Most recorded foodborne outbreaks are caused by raw and unprocessed or minimally processed foods prepared and consumed at home, in street vending, catering establishments or various types of institutions. As illustrated in several reports and publications, industrially manufactured foods have, however, also been involved in such outbreaks.
Surveys performed over the last 20–25 years have shown that up to 25% of these outbreaks could be traced back to recontamination during preparation and handling. In the case of industrially manufactured products, factors such as cross-contamination from contaminated processing environments and/or food contact surfaces play important roles. However, raw materials or ingredients added after a kill step have also been identified as sources of contamination on several occasions. With respect to Salmonella, for example, published case studies range from cocoa powder used to prepare different chocolate-based products[1] and paprika used to sprinkle onto potato chips[2] to the very recent major issues related to contaminated peanut butter[3] used as a raw material in numerous other products. Similar examples exist for other pathogens but are not listed here.
Such raw materials and ingredients, usually called dry mix ingredients (DMIs), are essential in contributing to the safety of the finished products and thus require particular attention in their selection and purchase. Wet mix ingredients (WMIs) are subjected to a kill step, usually heat treatment, and are therefore considered less critical. These issues will be discussed in relation to food safety management systems as well as to microbiological specifications that are used in contracts between raw material suppliers and food manufacturers. The discussion will focus initially on the evolution of food safety management concepts and principles in the last decade and then review the role of microbiological criteria within this framework, followed by some practical considerations.
Food Safety Management Principles
 Over the last 10–15 years, the approach to microbiological food safety control defined by public health authorities (PHAs) has evolved due to the development and implementation of the Microbiological Risk Analysis as defined by Codex Alimentarius.[4] This process is a tool for PHAs to define the food safety control measures in a more structured, open and transparent way than in the past. During this period, new terminology was developed and introduced, allowing better descriptions of targeted public health goals. These elements are illustrated in Figure 1 and discussed below.
Over the last 10–15 years, the approach to microbiological food safety control defined by public health authorities (PHAs) has evolved due to the development and implementation of the Microbiological Risk Analysis as defined by Codex Alimentarius.[4] This process is a tool for PHAs to define the food safety control measures in a more structured, open and transparent way than in the past. During this period, new terminology was developed and introduced, allowing better descriptions of targeted public health goals. These elements are illustrated in Figure 1 and discussed below.
The appropriate level of protection (ALOP) has been defined as the level of protection deemed appropriate by a PHA to establish a sanitary or phytosanitary measure to protect human, animal or plant life or health within its territory. Such a level is usually expressed as the number of cases/100,000 people/annum and must be translated into more tangible limits to define the stringency of the hygiene measures to control hazards. This is done first by defining a Food Safety Objective (FSO) corresponding to the maximum frequency and/or concentration of a microbiological hazard in a food at the time of consumption.
The FSO, as defined by PHAs, can be considered the “final target or objective” for the manufacturer and is achieved through the implementation of appropriate hygiene control measures during the manufacture of food products. In order to establish the link or interface between the governmental objective and the food safety management during manufacturing, new elements have been defined over the last few years.
The first element is the Performance Objective (PO), which is equivalent to the FSO in the sense that it also defines a maximum frequency and/or concentration of a microbial hazard in a food. However, contrary to an FSO, it is specified for one or more steps in the food chain before the time of consumption. A PO can therefore be considered an intermediate step in achieving the FSO and can be determined by either PHAs or operational food safety managers. Either single or multiple POs, depending on the complexity of the food chain, will therefore contribute to achieving the established FSO and ultimately the ALOP.
Depending on the food chain and the conditions influencing the decrease or increase of a hazard, such as a kill step(s) or the possibilities for growth, the PO at a specific step of the food chain will be higher or lower than the established FSO. Under certain circumstances, such as dry products not supporting any growth and manufactured by mixing DMIs without any further kill step, the PO is equivalent to the FSO at the level of both the raw material suppliers and the manufacturer of the final product.
Both FSOs and POs are used to help determine the required control measures within the food chain. These control measures are nothing more than the traditional and well-established generic control measures, such as Good Agricultural Practices, Good Manufacturing Practices and Good Hygiene Practices (GHPs) as well as Hazard Analysis and Critical Control Points (HACCP) or similar systems to address specific microbiological hazards. While the control measures remain the same, the main difference from their past applications is an increased focus on the quantification of their effects.
