The big issue in the food industry today is food safety; and microbial contamination is the number one enemy of the food supply. Chemical treatments with compounds such as peracetic acid are among the most effective methods for control of microbial contamination.
Peracetic acid has the chemical formula CH3C(O)OOH and is formed in an equilibrium mixture of acetic acid and hydrogen peroxide.
CH3COOH + H2O2 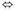 CH3CO3H + H2O
CH3CO3H + H2O
It is almost always used is water solution with acetic acid and vinegar, although the proportions of each component vary from one manufacturer’s blend to the next. Peracetic acid has a strong vinegar-like smell around 0.1 ppm but becomes significantly irritating above about 1 ppm. Peracetic acid is a very effective antimicrobial compound, even more effective than hydrogen peroxide.
In addition to being an effective against bacteria, any biocidal chemical that will be applied directly to food must not leave any harmful residues. Peracetic acid is very reactive and quickly decomposes to acetic acid (acid in vinegar), oxygen and water. This process is so complete that the Environmental Protection Agency (EPA) regulations exempt peracetic acid residues from tolerances in food products treated up to 100 ppm per application on fruits, vegetables, tree nuts, cereal grains, herbs and spices and with solutions of less than 500 ppm applied to equipment;[1] and U.S. Department of Agriculture regulations provide that peracetic acid may be directly applied to foods that are NOP certified as organic.[2] These features of good efficacy, essentially no toxic residues, easy application (in water solution) and relatively low cost have made peracetic acid an increasingly popular antimicrobial in many food and beverage-related industries. Peracetic acid blends have been approved by the U.S. Food and Drug Administration (FDA) approved as sanitizer on food contact surfaces (21 CFR 178.1010) and for direct food contact with fruits, vegetables (21 CFR 173.315) and meat, poultry and seafood (21 CFR 173.370).
Bottling and Beverage
Many bottling plants use peracetic acid as a sanitizer and disinfectant to reduce microbial contamination. Peracetic acid is also widely used as a sanitizer for the brewing industry.
One of the more recent trends has been toward completely eliminating microbial contamination with aseptic packaging whereby both the container and the bottle are sterilized. Aseptic packaging offers longer shelf life and reduced shipping costs and is used for milk products, low acid and high acid beverages.[3, 4] For example one large midwestern company produces dairy products aseptically using peracetic acid and can ship their product anywhere in the U.S. without the need to refrigerate, thus greatly reducing shipping costs. The items of course are refrigerated at the point of sale in the grocery store in order to meet customer expectations.
Fresh Produce
To reduce pathogens, most fresh vegetables are washed in an antimicrobial solution. The traditional standby has been chlorine (aqueous hypochlorite solution) and in recent years there has been an increasing switch to peracetic acid washes.[5, 6] Most fresh vegetables such as lettuce, spinach and fruit such as strawberries are washed after picking before shipment.[7] However, it has been recognized in some products such as strawberries that the fungus that causes the rot are already in the berry before harvest and that treating the strawberries several days before harvest increases shelf life.[8]
Meats, Poultry and Seafood
In the meat production industry, antimicrobials such as chlorine and peracetic acid have been shown to be very effective at reducing pathogen levels if applied directly to the meat,[9, 10] and in the U.S., they are now being used for direct intervention.[11]
Some research has found that chlorine/hypochlorite can react with fats and proteins to form various chlorinated compounds[12] some which are potentially carcinogenic.[13] Even treatment of vegetables such as bean sprouts and cabbages with sodium hypochlorite were found to produce chloroform (CHCl3) a known carcinogen.[14] However, FDA does not believe that carcinogens are formed to a significant extent at the concentrations used in food production and chlorine and hypochlorite are both approved for direct intervention in fresh produce, meat and poultry in the U.S.[15]
As a consequence, Russia banned imports of chicken from the U.S. Intervention using peracetic acid does not have this problem, and in June 2010, Russia lifted its ban on poultry imports to allow chicken treated with cetylpyridium chloride, peroxyacetic acid or hydrogen peroxide.[16] Similarly, the EU had also not imported U.S. chicken since 1997 because of its concerns about the use of chlorine washes for poultry. The European Food Safety Authority has said that the use of pathogen reduction treatments such as peracetic acid is safe, but the EU has yet to lift the ban in large part because chicken processing plants in the EU focus on tighter process controls to limit bacterial contamination rather than broad chemical intervention.[17] Processing chickens without chemical intervention is not an option for US poultry plants wanting to meet the U.S. Department of Agriculture’s requirements for pathogen reduction.[18]
Peracetic acid has been found effective for reducing microbial contamination of seafood and for keeping seafood fresher for longer[19] and has been approved for direct contact with fish by FDA.[20]
Occupational Exposures to Peracetic Acid
As discussed above, the two key strengths of peracetic acid are its effectiveness as a biocide and its high reactivity which causes it to quickly degrade to harmless byproducts. This high reactivity though presents a risk to workers using peracetic acid food processing environment and anecdotal reports indicate that some workers in have been adversely affected by exposure to peracetic acid and similar chemicals.[21, 22]
The vast majority of employers wants to keep their people safe and would do so even without the legal duty to provide a safe work environment imposed by the 1970 Occupational Safety and Health Act (OSHA). Peracetic acid has only recently become widely adopted and OSHA has not yet promulgated permissible exposure limits for peracetic acid. Until recently employers had to rely on their best judgment which often meant cranking up the ventilation until peracetic acid could not longer be smelt.
