Ensuring that the materials which we put into contact with foods will not contaminate the foods has been the subject of a significant effort by regulators, particularly over the last 30–40 years.
The regulations developed by the U.S. Food and Drug Administration (FDA) cover nearly all classes of materials that are commonly used in contact with food, ranging from plastics, through coating materials, certain adhesives, paper and many other types of food contact material. These regulations are laid out in great detail in FDA 21 CFR Parts 170–199, and for many material types, these regulations provide the detailed safety rules to show how a food contact material can be placed safely and legally on the U.S. market. However, new food contact substances are continually being developed and existing food contact substances can find new uses which are not covered by existing approvals. FDA has several options that can be used in these cases where formal approval is required.
In this article, we aim to review the options for formal approval through FDA of new food contact substances or applications and describe how the level of migration of the substance into foods and the subsequent calculation of a dietary exposure estimate leads to the selection of an appropriate regulatory pathway.
Regulatory Options for Food Contact Materials
In the U.S., three principal options exist to gain formal FDA clearance for new food contact substances, which are not already regulated under a relevant part of the 21 CFR:
• Submission of a Food Contact Notification (FCN)
• Submission of a Food Additive Petition (FAP)
• Submission of a Threshold of Regulation Exemption (ToR)
The starting point in each of these routes is to determine the level of the food contact substance that migrates into foodstuffs. The FDA has described the protocols of how to carry this out very extensively in the Chemistry Guidelines of 2007. In essence, it is possible to assess migration in one of three ways:
• By measurement in a migration test, or
• By calculation assuming 100% migration of the substance, or
• By using a more sophisticated model based on diffusion physics.
Each of these approaches has its own strengths and weaknesses. Although the measurement of migration is the most expensive of these options, it will lead to the lowest estimate of the level of the substance which passes into foods. As we will see later, this will lead to a lower estimate of dietary exposure and can open up more regulatory options and also reduce the need for expensive toxicological tests.
FDA Migration Tests
When carrying out FDA migration tests, it is important to start with representative samples containing the food contact substance used at its maximum envisaged rate in a representative material type. For example, when approaching a plastics additive that may be used in polyethylene, the test pieces for the migration tests would be made from polyethylene and would contain the additive at its highest envisaged level. The extraction tests are then carried out by exposing the test pieces to food simulating solvents under conditions of mild agitation. To assist the petitioner, the FDA has clearly defined several different options for food simulating solvents including 10 percent ethanol, 50 percent ethanol and vegetable oil. The choice of which simulant is used for these tests depends on which foods the substance is likely to find itself in contact with.
Additionally, when planning the extraction test, there are choices to make for combinations of exposure times and temperatures. These are described in protocols A–H that cover a range of scenarios including high temperature heat sterilisation, through ambient usage to frozen storage, with special protocols designed for substances which may be subject to irradiation and also those used in cooking.
Once the level of the food contact substance migrating into food simulants has been determined, the next key step is to calculate the level of the substance in the consumer’s diet using the FDA exposure model.
FDA Exposure Model
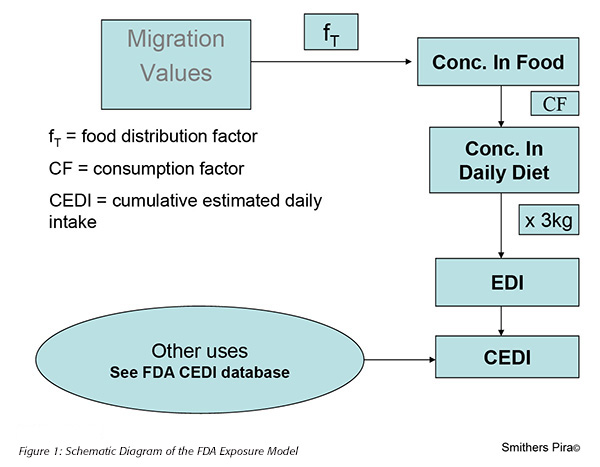 In contrast to the EU where dietary exposure has not been effectively reflected in the regulatory assessment of new food contact substances, FDA has long operated a fairly straightforward and pragmatic approach to the estimation of dietary exposure. This model is based on three key elements (Figure 1):
In contrast to the EU where dietary exposure has not been effectively reflected in the regulatory assessment of new food contact substances, FDA has long operated a fairly straightforward and pragmatic approach to the estimation of dietary exposure. This model is based on three key elements (Figure 1):
1. The migration levels of the food contact substance measured in the migration study
2. The proportion of each type of food to which individual packaging materials might be exposed
3. The proportion of each type of packaging material to which the consumer would be exposed through daily diet
Starting with migration values into the different food simulating solvents, FDA model applies a food distribution factor, which describes the proportion of acidic, fatty and alcoholic foods to which an individual food contact material might be exposed. The model then applies a consumption factor that describes the percentage of food in the daily diet which has been packed in each type of food contact material. There is then an assumption that the average consumer is exposed to 3 kg of packed foods or beverages per day made up of 1 kg of food and 2 kg of beverages. Using these figures it is then possible to calculate an estimated dietary intake (EDI) for the food contact substance. Infants aged 0 to 6 months have a higher consumption of food when compared to their body weight and therefore a different exposure calculation is applied for food contact substances which will be used with infant formula products.