The effect of a single control measure or a combination of several of them is characterized by the Performance Criterion (PC). The PC relates to the initial level of a pathogen, for example, in a raw material, and is used by food safety managers to define the most appropriate control measure (or combination thereof) to determine the required reduction in order to achieve the desired target. An example of such a PC is the 12-log reduction of Clostridium botulinum in canned, low-acid foods.
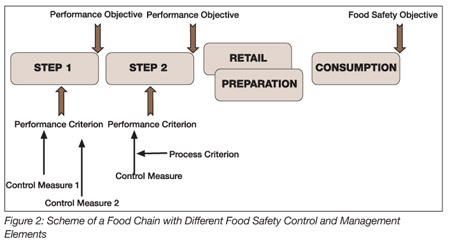 The different elements outlined above are summarized in Figure 2, which illustrates that production of foods complying with the hazard concentration tolerated at the moment of consumption is a shared responsibility for all stakeholders in the food chain. Starting with a defined FSO, one can, by moving upstream along the different steps of the food chain, evaluate the impact of, for example, the preparation conditions, the distribution and intermediate storage conditions, the manufacture of the food product, the manufacture of individual ingredients and the primary production of the food product. By doing so and taking into account the factors and conditions affecting the fate of relevant pathogens, it becomes possible to determine the needs and requirements at each step of the chain to ensure the safety of the final product.
The different elements outlined above are summarized in Figure 2, which illustrates that production of foods complying with the hazard concentration tolerated at the moment of consumption is a shared responsibility for all stakeholders in the food chain. Starting with a defined FSO, one can, by moving upstream along the different steps of the food chain, evaluate the impact of, for example, the preparation conditions, the distribution and intermediate storage conditions, the manufacture of the food product, the manufacture of individual ingredients and the primary production of the food product. By doing so and taking into account the factors and conditions affecting the fate of relevant pathogens, it becomes possible to determine the needs and requirements at each step of the chain to ensure the safety of the final product.
This established objective will therefore have a direct impact on the microbiological requirements for raw materials that is determined by the way they are further processed. These requirements will shape the interface between the suppliers and users of raw materials and ingredients.
Microbiological Criteria
How do microbiological criteria fit into this system? In fact, they represent one form of acceptance criteria for foods, and ideally, they should be based upon FSOs or POs. They define, according to Codex Alimentarius,[5] the acceptability of a product or a food lot, based on the absence or presence or number of microorganisms or quantity of their toxins/metabolites per unit of mass, volume, area or lot of the product. They can, in principle, be applied after different steps of the food chain, such as raw material, finished products at the factory or at the point of consumption.
 Although FSOs and POs are defined as the maximum frequency and/or concentration of a microbial hazard in a food and seem therefore similar to a microbiological specification used in contracts between two commercial partners, they are in fact quite different in function and content as summarized in Table 1.[6]
Although FSOs and POs are defined as the maximum frequency and/or concentration of a microbial hazard in a food and seem therefore similar to a microbiological specification used in contracts between two commercial partners, they are in fact quite different in function and content as summarized in Table 1.[6]
Due to their statistical limitations, microbiological criteria by no means represent a tool to ensure the safety of the material in question.
Contracts between commercial partners are thus not established based on FSOs or POs but based on microbiological criteria. It is therefore important to understand the meaning of such criteria or specifications (usual terminology in the case of raw materials) as well as some of their underlying considerations.
The International Commission on Microbiological Specifications for Foods (ICMSF) began providing guidance on the development of sampling plans and microbiological criteria for foods in international trade about 30 years ago. The result of these activities is reflected in publications like Microorganisms in Foods 2: Sampling for Microbiological Analysis: Principles and Specific Applications.[7] In this book, the ICMSF introduced concepts of probability and developed a scheme for the selection of cases and attributes plans in order to establish microbiological criteria for food lot acceptance. These plans take into account the conditions under which the product is expected to be handled and consumed. These conditions will cause no changes in the relevant organisms (e.g., in shelf-stable products), lead to an increase if there is an opportunity for growth or lead to a decrease if a kill step (e.g., cooking) is applied just before consumption. Another element considered is related to the degree of concern relative to a microorganism, that is, a food utility/hygiene indicator and a mild hazard up to a severe hazard. Using this approach, 15 cases were found to require an increased stringency for sampling plans applied for lot acceptance. The strictest requirements (absence in 60 × 25 g) have been recommended for Salmonella intended for the most sensitive population of foods, such as a infant formula reconstituted with water, where conditions may be conducive to increased risk if product is mishandled, allowing for growth.