Some guidance was available from the EPA which issued Acute Exposure Guideline Levels (AEGLs) for peracetic acid. There are three levels of AEGL corresponding to the severity of symptoms of exposure.[23] These levels can be loosely translated as irritation (AEGL -1), disabling irritation or permanent injury (AEGL-2) and serious injury or death (AEGL-3). The 8 hour time-weighted average (TWA) AEGL.s for peracetic acid are AEGL 1: 0.52 mg/m3 (0.17 ppm), AEGL 2: 1.6 mg/m3 (0.51 ppm) and AEGL 3: 4.6 mg/m3 (1.3 ppm).[24] The AEGLs however are designed to assist emergency responders asses the risk to a person exposed to the chemical during an accident. The AEGLs are thus intended for a one time exposure and not for repetitive exposure as with an occupational exposure limit.
Recently (spring 2014[25]) however, the American Conference of Government Industrial Hygienists (ACGIH) issued a threshold limit value short-term exposure limit (STEL) for peracetic acid, calculated as a TWA over 15 minutes of 0.4 ppm. This STEL is the first occupational exposure limit peracetic acid and it gives employers a rational metric that they can use to ensure that their people are not over exposed to peracetic acid vapor.
A metric is great, but to be useful there must be a reliable means to measure the peracetic acid concentration. The large manufacturer aseptic dairy products discussed above had received several complaints from their workers using peracetic acid, and so they sought out a gas monitoring company that could produce a continuous monitor for peracetic acid that met their requirements.
The result of this collaboration was the ChemDAQ Steri-Trac® monitor for peracetic acid which continuously provides real time measurements of the peracetic acid concentrations. The monitor has a minimum detection limit of 0.04 ppm, resolution of 0.01 ppm and range 0 to 6 ppm so it can readily detect peracetic acid concentrations around the STEL. The dairy products manufacturer now has the ChemDAQ monitor installed on six aseptic production lines. The 18 peracetic acid monitors and six hydrogen peroxide monitors (hydrogen peroxide is used to sterilize bottle caps) are connected back to the ChemDAQ DAQ® computer which log data, provides for data collection, TWA calculations, graphing/reports and data export.
In addition to continuously monitoring the peracetic acid vapor concentrations, the ChemDAQ system also allows facilities to adjust their air handler to ensure that sufficient ventilation is employed to keep the peracetic acid concentrations within safe limits, but without wasting excess conditioned air. Some facilities perform the adjustments manually, whereas others use the in-built data export functions (SQL data base or Modbus®[26]) available from the ChemDAQ DAQ computer. The individual monitors also have two relay outputs that are activated whenever the peracetic acid concentration exceeds the user adjustable low- and high-alarm set points.
One of the difficulties that ChemDAQ faced in developing this monitor was how to calibrate it. Most gas and vapor monitors are calibrated using certified gas mixtures that come in compressed gas cylinders, but peracetic acid is too unstable to be provided in that form. ChemDAQ therefore generates their own peracetic test gas and analyzes it, and then calibrates the peracetic acid sensors. Field calibration of reactive gas sensors is often problematic, because the reactive gases can react with contaminants and is probably the number one service issue for the manufacturers of monitors for reactive gases. ChemDAQ solves this problem by providing a calibration service whereby freshly calibrated sensors are sent to customers, who swap out the expired sensors and return the latter to ChemDAQ to be refreshed. This process works very smoothly, and ChemDAQ supplies calibrated sensors around the world to Europe, Asia, Australia and the Americas.
Another challenge that the developers at ChemDAQ faced was cross-sensitivity to hydrogen peroxide. Both peracetic acid and hydrogen peroxide are strong oxidizing agents, and so there is some cross-sensitivity of hydrogen peroxide on the peracetic acid sensor. ChemDAQ developed a proprietary chemical filter that allows the peracetic acid to pass through but selectively removes essentially all of the hydrogen peroxide from the gas stream.
