For centuries, the benefits of enhancing meat flavor and texture with injected brines, marinades and phosphates has been well accepted as a way to add value to customers, while also improving marketability by improving color, texture and shelf life. The modern injection process typically consists of five main components: injector needles, brine or marinade mixing tanks, filters, heat exchanger and saddle tank, all connected in a recirculation loop with sanitary clean-in-place (CIP) piping.
The injection process is inherently susceptible to microbiological challenges in the brine/marinade and equipment components. Therefore, a thorough microbiological testing and sanitation protocol is important to achieve and maintain product quality and integrity.
Brine Dumping
Throughout the course of a processing day, the microbial levels of the injected solution increase as it is recirculated, until it reaches a predetermined level. At this point, the solution must be discarded or “dumped,” and a new batch of solution mixed before continuing production. While this procedure ensures that the injected solution is maintained at safe levels, it does not address the sanitation component of the injection process.
Sanitation of the Injection Loop Components
By its nature, the injection process allows the needles to transfer surface skin pathogens and spoilage bacteria into the muscle tissue. While proper cooking temperatures mitigate the food safety risks associated with this contamination, shelf life of fresh injected product can suffer dramatically as a result. Therefore, it is critical to adopt a thorough cleaning regimen for the injector and its components.
To clean the injectors, a common approach is to use a chlorinated alkaline cleaner recirculated through the system, followed by a water rinse. A vinegar solution is often used to soak the needles overnight. This helps unclog needles by loosening bone, cartilage and fat that has accumulated during the course of the day. The needles are then rinsed and blown out with compressed air.
However, the approach outlined above is often not sufficient to eliminate all spoilage organisms from the injection system.
The Problem: Biofilm
Biofilms are a complex matrix of bacteria, fungi and algae bound together in a sticky gel of polysaccharide and other organic contaminants attached to a surface. Organisms produce a protective, slime-like layer, in which they live, that anchors them firmly to a surface and provides a protective environment to grow and reproduce.[1] Biofilms generally form on any surface that is exposed to water or other liquids. Brine/marinade injection systems are particularly at risk to biofilm buildup due their nutrient enriched make-up. Pathogens in biofilm can be up to 1,000 times more resistant to traditional cleaners, sanitizers and disinfectants as compared to their free floating (planktonic) counterparts living in water based environments.[2]
Given these characteristics, biofilm becomes a natural foe in the injection process. Needles, tubing, CIP piping, heat exchangers and saddle tanks are at risk, and it is not economical to disassemble and hand scrub to remove biofilm. As such, an effective biocidal, biofilm removal technology becomes crucial.
PerQuat® Technology
Sterilex® PerQuat® technology contains synergistic components that lyse open the biofilm structure, kill pathogenic organisms, collapse the biofilm polymer matrix and physically lift the biofilm structure from surfaces.
In 2009, under a U.S. Department of Agriculture (USDA) National Research Initiative Grant, Judy Arnold of the USDA-Agricultural Research Service Poultry Microbiological Safety Research Unit in Athens, GA conducted a study with multiple compounds to compare their efficacy against resistant biofilm containing Listeria monocytogenes, a foodborne pathogen found in some ready-to-eat-meats. She published a paper entitled “Biofilms Have a New Foil” in which she reported:
“Results showed that the formulation (Sterilex) was 100 percent effective, providing total kill and more than 90 percent biofilm removal. This disinfectant is more effective than currently used disinfectants in reducing L. Monocytogenes biofilm growth, thus minimizing the risk of pathogenic contamination…”[3]
Injector Case Study
To validate the benefits of using PerQuat sanitation chemistries in injection systems, a major U.S. poultry processor conducted a shelf-life study in 2012 using Sterilex Ultra Disinfectant Cleaner & Sterilex Ultra Activator Solution (a liquid PerQuat solution) to treat an injector and components for approximately two weeks, nightly. Baseline samples were collected and compared against samples of raw dark poultry meat after injector treatment with the Sterilex product. Shelf life of meat was determined by looking at both microbiological metrics [aerobic plate counts (APC) and total coliform counts] as well as organoleptic observations. The results are shown below (Tables and Figures; see also sidebar).
Discussion
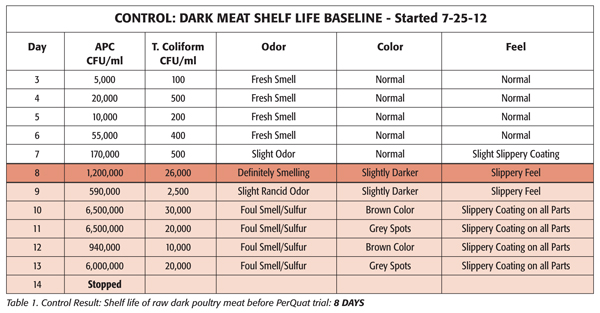 Micro Results
Micro Results
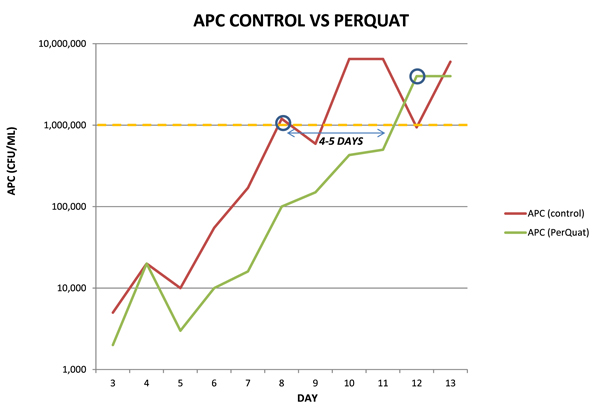
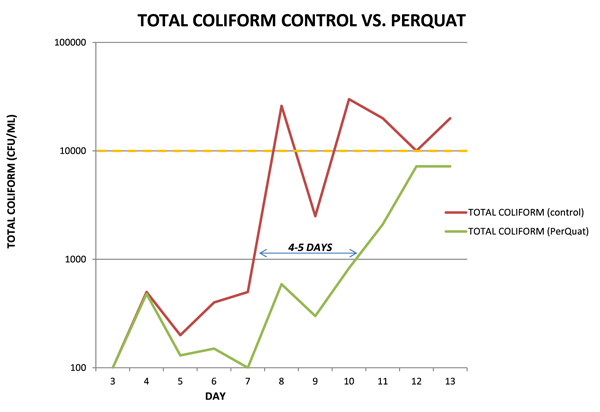
From days 3 through 12, dramatic reductions in APC counts and total coliform counts by day were observed. In general, microbiological thresholds were reached 4–5 days later after PerQuat treatment as compared to the control sample, leading to an increase in shelf life.