The concepts outlined by the ICMSF have also been the basis for the elaboration of the Codex Alimentarius document Principles for the Establishment and Application of Microbiological Criteria for Foods.[5] This document is still a useful basis for the establishment of microbiological criteria. It is currently undergoing revision to take into account the current approaches in food safety management as well as the current knowledge on statistical performances of sampling plans.
According to these Principles, microbiological criteria should only be established when there is a definite need and where an application is practical. They should be established in a transparent way to ensure fair trade and should undergo regular revision in order to take into account changes in the scientific knowledge, in processing technologies as well as the occurrence of emerging pathogens. Irrespective of the type of criterion, the elements outlined in Codex Alimentarius and summarized below represent a suitable basis for their establishment.
• Evidence of actual or potential hazards to health
• The microbiological status of the raw materials
• Effects of processing on the microbiological content of a food
• The likelihood and consequences of microbial contamination and/or growth during subsequent handling, storage and use
• The intended use of the food
• Category(s) of consumers concerned
• Cost-benefit ratio associated with the application of the particular criterion
• The need to inform stakeholders along the entire food chain
With respect to their content, microbiological criteria consist of different elements:
• The microorganism of concern (and/or their toxins) and the reason for that concern
• Where appropriate indicators can be included
• Appropriate microbiological limits defined for specific point(s) of the food chain
• The number of analytical units that should conform to the established limits
• A sampling plan defining the number of samples taken, methods of sampling and handling as well as the size of the analytical unit
• The analytical methods of detection and/or quantification
While Codex Alimentarius only refers to microbiological criteria, it is commonly accepted that they can be differentiated into different types depending on the context in which they are used. However, the basic principles for their establishment are the same:
• Microbiological Standard – a mandatory criterion that is included in a regulatory document
• Microbiological Guideline – an advisory criterion used to inform food manufacturers on levels that can be achieved when applying best practices
• Microbiological Specification – a part of a commercial agreement between a buyer and a supplier
In the case of food processors, these standards represent the target to be achieved and can be used to validate and/or verify the efficacy of their preventive measures, that is, GHPs and HACCP, and to confirm compliance with existing regulations.
Two general types of microbiological testing and sampling plans are used to make decisions concerning the safety and quality of foods. On one side, there are attributes plans evaluating qualitative (i.e., presence/absence) or grouped quantitative data (e.g., < 10 or < 100 colony-forming units/g) and, on the other hand, variable plans evaluating non-grouped quantitative data.
Two-class attributes sampling plans are usually applied for pathogens and the decision to accept or reject a lot tested is defined by two parameters. The first is n, the number of independent and random units drawn from the lot. The second is c, the maximum allowable number of samples providing an unsatisfactory result, that is, the presence of the relevant organism. In the case of pathogens, c = 0 is usually applied, for example, n = 60, c = 0, m = 0 (in 25 g) in the case of Salmonella. In the case of grouped quantitative data, m is defined, a limit separating acceptable from unacceptable quality; n = 5, c = 0, m = 100 is an example of plans applied for mild pathogens such as Staphylococcus aureus in certain foods.
Three-class plans are used as an alternative to two-class plans where a subdivision into three classes is possible since a certain proportion of product with a marginal deviation is still acceptable. In such plans, the limit m separates good from marginally acceptable product and an additional limit M is defined to further distinguish unacceptable product. An example of a three-class plan for Enterobacteriaceae, a widely used hygiene indicator, is c = 5, n = 2, m = 10 and M =100, where two samples yielding a marginal result is acceptable, while three or even a single result exceeding M would lead to the rejection of the lot.
The sampling plans should be seen as a tool to assist in the acceptance/rejection process whether they are applied to raw materials or finished products at different steps of the food chain. Their design, that is, the definition of n, c, m and M, will depend on the purpose and will determine the stringency of the requirements. While the values for m and M (if used) provide a rather “visible” indication of stringency, n and c are also important in expressing stringency: the higher the n and the lower the c (for a given n), the more stringent the requirement. These parameters not only describe stringency but also performance or ability of a given sampling plan to detect the targeted microorganism, usually assuming a normal distribution in the food product, present at a certain microbial concentration. The relation of the performance of such sampling plans to a specific concentration to determine the proportion of defective samples in a lot of food product has been discussed previously.[8,9] Based on these premises, it has been shown that it is possible to calculate probabilities of acceptance (or rejection) of lots and relate them to a mean concentration of organisms in the lot sampled.
