With the signing by President Barack Obama of the Food Safety Modernization Act (FSMA) on January 4, 2011, the importation of safe and sanitary foods became one of the focal points of the government. Both the U.S. Food and Drug Administration (FDA) and the U.S. Department of Agriculture (USDA) are increasing inspections at points of entry and have received authorization to make importers responsible for the certification of manufacturing food plants in foreign countries. These very simple directives have implications not only for food safety but also for all the procedures that need to be in place to produce safe foods, normally known in the industry as Good Manufacturing Practices or GMPs.[1]
So how can foreign food manufacturers comply with U.S. regulations? First and most importantly is for these enterprises to know the regulations by which their products will be inspected and judged to allow their importation into U.S. markets. In this particular case, the response is obvious as compliance is the law of the land.
Secondly, but completely under the exporters’ control, is the manufacturing phase of such food products. In this case, the local food manufacturing entity might believe and think that it has complete liberty, judgment and jurisdiction as to how, when and what goes into the manufacturing process, but if the food product must comply with U.S. food laws and regulations, that “liberty” is limited by the way in which the produced food item complies with the specifications for sale of such products in U.S. markets.
The following is one definition of food manufacturing:
Industries in the food manufacturing sector transform livestock and agricultural materials into products for intermediate or final consumption.
Food industry groups are distinguished by raw materials (generally of animal or vegetable origin) processed into food products, such as dairy, meat, etc. The food products manufactured in these establishments are typically sold to wholesalers or retailers for distribution to consumers. Establishments primarily engaged in retailing bakery, confectionary and other products made on the premises are included in this concept of food manufacturing as well as catering. The concept also includes similar parties producing ready-to-eat products.
Therefore, proper manufacture of any exportable food product becomes essential to its acceptance by the importing country. Additionally, to obtain this level of acceptability, the manufacturing process must proceed under the best manufacturing and sanitary conditions. The array of these conditions is defined under GMPs, the “specifications” by which an industry produces its products, which comprise a wide array of subjects: from the construction phase to the logistics and distribution phase; from the walls and floors of the plant to the packaging, labeling and palletizing of the product; and the sanitation of the distribution vehicles. Obviously, many of these practices differ, depending on the product, location, physical structure of the manufacturing facility, etc. Under these conditions, industry practice dictates that each location and product requires an individual set of GMPs and, in each area of the operation, specific Standard Operating Procedures (SOPs) must exist.
As defined by FDA, current food GMPs are published in Title 21 of the Code of Federal Regulations Part 110 (21 CFR 110). GMPs describe the methods, equipment, facilities and controls for producing processed food. As the minimum sanitary and processing requirements for producing safe and wholesome food, they are an important part of regulatory control over the safety of the nation’s food supply. GMPs also serve as one basis of FDA inspections.[2]
Current GMP Objectives
As GMPs are a set of generally recognized rules and procedures defining what is acceptable in the food industry, they reflect the knowledge gained from experience and scientific study in food preservation, technology and hygiene. The code’s preventive approach links GMPs closely to quality assurance, with the aim of safe, wholesome food via well-controlled operations that avoid waste and contamination.
Current GMP objectives are the following:
• To understand key issues in quality assurance/GMPs/quality control
• To understand specific requirements on quality management and quality assurance, including organization, procedures, processes and resources
• To develop actions to resolve actual and current problems/concerns
It is clear why each manufacturing process requires its own set of specifications as mentioned above, including:
• GMPs
• SOPs
• Hazard Analysis and Critical Control Points (HACCP)
• Nutritional labeling
• Origin of product regulations
• USDA regulations
To illustrate why these practices are so important, processed food sales worldwide were approximately $3.2 trillion in 2004. In the U.S., consumers spent approximately $1 trillion annually for food (almost one-third of worldwide sales), or nearly 10 percent of the U.S. gross domestic product, in an industry that employs over 16.5 million people. Of this $1 trillion, imported foods comprise around 50 percent of all fish and shellfish, 33 percent of all fresh fruit and 12 percent of all vegetables
The FSMA objectives are that the exporting food industry meets the following expectations:
• Manufacture safe and reliable (reproducible) good-quality food products
• Attain and maintain agreed-to specifications
• Follow techniques used in industrialized countries to accomplish compliance with laws and regulations
• Develop capabilities to secure inspection approval from governmental and private food inspectors/entities
However, experience has shown that the food industry in developing countries:
• Is under few or no rules and regulations, although this tendency is decreasing
• Does not strictly keep sanitation and good personnel practices (i.e., uniforms, cleanliness, pocket items, etc.)