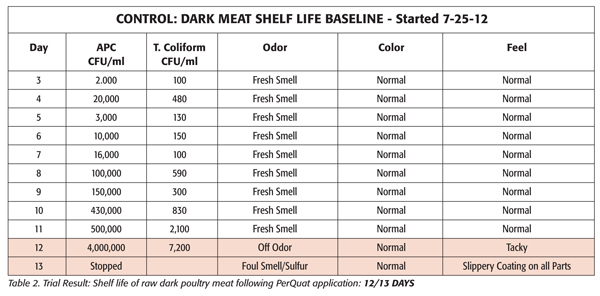 Organoleptic Results
Organoleptic Results
Organoleptic differences between the two sets of samples were even more dramatic. The pretreatment samples began to emit odors at day 7, while the post treatment samples remained fresh smelling until day twelve.
 The color of the control samples remained normal until day eight, at which time the meat began turning brown in color. Grey spotting was also observed until day 13 when the testing was stopped. There was no discoloration of the treated samples. Color remained normal from day 3 through day 13.
The color of the control samples remained normal until day eight, at which time the meat began turning brown in color. Grey spotting was also observed until day 13 when the testing was stopped. There was no discoloration of the treated samples. Color remained normal from day 3 through day 13.
The surface characteristics of the control samples only remained normal until day 7, at which time a slippery coating was observed on all parts. The feel of the treated samples remained normal until day 12.
Conclusions
As observed in this trial, treatment of the injector loop with PerQuat technology on a daily basis for 2 weeks resulted in a 4/5 day extension of shelf life for raw, dark poultry meat. Delays in microbiological thresholds as well as significant improvements in organoleptic properties were seen throughout the trial.
 Similar results may be observed in any plant with a brine injection process by following a similar injector treatment protocol.
Similar results may be observed in any plant with a brine injection process by following a similar injector treatment protocol.
References
1. Chmielewski, R.A.N. and J.F. Frank. 2003. Biofilm formation and control in food processing facilities. Comp Rev Food Sci Safety 2:22–30.
2. Jefferson, K.K., D.A. Goldmann and G.B. Pier. 2005. Use of confocal microscopy to analyze the rate of vancomycin penetration through Staphylococcus aureus biofilms. Antimicrob Agents Chemother 49(6):2467–2473.
3. Durham, S. 2009. Biofilms have a new foil. USDA: Agric Res Mag 59(9).
You may contact Sterilex Corporation directly for more information at 800.511.1659 or Sales@Sterilex.com.
Sidebar:
Case Study #1
Type of Needles Used: MEPSCO Ultracat 488
Type of Product Tested: Dark poultry meat (legs and thighs) in packaged bags
Control Treatment of Needles/Lines:
• Needles removed from the injection lines.
• Needles soaked in vinegar solution overnight for 6 hours (Heinz white vinegar undiluted at 5% acidity), then rinsed and blown out with compressed air in the morning.
• Chlorinated alkaline foam cleaner (1.8% active chlorine, 6.9% caustic) recirculated through entire CIP injection loop for 1 hour followed by a thorough water rinse.
Control Sample Shelf Life Observed: 8 days
Sterilex PerQuat Treatment of Needles/Lines:
• Needles remain in place connected to lines.
• Chlorinated caustic foam cleaner (1.8% active chlorine, 6.9% caustic) recirculated through entire CIP injection loop for 1 hour followed by a thorough water rinse.
• Sterilex Ultra Disinfectant Cleaner & Activator Solution NF added to brine tank at a 1:1:10 concentration and circulated through CIP injection loop , filters, heat exchanger, sanitary piping and needles for 1 hour. The brine mixing tank was filled to the 100-gallon mark, and 10 gallons Sterilex Ultra Disinfectant Cleaner NF and 10 gallons Sterilex Ultra Activator Solution were added.
• Lines and needles rinsed thoroughly.
• Needles soaked in vinegar solution overnight for 6 hours (same concentration as above), then rinsed and blown out with compressed air in the morning.
Frequency of Use of Sterilex PerQuat to Needles/Lines:
• Sterilex treatment used daily for 2 weeks for trial. Samples pulled following 2-week shock treatment.
• Ongoing, Sterilex PerQuat treatment applied 1–3× per week following initial shock treatment application.
Sterilex Sample Shelf Life Observed: 13 days
Increasing Shelf Life of Injected Meats