Considering the example of the sampling plan for Salmonella described above, this would provide values for the performance expressed as geometric mean concentration [e.g., the amount (in g) containing one cell] at which the defined sampling plan will reject a lot with a 95% confidence (see Table 2). These concentrations are calculated for different distributions within the lot, as expressed by the standard deviation (?) of the log-normal distribution: the larger this value, the wider the distribution. Such a distribution is considered the reflection of the level and effectiveness of control measures during processing.
 As shown by the values in Table 2, the mean concentrations do not correspond to one cell in 1,500 (i.e., to 60 × 25 g) as frequently and erroneously assumed; this “1 in 1,500 g” would rather correspond to a limit of detection.
As shown by the values in Table 2, the mean concentrations do not correspond to one cell in 1,500 (i.e., to 60 × 25 g) as frequently and erroneously assumed; this “1 in 1,500 g” would rather correspond to a limit of detection.
Manufacturers are certainly interested in achieving a mean value as low as possible with a distribution as tight as feasible, allowing consistent safety of products over time, thus in line with the elements characterizing a FSO as in Table 1.
A full description of the statistical basis and operation of such sampling plans has been described previously,6 and further calculations and examples for different sampling plans will become available in 2011.[10] In addition, Excel spreadsheets that allow this type of calculation have been posted on the website of the Commission at www.icmsf.iit.edu/main/home.html. Similar tools are being developed by other organizations, such as the Food and Agriculture Organization and World Health Organization, and will be available soon.
The ICMSF has been exploring the relationship between microbiological criteria and an FSO or a PO.[11] For further details, the reader is referred to this publication, which provides information on the data that are necessary and the types of decisions that must be made, to develop meaningful sampling plans and ensure that microbiological criteria based on within-lot microbiological testing are being used appropriately.
Practical Considerations
The understanding of the relationships between different elements of the food safety management systems as well as of underlying principles is certainly evolving. The following section is by no means an exhaustive discussion of questions (and debates) arising in discussions around microbiological specifications. The two examples below are only illustrations of the type of questions that may occur between providers and users of raw materials. However, in the context of relationships between suppliers of raw materials and users, more practical questions are often raised on the establishment and application of microbiological specifications.
With respect to the establishment of such specifications, questions are usually related to the stringency of requirements and their justification. Generic specifications, such as “absence of Salmonella” or small number of samples, are then opposed to stringent requirements of n = 60. In other cases, specifications with varying stringencies originating from different users are compared, and difficulties in understanding the different requirements may arise.
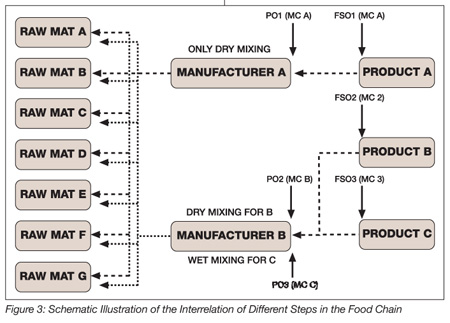 In such situations, it should be understood by all parties that the requirements expressed by PHAs, whether as an FSO, PO or microbiological criteria, represent the final objective to be achieved. These requirements must be traced back to the preceding steps of the food chain. Situations can become complex as schematically illustrated in Figure 3.
In such situations, it should be understood by all parties that the requirements expressed by PHAs, whether as an FSO, PO or microbiological criteria, represent the final objective to be achieved. These requirements must be traced back to the preceding steps of the food chain. Situations can become complex as schematically illustrated in Figure 3.
If, for example, three different products with different microbiological requirements, whether expressed as an FSO or as a microbiological requirement, were considered, then this would need to be translated upstream in a different manner. For this example, it is assumed that no change in the microbiological flora is occurring during distribution to the consumer. For manufacturer A, manufacturing product X through the application of dry mixing technologies, that is, without an additional kill step, would mean that the PO1 would need to be equivalent to the established FSO1, hence the microbiological specifications would need to be equivalent and therefore be reflected in the microbiological specification of the raw materials.
In the case of manufacturer B manufacturing products Y and Z with different technologies (e.g., wet mixing including a kill step), in principle, different POs or microbiological specifications would be derived. In the case of the dry mixing process, the requirements would also be equivalent and translated into microbiological specifications. In the case of the wet mixing process, the requirements of the finished products would of course need to be equivalent to those of the FSO/microbiological criteria. However, in this case, the requirements for the raw materials could be adjusted based on the effect of the heat treatment (control measure) and hence be less strict. This would mean that, in the case of DMI, the POs and microbiological criteria at the step of the raw material suppliers would need to be equivalent to those “downstream,” while the POs and microbiological criteria for WMI would be less stringent.