For many petitions that FDA is called on to review, the food contact substance is not totally new, but has previously been assessed for use in a different food contact application. The consumer may, therefore, already have been exposed to the substance in their daily diet. For this reason, the FDA maintains a database of cumulative estimated dietary intake (CEDI) figures for a range of existing food contact substances. When approaching one of these substances, the EDI for the substance in its new application needs to be added to the CEDI for all the previous applications to arrive at a new estimate of CEDI.
Once the incremental EDI and the CEDI have been determined, these drive the choice of the route through which the petitioner would choose to seek FDA approval:
• If the CEDI is greater than 1 part per million (ppm), the FDA requires a full food contact petition for the substance.
• If CEDI is lower than 1 ppm in the diet, the principal route for seeking approval is through a process known as the food contact notification (FCN) system.
• Where the incremental EDI is lower than 0.5 ppb, there is an additional option for approval through the ToR.
In this way, we can see that the migration values and FDA’s dietary exposure model drive the choice of regulatory options: the lower the calculated dietary intake, the greater the number of options available to petition FDA. We will now describe the strengths and weaknesses of these different options.
The Food Additive Petition (FAP) Process
Until 1999 when the FCN system was fully implemented, the main option to seek regulatory approval from FDA for a new food contact substance was through a full food additive petition. The system was largely unpopular with industry because it was usually quite slow with a typical submission taking 2 to 4 years for FDA to grant an approval. This was considered a barrier to innovation. Additionally, if FDA considered a new food contact substance was suitable for use in food contact applications, the approval would be acknowledge with a new regulation. This is the process which led to the large volume of approvals detailed in 21 CFR parts 170–199. The upshot of this approach was that a business operator could petition for the approval of a new substance at some considerable expense and then as soon as approval was granted, competitors could use that regulation at little or no expense to justify use of their own products. Again, this was thought to be a disincentive to innovation.
The FAP process is still open, but in practice, FAPs are only submitted when the CEDI is greater than 1 ppm, in which case FDA permits no other options, or when the expected number of applications for a new food contact substance exceeds those which could be covered within a reasonable number of FCNs. It is also a relatively expensive process requiring extensive toxicological data.
The FCN System
The most commonly used route to obtain approval for a new food contact substance at the present date is through the submission of a FCN. In principal, this contains many of the elements of the FAP process, including an assessment of CEDI and a toxicological safety assessment. However, it is important to remember that under the FCN process there has been a transfer of workload from FDA to the petitioner. Under the FCN process, the petitioner is responsible for ensuring that the migration study and the toxicological assessment have been carried out and described correctly. The role of FDA is then to confirm that this has been carried out satisfactorily.
The FCN process has two key advantages over the FAP process:
1. First, it leads to a proprietary approval that can only be used by the company that submitted the application. If competitors wish to place an identical product on the market, they must seek their own FCN, even where the application is identical.
2. The FCN process is also relatively quick. Once the notification has been submitted to the FDA, it has 120 days to accept or reject the submission. If this deadline is not met, the product is automatically considered legal.