• Maintains sometimes weak or nonexistent controls of production and process (i.e., processing times and temperatures, etc.)
• Has a system of warehousing and distribution of food products that is deficient in many aspects (i.e., temperatures, transport cleanliness, etc.)
• Maintains deficient laboratory procedures or does not follow them at all
• Maintains deficient and insufficient record keeping (i.e., recalls, product codes, etc.)
Thus, compliance with GMPs becomes paramount for such companies to export food products in a safe and sanitary manner.
GMPs and Quality
Quality must be linked to the consumer. For exports, the “consumers” are the existing markets in the importing countries. The quality expectations that already exist must be met.
To identify the basics of a quality system in which to build mandatory specifications, industry must determine 1) who are the customers, 2) what are the customers’ needs, 3) what are the products and services that must be developed to meet those needs and 4) how can we transfer these findings to our operations?
The basics of a quality system include a definition of quality:
• The economic production of consistent goods and services that delight the customer
• Compliance with specifications
Quality systems are based in control, and control requires that performance be evaluated, that this performance be compared to a standard and that you act on these differences. These principles are applied to the process control, the product inspection and the quality assurance procedures to attain compliance with specifications. If the industrial enterprise is not starting from scratch—as most concerns entering the export field are—then the easier route is to improve what is already there. Those improvements must be developed by steps and in a sequence that eliminates excessive and costly modifications. A suggested way to proceed would have the following steps:
• Establish an infrastructure for continuous improvement
• Verify specific improvement needs
• Provide resources to accomplish the following:
• Diagnose the cause
• Determine the remedy
• Establish controls to maintain modifications/improvements
The above steps must be applied to each of the operations in both manufacturing and transportation of the food product.
 The basic elements of quality management include an appropriate infrastructure or “quality system” that encompasses the organization’s structure, procedures, processes and resources (Figure 1). They also include the systematic actions necessary to ensure adequate confidence that a product will satisfy the given requirements or specifications for quality. The totality of these actions is termed Quality Assurance (QA).
The basic elements of quality management include an appropriate infrastructure or “quality system” that encompasses the organization’s structure, procedures, processes and resources (Figure 1). They also include the systematic actions necessary to ensure adequate confidence that a product will satisfy the given requirements or specifications for quality. The totality of these actions is termed Quality Assurance (QA).
To achieve a quality operation, compliance is essential with regard to regulatory requirements, basic customer requirements and basic environmental requirements.
The remainder of this article will discuss the regulatory requirements that encompass the GMPs or prerequisite areas that assure the wholesomeness of the food by including specifications for the following:
• Premises
• Receiving and storage
• Equipment performance
• Sanitation
• Personnel and training
• Health and safety recalls
A QA system should therefore ensure that products are designed and developed correctly to comply with specifications; that the enterprise is complying with GMPs; that production and control operations are defined; that managerial responsibilities are defined and include job descriptions; that the manufacture, supply and use of appropriate raw and packaging materials are under control and at appropriate costs; that in manufacturing, the necessary controls, both of processes and product quality, are in place; that correct processing and verification of the finished product quality are attained by keeping proper records and recall information; that proper storage, distribution and handling of the finished product have standards and that these are followed by third parties as well as the company itself.
How to Implement GMPs
To develop appropriate GMPs, there are prerequisites with which the food manufacturing entity must comply. These prerequisites are related to all sections comprising the manufacturing entity or company, and are like the foundations of a building that must be in place before the walls are even built.
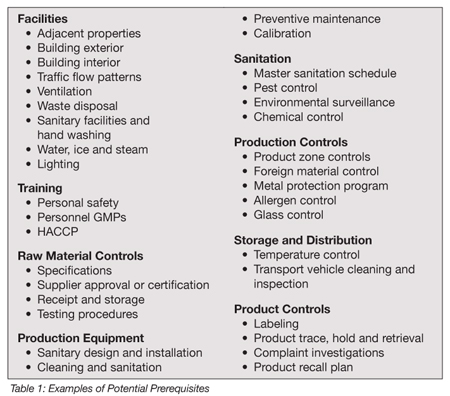 Examples of potential prerequisites are found in Table 1. Many of these prerequisites in the quality program are defined in each country by different laws, rules and regulations. For example, sanitary construction regulations for food manufacturing plants in many countries specify how the exposed walls of a manufacturing hall must be built, that is, “the surface/facing in a manufacturing room has to be of a washable material and should not cover less than 10 feet from the ground level to the end of the washable surface.” Under these or similar conditions, if the plant already complies with the local regulations, it has achieved those prerequisites.
