Nonalcoholic beverages include water and carbonated water, fruit and vegetable juices and nectars, water-based, flavored carbonated and noncarbonated drinks and water-based brewed or steeped beverages, such as coffee and tea. Safety for the human consumption of these products is regulated in each country by national regulations based on codes and standards derived by the Codex Alimentarius Commission.[1] European Union (EU) member states use European legislation for microbiological criteria, food additives and general hygiene requirements for the production, storage and trade of food products, as well as specific requirements for safety and quality of such beverages. The Republic of Macedonia has developed national legislation for food safety, primarily based on European legislation and Codex standards, so the safety of nonalcoholic beverages is covered by these regulations.[2, 3]
The basic ingredients of a beverage are water, sweetener, acid and flavor. Optional ingredients often include fruit and/or fruit juice, carbon dioxide, preservatives and color. Water is always the major ingredient and represents approximately 86% of a carbonated drink, 90% of a fruit juice and 100% of bottled waters. Nonalcoholic drink ingredients can be divided into two categories: food substances, like fruit, juice, sugars and starches, and additives, like sweeteners or preservatives. Additives are defined as substances added to food to maintain quality, texture, consistency, appearance, taste, alkalinity, acidity, etc. Some additives have adverse health effects in humans and have been banned for use in beverage production. International standards and national legislation prescribe detailed rules for the mandatory labeling of additives used in a product to enable informed choices by consumers and avoid consumption of additives when necessary.[1, 4]
Preservatives in Nonalcoholic Beverages
Since nonalcoholic beverages are high in water activity and some are rich in vitamins and minerals, they are an attractive environment for microbes. However, the usually low pH of beverages, due to carbonation, the sugar content in some of them and the addition of preservatives help inhibit the growth of microbes. The type of chemical preservative that can be used in beverages depends on the chemical and physical properties of both the antimicrobial preservative and the beverage. The pH of the product, the presence of vitamins, the packaging and the storage conditions will determine whether preservatives are necessary and what type should be used to prevent microbial growth. The main preservatives allowed and used in nonalcoholic beverages are sorbic and benzoic acids and their salts.[2, 4]
Sorbates are very effective preservatives against yeasts, molds and bacteria. The antimicrobial effectiveness of sorbates depends on the physical and chemical properties of the beverages. Sorbates and benzoates are often used in combination, especially in highly acidic drinks. Benzoic acid occurs naturally, notably in cranberries, cinnamon, plums and currants, and it has long been used to inhibit microbial growth in many products, including nonalcoholic beverages. Benzoate salts are more stable than the acid form and more soluble in water, making benzoates a favorable choice for the beverage industry. The salts are particularly well suited for use in carbonated, nonalcoholic and juice beverages because they work best between pH levels of 2 and 4.4
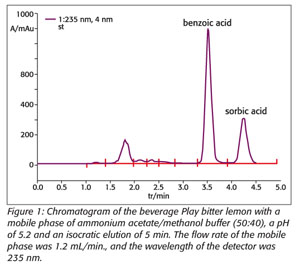
Therefore, we need techniques to detect and quantify benzoic and sorbic acids in beverages to prevent excessive human exposure. Traditional techniques such as titration and spectrophotometry usually require extensive sample pretreatment. Gas chromatography methods require complex pretreatments, such as several extraction, evaporation and derivatization steps, which might reduce analytical precision. High-performance liquid chromatography (HPLC) methods are more attractive for that purpose, offering the possibility to analyze the additives without prior steps but with high precision and accuracy. At the Institute of Public Health of the Republic of Macedonia, in the laboratory for food quality testing, we use an HPLC method for the quantification of additives. Samples are filtered through a membrane filter with a pore diameter of 0.45 μm and chromatographed. A method for benzoic and sorbic acid determination in beverages by liquid chromatography using a diode array UV-VIS detector (UV-VIS-DAD) involves a reversed-phase mode and a mobile phase of ammonium acetate/methanol buffer (50:40), with a pH of 5.2 (Figure 1). This HPLC method is complete within 5 minutes after sonication and filtration. The results showed sorbic and benzoic acid concentrations vary with different kinds of beverages, with lower than the maximum levels allowed by national legislation.[5, 6]
 When ascorbic acid (vitamin C) is present as an ingredient in beverages along with sodium benzoate, benzene formation may occur under certain conditions. Formation of benzene is exacerbated in beverages if they are stored for extended periods at elevated temperatures. Although the levels and frequencies at which such benzene formation has occurred in the past have not posed a public health risk, the beverage industry has developed methods to prevent or minimize its occurrence. In recent years, the use of benzoates has been reduced because of new processing techniques, but it is still necessary to use these preservatives in some beverages to maintain quality.[7]