Summary
In summary, peracetic acid offers many benefits to food producers and processors from sanitization to sterilization. Peracetic acid is a very effective biocide but it quickly reacts in the environment to break down to harmless byproducts. Peracetic acid is finding application across the food spectrum from dairy to produce to meat processing and is being increasingly adopted. Its high reactivity makes it a potential hazard for workers using it; in the spring of 2014, the ACGIH issued a STEL TWA of 0.4 ppm. ChemDAQ also recently released a continuous monitor for peracetic acid that provides real-time readings with a range of 0 to 6 ppm and a minimum detection limit of 0.04 ppm, so well suited for detecting peracetic acid near the STEL. The ChemDAQ peracetic monitor helps employers continue to use peracetic acid safely.
References
1. www.federalregister.gov/articles/2000/12/01/00-30679/peroxyacetic-acid-exemption-from-the-requirement-of-a-tolerance.
2. USDA National Organic Program, certified organic (7CFR 205.601).
3. www.procomac.it/procomac/cmsdoc.nsf/WebDoc/ndkw73ehmt, retrieved 4/9/2014.
4. www.krones.com/en/products/filling-technology/29897.php, retrieved 4/9/2014
5. www.sciencedirect.com/science/article/pii/S0260877409002374
6. books.google.com/books?id=Xobj2N_gOxsC&pg=PA180&lpg=PA180&dq=peracetic+acid+strawberries&source=bl&ots=3ag35d1rmu&sig=XJ3zT2aUlG2NVpRWECTyGrEnBc0&hl=en&sa=X&ei=wBQ_U6CKMNOxsAS1woGQBw&ved=0CGYQ6AEwCQ#v=onepage&q=peracetic%20acid%20strawberries&f=false.
7. /lib3.dss.go.th/fulltext/Journal/Journal%20of%20food%20science/2000%20v.65/no.8/jfsv65n8p1376-1379ms20000337%5B1%5D.pdf.
8. hortsci.ashspublications.org/content/42/3/617.full.
9. www.citrobio921.com/docs/seafood.pdf.
10. www.ncbi.nlm.nih.gov/pubmed/18931192.
11. www.foodquality.com/details/article/1352287/Multiply_Your_Meat_and_Poultry_Attack.html?tzcheck=1.
12. www.ncbi.nlm.nih.gov/pmc/articles/PMC1474307/pdf/envhper00440-0256.pdf.
13. www.fda.gov/ohrms/dockets/dailys/04/july04/072904/04N-0258-emc00009-01.pdf.
14. www.jstage.jst.go.jp/article/shokueishi1960/33/3/33_3_267/_pdf
15. www.fda.gov/Food/FoodScienceResearch/SafePracticesforFoodProcesses/ucm091363.htm.
16. www.foodsafetynews.com/2010/06/russia-agrees-to-lift-ban-on-us-poultry-imports/#.Uz7_OahdWAg and www.fsvps.ru/fsvps/press/1516.html?_language=en.
17. Key language in the regulations reads, in part, “[f]ood business operators shall not use any substance other than potable water”—or, when otherwise permitted, “clean water—to remove surface contamination from products of animal origin,” unless use of another substance has specifically been approved.[ EC Regulation No. 853/2004, Article 3 (part).]
18. www.nationalchickencouncil.org/wp-content/uploads/2013/10/Bill-Roenigk-T-TIP-Hearing-of-the-Finance-Committee-U-S-Senate-3.pdf?utm_source=WhatCounts+Publicaster+Edition&utm_medium=email&utm_campaign=NCC%E2%80%99s+Roenigk+Expresses+Concerns+about+US-EU+Trade+Deal&utm_content=in+testimony+delivered+today.
19. www.envirotech.com/pdf/PAA%20for%20Seafood.pdf.
20. www.accessdata.fda.gov/scripts/fdcc/index.cfm?set=fcn&id=699.
21. www.huffingtonpost.com/food-integrity-campaign/usda-poultry-inspector-sp_b_3201436.html.
22. scienceblogs.com/thepumphandle/2013/02/19/a-food-safety-inspectors-warning-about-chemicals-and-bacteria-under-usdas-proposed-poultry-inspection-modernization/
23. www.epa.gov/oppt/aegl/pubs/define.htm.
24. www.epa.gov/oppt/aegl/pubs/results80.htm.
25. 2014 Guide of Occupational Exposure Values, Compiled by ACGIH
26. Modbus is a registered trademark of Schneider Electric USA, Inc.; USPTO Trademark registration number 73331239.
For more information, please visit www.chemdaq.com.




