. “Novel Thermostable Y-Family Polymerases: Applications for the PCR Amplification of Damaged or Ancient DNAs.” Nucleic Acids Res 34:1102–1111.
Pre-Analytical Sample Preparation in Food Microbiology: An Overview