When ascorbic acid (vitamin C) is present as an ingredient in beverages along with sodium benzoate, benzene formation may occur under certain conditions. Formation of benzene is exacerbated in beverages if they are stored for extended periods at elevated temperatures. Although the levels and frequencies at which such benzene formation has occurred in the past have not posed a public health risk, the beverage industry has developed methods to prevent or minimize its occurrence. In recent years, the use of benzoates has been reduced because of new processing techniques, but it is still necessary to use these preservatives in some beverages to maintain quality.[7]
In 2006, the United Kingdom Food Standards Agency published the results of its survey of benzene levels in soft drinks, which tested 150 products and found that four contained benzene levels above the World Health Organization guidelines for drinking water (10 μg/L).7 The U.S. Food and Drug Administration (FDA) released its own test results of several soft drinks containing benzoates and ascorbic acid. Five tested drinks contained benzene levels above the U.S. Environmental Protection Agency-recommended standard of 5 ppb; despite these findings, as of 2006, FDA stated its belief that “the levels of benzene found in soft drinks and other beverages to date do not pose a safety concern for consumers.”[8] At present, we don’t have available data for benzene concentrations in nonalcoholic beverages from the market in the Republic of Macedonia.
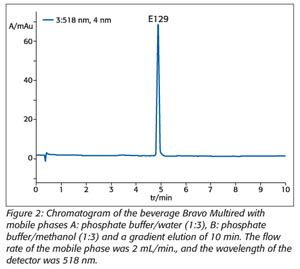
Dyes are added to some nonalcoholic beverages, but they must be labeled. Consumers thereby have the opportunity to decide whether they will consume beverages with added dyes. HPLC with a UV-VIS-DAD was employed for the determination of synthetic dyes as well,9 using reversed-phase conditions and a short monolith column (Chromolith RPe; 50-4.6 mm). Sample preparation by means of typical membrane filtration is unacceptable, as the synthetic dyes will be absorbed. To prevent the decreased intensity of dyes, samples are dissolved in a solution of methanol/water (1:1, v/v) (Figure 2). Using this method, we detected a few samples from imported fruit juices with added colors like E102 and E122, which are not allowed for use in this kind of product in Macedonia.[9]
Low-Calorie Sweeteners
Sucrose has been widely used in beverages as a sweetener. However, the special dietary requirements of diabetics and health concerns about obesity and dental caries have triggered considerable research into the development of alternative sweeteners. Most low-calorie beverages on the EU market frequently use intense sweeteners, which were approved for use in the EU in 1994 as governed by an EU Directive for sweeteners in food. This legislation sets limits for the use of each intense sweetener in food and drink products. The law also states that when any intense sweetener is used, there must be a minimum 30% reduction in sugar content compared with regular products. The use of intense sweeteners in Macedonia is regulated by national legislation for food additives and food labeling, which has been harmonized with European standards. Acceptable daily intake levels are set for each intense sweetener by Codex Alimentarius Commission standards, which indicate safe consumption of intense sweeteners every day over a lifetime.[1, 3]
Because of high consumer demand and acceptance of low-calorie beverages, the market for artificially sweetened foods has increased significantly. The demand for detection and quantification of these additives in beverages has also increased for safety reasons. The majority of recently published protocols for the determination of sweeteners are based on HPLC methods, which offer great advantages over more traditional analyses. HPLC methodology may not require very complicated sample preparation when beverages are analyzed since it is easier to work with a liquid matrix.[10]
There are many methods available for determination of artificial sweeteners. Most of the HPLC methods used are based on isocratic- or gradient-grade reversed-phase chromatography with UV detection. However, methods capable of determining several sweeteners simultaneously are required because there are more compounds available that are used as a blend. Thus, there is a continuous search for simpler, faster and more sensitive methods capable of detecting several sweeteners in a single analytical run for routine analysis in the quality control of various food products.[10–12]
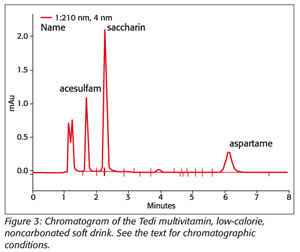
One method that has been successfully applied at the Institute of Public Health performs the simultaneous determination of saccharine, acesulfame K and aspartame in a phosphate buffer (0.1 M NaH2PO4) acidified to pH 3.5 by phosphoric acid. The mobile phase consists of methanol and buffer in a ratio of 25:75. Separation of the sweeteners is achieved using a reversed-phase select B chromatography column (125 min., 4 mm). Isocratic elution is applied with a flow rate of 1.0 mL/min. The compounds of interest are detected at a wavelength of 210 nm. A chromatogram of a noncarbonated soft drink containing saccharine, acesulfame K and aspartame is shown in Figure 3.