Where the CEDI has been determined to be between 0.5 ppb and 1 ppm, FDA has some very clear Toxicology Guidelines. Extensive discussion of this guidance is beyond the scope of the present article, but there is a general principle that the amount of information the FDA requires on an individual food contact substance depends on the CEDI of that substance. Low exposure food contact substances with a dietary concentration between 0.5 and 50 ppb require only straightforward in vitro toxicology tests such as the Ames test and the mouse lymphoma thymidine kinase gene mutation assay. Food contact substances with a dietary concentration between 50 ppb and 1 ppm will require more extensive toxicological testing including studies in animals, which inevitably leads to a higher cost for these higher exposure chemicals.
The key outcome of the toxicological assessment is an acceptable dietary intake (ADI) for the food contact substance. In order for the FDA to approve the substance, they need to be convinced that the CEDI calculated from the migration study for the food contact substance is lower than the ADI indicated by the toxicological study. Likewise, the same process needs to be repeated for the major contaminants and degradation products in the food contact substance.
When submitting an FCN for a substance with a CEDI below 0.5 ppb, only a toxicology literature search is required as part of the FCN. This obviously gives rise to significant cost savings, but this is only viable for these very low exposure food contact substances.
One other important feature of the FCN process is that FDA is willing to provide a prenotification consultation meeting during which they will agree to carry out an initial review of an FCN dossier prior to its submission. This can either be carried out on FDA premises or remotely. During this meeting, FDA will identify any gaps so that the petitioner can address these prior to submission. This is clearly a win-win situation for industry and FDA and means that the process can run as smoothly and quickly as possible after the FCN has been submitted.
The ToR Process
Where a food contact substance has a very low incremental EDI (< 0.5 ppb), there is another approach which can be used to seek approval from the FDA through the ToR mechanism. In many ways, the level of toxicological information required for a ToR is identical to that required for an FCN for substances with a dietary exposure lower than 0.5 ppb. However, it is worth noting that approvals granted under the ToR process are non-proprietary and hence petitioners would normally prefer to opt for the FCN process unless a generic approach with a wide range of applications is the required outcome.
Non-Regulatory Approaches for Clearance of Food contact Substances
In addition to the formal regulatory options described in this article, there are a number of other options whereby a food contact substance can be used without seeking formal approval from FDA. These non-regulatory options are sometimes used when confidentiality is required.
One of the most commonly used routes is through the so called “no migration exemption.” If the new food contact substance is not detected in a migration study and provided that the method of analysis in the migration study is capable of measuring the substance at levels of either 10 ppb or 50 ppb of migration, depending on the nature of the substance and the application, it is possible to argue that the substance has not formally become a component of food under the definition of the Federal Food, Drugs & Cosmetics Act. In these circumstances, the substance can be argued to be exempt from FDA regulation, but some care and professional advice is usually required before reaching this conclusion.
Summary
In reviewing the various options available for formal clearance of a new food contact substance through FDA, each of the available mechanisms depends on a measure of the dietary exposure of the food contact substance. In general, the lower the dietary exposure of a substance, the greater the number of regulatory options that are available to the petitioner. However, the principal route that is currently used to petition FDA for approval of new food contact substances is the FCN system that has the twin advantages that it is rapid and that, if successful, results in a proprietary approval.
In addition, since the toxicological requirements increase with increasing exposure, so does the cost of submitting a petition. Intuitively, this makes perfect sense as it is quite rational that the regulator, industry and consumers would all wish to pay the most attention to the risk assessment of high exposure to food contact substances.
 Dr. Alistair Irvine manages the Food Packaging Safety Section within Smithers Pira and advises clients on the safety legislation which applies to food packaging in a wide range of different countries throughout the world.
Dr. Alistair Irvine manages the Food Packaging Safety Section within Smithers Pira and advises clients on the safety legislation which applies to food packaging in a wide range of different countries throughout the world.
 Ian Cooper leads a team of analytical chemists at Smithers Pira in research projects and to generate migration data for gaining EFSA/food contact clearances for new substances/materials and non-routine investigations, such as safety evaluation of recycled materials, nonintentionally added substances and printing inks.
Ian Cooper leads a team of analytical chemists at Smithers Pira in research projects and to generate migration data for gaining EFSA/food contact clearances for new substances/materials and non-routine investigations, such as safety evaluation of recycled materials, nonintentionally added substances and printing inks.
>




