For many companies, a circled calendar date signifying a pending visit from an independent or customer auditor is akin to having an audience with the Internal Revenue Service. This trepidation is all too understandable given the glaring fact that many companies are inundated with “redundant” food safety audits each year. Nonetheless, companies must remain focused on the big picture. In our global economy, food plants must embrace every audit as an opportunity to showcase their commitment to producing safe and quality products.
Decades ago when food safety audits were in their infancy, some companies largely viewed them as nuisances whose principal function was to derail production deadlines. However, with the growing emphasis on improving food safety standards in the United States and abroad, audits soon evolved into an indispensable component in effective food safety programs. Over time, food safety audits have grown in scope and sophistication, significantly raising the stakes and challenges for food companies. Today, companies are charged with:
• Assuring food safety and quality at multiple sites
• Maintaining expertise in numerous products and processes
• Managing QA data to monitor and improve product quality
• Staying abreast of industry and regulatory guidelines
• Meeting training requirements for an increasingly diverse workforce
Teamwork and preparation are necessary to meet these constant and enormous challenges. The following section, gathered from years of front-line experience, covers opportunity areas commonly associated with food safety audits. By being more aware, companies can enhance the auditing experience and strengthen their processes.
Someone’s Knocking on the Door
Generally, companies and auditors mutually agree on audit dates and start times. Unfortunately, things come up and fires need to be fought, leaving auditors sitting in the conference room. While these occurrences are to be expected, time is money in the bottom-line world of business. To protect their investment and complete the audit within the allotted time frame, companies should give consideration to establishing contingency procedures in the event the primary escort is unavailable.
General audit protocols should encompass: 1) Plans for ensuring the auditor is always accompanied through the plant; 2) Audit schedules for appropriate plant personnel; 3) A private and secure location for document review; 4) A proactive course of action to reschedule audits when key personnel are unexpectedly unavailable or serious in-plant emergencies occur; and 5) Identification of key personnel to participate in the audit debriefing.
All in the Details
There should be few, if any surprises, in food safety audits. Through audit forms and other similar documents, plants are given information about the attributes on which they will be evaluated. Without question, one of the most critical categories is document review, which includes sanitation standard operating procedures (SSOPs), Hazard Analysis and Critical Control Points (HACCP), Good Manufacturing Practices (GMPs), allergen control, and other prerequisite programs. Despite this, auditors are often blindsided by procedures and records that are disorganized, incomplete or inaccessible. From undocumented mock audits to missing training records, these costly transgressions can adversely affect audit outcomes.
Quality implies documentation that can be verified; in our heightened regulatory environment, this point cannot be overstated. Well-organized and up-to-date operation manuals detailing quality assurance and quality control (QA/QC) program policies and procedures must be readily available during audits. Manuals should also include the roles and responsibilities of the various functional areas impacting food safety and quality, as well as procedures for corrective actions and continuous improvements. If the manual does not contain all the records, the auditor must be provided with cross-references indicating the location for these documents. Plants may be asked to provide records for up to two or three years based on applicable regulations. In today’s electronic world, document retention processes should also include procedures for secured back-ups of electronically retained records.
Regrettably, there is no magic bullet to ensure effective record keeping. Plant management must continuously roll up its shirtsleeves, button down the hatch, and stay on top of this unglamorous though essential task. The tangible benefits of doing so far outweigh the repercussions of providing auditors with documents that have more holes than Swiss cheese. Moreover, it is important to have complete and accurate documentation in the event of a food safety emergency.
Fear of the Black Hat
Audit reports contain valuable information to assist in improvements. However, reports are often filed away and are not used as a source for process improvements, reducing risks and strengthening customer relations. In many instances, organizations do not possess a clear understanding or concise road map for implementing corrective actions.
As an invaluable reservoir of knowledge, trained and experienced auditors can help identify areas of opportunity for setting plants on the right path. Ideally, the entire plant management team should be present during audit finding meetings. This is an opportune time for staff to exchange information, ask questions, begin to develop corrective action plans, and establish timelines for their implementation.
Like black-hat-wearing villains in old “B” movie cowboy westerns, plant personnel sometimes view auditors as distant and disruptive forces that should be avoided at all costs. This attitude is both pointless and counterproductive. Auditors can provide plants with important information on food safety measures that can impact daily operations and serve as sounding boards for new ideas. Through open communication and a willingness to build bridges, companies can cultivate positive working relationships with auditors that can enhance their food safety and quality management programs.