In the case of DMI, the interactions between the supplier and the user of the raw materials would certainly go beyond the simple establishment of microbiological specifications and the testing of samples. It would certainly also include, from the user, a review and assessment on the type and effectiveness of hygiene control measures as implemented by the supplier.
It is therefore clear that requirements such as microbiological specifications for several applications may differ. Milk powder used, for example, to manufacture chocolate, ice cream, infant formula, beverages for adults, ultra-high temperature products, etc. would need to fulfill different requirements. The way to manage and the degree of stringency of control measures as well as verification measures may therefore be different and must be appropriate for their specific purpose.
A frequently discussed question relates to the practical implementation of sampling plans, for example, n = 60, c = 0, m = 0 (in 25 g) and the actual (physical) sampling. How can the intrusive sampling, leading to a risk of contamination of the sampled container, be minimized? Can such a sampling plan be modified by taking fewer samples, for example, n = 10, c = 0, m = 0 (150 g)?
This type of question can certainly be discussed using calculation tools as presented above. Such tools will enable users to calculate alternative but equivalent sampling plans. However, care must be taken in considering the underlying assumptions related to the distributions of contamination and mean concentrations within the lot.
Another frequent question is related to dilution effects from additions of small quantities of ingredients. While the question is certainly legitimate, the application of a “corrective factor” to microbiological specifications to take into account an addition rate is usually not possible, as numerous elements would need to be taken into consideration for which the data are not often available. Such elements include a thorough understanding of the microbiological status of a raw material and the effectiveness of control measures influencing it, such as processing conditions, contamination from processing environment and food contact surfaces, the variability within individual or between subsequent lots, the distribution of contamination within a lot as well as other elements related to sampling, handling and testing.
Conclusions
Microbiological specifications used in the frame of commercial agreements fulfill a specific and important role in ensuring the safety of finished products. They should be based on sound principles and the role and limitations understood by the different stakeholders in the food chain.
Jean-Louis Cordier, Ph.D. is the food safety manager of Quality Manage-ment/Operations at Nestlé Nutrition. He received his Ph.D. at ETH Zürich, Institute for Microbiology and can be contacted at jean-louis.cordier@nestle.com.
References
1. Gästrin, B., Å. Kämpe, K. G. Nyström, B. Odén-Johanson, G. Wessel and B. Zetterberg. 1972. Salmonella durham-epidemi spridd genom kakaopulver. Läkartidningen 69:5335–8.
2. Lehmacher, A., J. Bockemühl and S. Aleksic. 1995. Nationwide outbreak of human salmonellosis in Germany due to contaminated paprika and paprika-powdered potato chips. Epidemiol Infect 115:501–11.
3. Centers for Disease Control and Prevention. 2009. Multistate outbreak of Salmonella infections associated with peanut butter and peanut butter-containing products – United States, 2008–2009. Morb Mortal Wkly Rep 58:1–6.
4. Codex Alimentarius Commission. 2007. Principles and guidelines for the conduct of microbiological risk management (MRM). CAC/GL-63. FAO: Rome.
5. Codex Alimentarius Commission 1997. Principles for the establishment and application of microbiological criteria for foods. CAC/GL-21. FAO: Rome.
6. International Commission on Microbiological Specifications for Foods. 2002. Microorganisms in Food 7. Microbiological testing in food safety management. Kluwer Academic-Plenum: NY.
7. International Commission on Microbiological Specifications for Foods. 1986. Microorganisms in Foods 2: Sampling for microbiological analysis: Principles and specific applications (2nd ed). University of Toronto Press: Toronto.
8. Hildebrandt G., L. Böhmer and S. Dahms. 1995. Three-class attributes plans in microbiological quality control: Contribution to the discussion. J Food Prot 58:784–90.
9. Legan, J.D., M. H. Vandeven, S. Dahms and M. B. Cole. 2001. Determining the concentration of microorganisms controlled by attributes sampling plans. Food Control 12:137–47.
10. International Commission on Microbiological Specifications for Foods. In Press. Microorganisms in food 8. The use of data for assessing process control and product acceptance.
11. Van Schothorst, M., M. H. Zwietering, T. Ross, R. L. Buchanan, M. B. Cole and International Commission on Microbiological Specifications for Foods. 2009. Relating microbiological criteria to food safety objectives and performance objectives. Food Control 20:967–79.
Customer-Supplier Relationships: Microbiological Specifications for Raw Materials and Ingredients