Conclusions
Laboratory techniques for the detection and quantification of chemicals in nonalcoholic beverages are essential to identify potential health risks. Foodborne diseases constitute a substantial public health concern, and most are largely preventable by proper application of appropriate food technologies. At an industrial level, applications of Hazard Analysis and Critical Control Points systems combined with controls performed by health and food control authorities are effective in ensuring the safety of nonalcoholic products. Consumers are still in need of further education on the use of additives in nonalcoholic products and their potential effects on human health. The food industry can help inform consumer choice by emphasizing this topic in health education programs and posting relevant information on company websites.
 Gordana Ristovska, M.D., Ph.D., is a specialist in hygiene and environmental health and head of the department for food safety at the Institute of Public Health of the Republic of Macedonia.
Gordana Ristovska, M.D., Ph.D., is a specialist in hygiene and environmental health and head of the department for food safety at the Institute of Public Health of the Republic of Macedonia.
 Maja Dimitrovska, M.Sc., is a specialist in sanitary chemistry in the food control laboratory at the department for food quality at the Institute of Public Health of the Republic of Macedonia.
Maja Dimitrovska, M.Sc., is a specialist in sanitary chemistry in the food control laboratory at the department for food quality at the Institute of Public Health of the Republic of Macedonia.
 Anita Najdenkoska, B.Sc., is a senior food analyst at the department for food quality at the Institute of Public Health of the Republic of Macedonia.
Anita Najdenkoska, B.Sc., is a senior food analyst at the department for food quality at the Institute of Public Health of the Republic of Macedonia.
2. European Parliament and Council Directive 95/2/EC (1995) on food additives other than colours or sweeteners. Official Journal of the European Communities L61, 18.3.95, 1–40.
3.Regulation (EC) No. 1333/2008 of the European Parliament and of the Council of 16 December 2008 on food additives. Official Journal of the European Union L354, 31.12.2008, 16–33.
5. Pan, Z., L. Wang, W. Mo, C. Wang, W. Hu and J. Zhang. 2005. Determination of benzoic acid in soft drinks by gas chromatography with on-line pyrolitic methylation technique. Anal Chim Acta 545:218–223.
6. Saad, B., F. Bari, M. Saleh, K. Ahmad and K. Talib. 2005. Simultaneous determination of preservatives (benzoic acid, sorbic acid, methylparaben and propylparaben) in foodstuffs using
high-performance liquid chromatography. J Chromatogr A 1073:393–397.
7. webarchive.nationalarchives.gov.uk/ 20120206100416/http://food.gov.uk/multimedia/pdfs/fsis0606.pdf.
9. Kiseleva, M. G., V. V. Pimenova and K. I. Eller. 2003. Optimization of condition for the HPLC determination of synthetic dyes in food. J Anal Chem 58:685–690.
10. Zygler, A., A. Wasik and J. Namies’nik. 2009. Analytical methodologies for determination of artificial sweeteners in foodstuffs. Trends Anal Chem 28(9):1082–1102.
11. Zhu, J., Y. Guo, Y. Mingli and S. J. Frits. 2005. Separation and simultaneous determination of four artificial sweeteners in food and beverages by ion chromatography. J Chromatogr A 1085:143–146.
12. Cantarelli, M. A., R. G. Pellerano, E. J. Marchevsky and J. M. Camiña. 2009. Simultaneous determination of aspartame and acesulfame-K by molecular absorption spectrophotometry using multivariate calibration and validation by high performance liquid chromatography. Food Chem 115(3):1128–1132.