Major Crossroad
While it would be possible to write at length on other common pitfalls, we would be amiss if we failed to touch upon a major crossroad in auditing. Seeking greater assurances in the safety of products they purchase, retailers everywhere are increasingly requiring suppliers to adhere to food safety management systems that have been benchmarked by the Global Food Safety Initiative (GFSI). GFSI is a group of major international retailers, including Wal-Mart USA, Food Lion and Hannaford USA, that is committed to strengthening consumer confidence in the food sold by retail outlets.
With certification programs administered by the Safe Quality Food (SQF), British Retail Consortium, International Food Standard, and Dutch HACCP, this retail driven initiative is designed to end the redundancy and confusion associated with a plethora of on-site inspection programs. According to SQF, the benefits afforded under its program include:
• A tool to build confidence and trust between retailers and suppliers
• A proactive tool for producers and manufacturers to demonstrate “due diligence” and compliance with regulatory and product traceability requirements
• An internationally recognized standard for all food suppliers operating in domestic and global markets
• A reduction in the number and frequency of inconsistent and costly audits
• A proven way for suppliers to gain an advantage over their non-certified competitors and increase profits by aligning their products to retailer/consumer requirements
• A management system that promotes cost efficiencies through “one system, one audit”
• A framework for demonstrating sustainable production and manufacturing best practices
Currently, these programs are better known and more widely embraced outside of the U.S. In the coming years, however, their prominence is expected to grow here as suppliers become more familiar with the savings, services and advantages that can be derived from certification. Companies wishing to learn more about can log on to www.sqfi.com or www.ciesnet.com.
Consumer Confidence Crisis
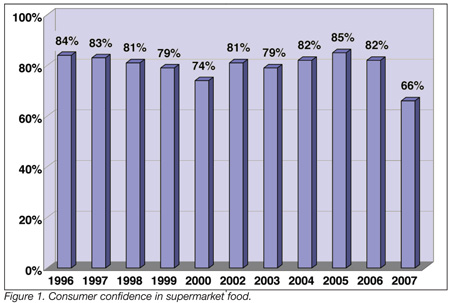 Following a series of high-profile foodborne illness cases last year, consumer confidence in the safety of the U.S. food supply is tumbling. According to a recent Food Marketing Institute (FMI) survey, 66% of shoppers are confident that the food they buy at grocery stores is safe, representing a 16% decline from last year and the lowest rating in nearly 20 years (Figure 1). Consumers are also extremely wary of restaurant food, with less than half of the participants (42%) expressing confidence in the safety of meals eaten away from home.
Following a series of high-profile foodborne illness cases last year, consumer confidence in the safety of the U.S. food supply is tumbling. According to a recent Food Marketing Institute (FMI) survey, 66% of shoppers are confident that the food they buy at grocery stores is safe, representing a 16% decline from last year and the lowest rating in nearly 20 years (Figure 1). Consumers are also extremely wary of restaurant food, with less than half of the participants (42%) expressing confidence in the safety of meals eaten away from home.
Finally, 38% of survey respondents indicated they have stopped buying produce, meat, poultry, and other commodities due to outbreaks and recalls that occurred in 2006. In the U.S, FMI members operate approximately 26,000 retail food stores with a combined annual sales volume of $340 billion.
Faced with this ebbing consumer confidence in the safety of the U.S. food supply, manufacturers and processors are investing additional resources into inspections—ranging from increased environmental monitoring and verification testing to food safety audits—to protect consumers and their brand. Like any investment, poise, preparation and planning are key to assuring the long-term success of these programs. As evidenced in this article, it is essential to always utilize the “Three P’s” in food safety audits.
Rena Pierami is vice president of technical services for Silliker, Inc. Prior to joining the Homewood, IL-based organization, she served as senior director of quality systems for the KFC Corp. in Louisville, KY. With over 25 years of industry experience, she is certified by the American Society for Quality and serves on the International HACCP Alliance Board.
John Williams Jr. is a Silliker senior communication specialist and has written extensively on a broad spectrum of food safety and quality subjects. Both authors can be reached via e-mail at info@silliker.com.
Guaranteeing a Great Audit