Examples of potential prerequisites are found in Table 1. Many of these prerequisites in the quality program are defined in each country by different laws, rules and regulations. For example, sanitary construction regulations for food manufacturing plants in many countries specify how the exposed walls of a manufacturing hall must be built, that is, “the surface/facing in a manufacturing room has to be of a washable material and should not cover less than 10 feet from the ground level to the end of the washable surface.” Under these or similar conditions, if the plant already complies with the local regulations, it has achieved those prerequisites.
Effectively implemented prerequisites are important in controlling hazards, for example, plant layout and prevention of cross-contamination, aids in chemical control, Listeria monocytogenes or Escherichia coli control in the environment, etc. No simple formula exists to say which prerequisites are essentially needed.
HACCP is a systematic preventive approach to food safety that addresses physical, chemical and biological hazards as a means of prevention rather than focus on finished product inspection. HACCP is used in the food industry to identify potential food safety hazards and take actions to eliminate the risk. The system is used at all stages of food production and preparation processes, including packaging and distribution.
Both the FDA and USDA say their mandatory HACCP programs for juice and meat are an effective approach to food safety and protecting public health. Meat HACCP systems are regulated by the USDA, while seafood and juice are regulated by the FDA. The use of HACCP is currently voluntary in other food industries.
Since its conception in the 1960s, HACCP has been recognized internationally as a logical tool for adapting traditional inspection methods to a modern, science-based, food safety system. Based on risk assessment, HACCP plans allow both industry and government to allocate their resources efficiently in establishing and auditing safe food production practices. In developing countries, especially for exporting food industries, the establishing of a HACCP systematic approach is highly recommended, inasmuch as the new FSMA requires that those plants receive an inspection and certification for the produced food products to enter the U.S.
Implementing and maintaining prerequisite programs addresses existence and effectiveness of prerequisites during design and implementation of a HACCP plan that must have SOPs, maintain records and periodically verify that the specifications are being met.
Implementing HACCP
 Innumerable books, magazine articles, experts, etc. indicate (show, train, etc.) how to implement a HACCP program. Table 2 summarizes the most common steps recommended.[3]
Innumerable books, magazine articles, experts, etc. indicate (show, train, etc.) how to implement a HACCP program. Table 2 summarizes the most common steps recommended.[3]
Besides the adoption and implementation of HACCP systems and programs, experience has indicated that the other deficiency most commonly found in developing countries’ food industries is proper record keeping. Production records provide auditable proof that the job was done. Specifications of raw and packaging materials are essential to confirm the quality and amount of what has been purchased. In-process and finished product specifications and records allow for product quality control. To maintain the proper records, the existing regulations and standards must be followed. Forms must be standardized and used. The common use of pieces of paper should not be allowed but should be discouraged as these and other records must be used and properly handled for auditing and recall purposes. Documentation provides a solid base for the quality management system.
Record keeping must be easy to manage, should provide the groundwork for improvement and must be linked together to form a total company system; to achieve this, employees need the latest, most correct and appropriate documents to do their jobs. Most importantly, keep documents simple and easily understandable.
For written documentation, one must define the process:
• Input
• Suppliers
• Raw materials and raw material requirements
• Activities
• Conversion of raw materials into products
• Measurements
• Outputs
• Customers and their requirements
• Products
• Other
• Key support units and responsibilities
In summary, exporting food industries can apply GMPs with management commitment and proper support. Programs that must be implemented have been mentioned above, and it is relevant to reiterate that there are several ways to attain “compliance with specifications.” The inspection of food products at the U.S. border by FDA and USDA is a fact, and so is the inspection of the manufacturing plants in their countries of origin. Therefore, compliance is expected, and the production of safe and compliant food with U.S. laws and regulations is a must for all exporters. Additionally, if top management commitment and assurance of economic support for these programs do not exist, if the support is not constantly reaffirmed and if funds are eliminated because of “budgetary reasons,” these programs will fail and ultimately affect product quality.
Herbert Weinstein, Ph.D., earned his chemical engineering degree from the Universidad Nacional Autonoma de Mexico and his M.Sc. and Ph.D. in food science and technology from the Massachusetts Institute of Technology. He can be contacted at HERBWEIN@aol.com.
References
1. www.fda.gov/food/foodsafety/fsma/default.htm.
2. www.fda.gov/Food/GuidanceComplianceRegulatoryInformation/CurrentGoodManufacturingPracticesCGMPs/ucm110907.htm.
3. Pierson, M.S. and D.A. Corlett Jr., eds. 1992. HACCP — Principles and applications. London: Chapman and Hall.
Food Plant GMPs: How Can Foreign Food Manufacturers Comply with U.S. Regulations?




