Nanotechnology involves the preparation and use of submicroscopic particles with sizes ranging in nanometer scale, conferring special physiochemical properties to these particles and positioning nanotechnology as a critical research endeavor of this century. Their importance also magnifies the efforts needed to study their effects on biological systems, as drug delivery is one of the prime areas for which nanoparticles are being researched.
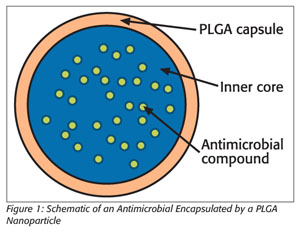
Nanoparticles can be prepared from a variety of substances, ranging from metals to polymeric compounds to elements. Much work has been done in elucidating the role of metal nanoparticles in material sciences. However, such studies in nanomedicine indicated adverse effects that made them unsuitable for use in therapeutic research. In addition, nanomedicine uses materials approved by the U.S. Food and Drug Administration (FDA) that are both biodegradable and biocompatible. PLGA (polylactic glycolic acid), an FDA-approved compound, is a copolymer of lactic and glycolic acids and has been used in nanotechnology extensively (Figure 1).
Biodegradation of PLGA Nanoparticles
PLGA biodegradation occurs both in vitro and in vivo in aqueous environments by hydrolysis of the backbone ester linkages. The biodegradation rate depends on the molar ratio of lactic and glycolic acids in the polymer, the molecular weight, the degree of crystallization and the glass transition temperature. PLGA degrades into lactic and glycolic acids; lactic acid enters the tricarboxylic acid (TCA) cycle and is then metabolized and eliminated from the system as water and carbon dioxide, whereas glycolic acid may undergo similar metabolism in the TCA cycle or is excreted by the kidneys unchanged.
Biocompatibility of PLGA Nanoparticles
PLGA nanoparticles have been shown to possess advantageous properties, such as low immunogenicity, good mechanical properties, low toxicity and predictable biodegradation mechanisms that make them ideal compounds for use in therapeutics.[9] Its very low immunogenicity makes it an extremely suitable candidate for use in biomaterials and biomedical applications, including sutures, nerve/dental/bone repairs, etc. Additionally, it is important to note that PLGA/PLA (polylactic acid) nanoparticles can easily be modified on their surfaces to further increase resistance to opsonization and macrophage-mediated degradation by coating with hydrophilic compounds.
Biodistribution of PLGA Nanoparticles
Because they’re so small, PLGA nanoparticles can cross extremely selective biological barriers and are hence able to deliver drugs to otherwise difficult-to-access body organs. PLGA nanoparticles are found in all major organs after administration, with highest concentrations seen in the liver and kidneys.[2] Further, once in the organs, they are readily degraded as described above, leading to no accumulation over time. In addition, the organs exposed to PLGA nanoparticles show no signs of reduced viability or alterations of metabolic processes, confirming studies conducted in cell culture models.
All studies to date have shown the beneficial effects of PLGA nanoparticles that make them suitable candidates for use in nanomedicine. PLGA has already been used to administer specific cancer drugs, nutraceuticals and drugs to treat brain ailments, not only elucidating their biosafety but also illuminating their release kinetics, increase in efficiency of the drug/compound and increased beneficial results with possible mechanisms of the test compounds delivered by these particles. Additional studies that can confirm their biosafety profile might lead to their use at a commercial level in a wide range of applications, spanning foods, drugs, dental/bone fills, etc. With this approach, PLGA/PLA nanoparticles definitely will have a revolutionary impact on the way molecules are delivered.
Application of Nanoparticles in Meat Systems
We have experimented with the use of phenolic compounds in PLGA nanoparticles in poultry meat systems, using broth studies. In both raw and cooked chicken, nanoparticle delivery of benzoic acid was efficient against Salmonella Typhimurium and Listeria monocytogenes [1.0 and 1.6 log colony-forming units (CFU)/g reduction in Salmonella Typhimurium and 1.1 and 3.2 log CFU/g reduction of L. monocytogenes compared with 1.2 log CFU/g without nanoparticles on days 9 and 14 of storage, respectively]. These findings demonstrate the efficacy of pathogen reduction by phenolics delivered by nanoparticles; however, since meat is a complex system and has shelf-life issues, better methods for delivering nanoparticles containing antimicrobials to meat systems might prove beneficial.
Edible films: An ideal solution for the food industry to overcome food safety and environmental problems is to incorporate antimicrobial substances into edible coatings.[3] Antimicrobial packaging as edible films can be a challenging form of active meat packaging. Direct surface application of antimicrobials diffuses them rapidly into the food mass, minimizing their protective effect. Incorporating these antimicrobials into edible films could effectively sustain their inhibitory effects for an extended period of storage by slow migration of compounds, which would help maintain high concentrations when required. Several antimicrobial agents, including lysozyme, nisin, pediocin, p-aminobenzoic and sorbic acids, have been incorporated into edible films and shown to inhibit Escherichia coli O157:H7, Lactobacillus plantarum, L. monocytogenes and Salmonella Typhimurium.[3, 4] Our laboratory has advocated the use of edible film technology to deliver malic and lactic acids containing nisin in soy protein films.[5] Malic acid (2.6%)-incorporated soy protein film has the fewest survivors of L. monocytogenes, Salmonella Gaminara and E. coli O157:H7 (5.5, 3.0 and 6.8 log CFU/mL, respectively). Soy protein films incorporated with grape seed and green tea extracts and nisin were tested on turkey frankfurters and provided more than 2.0 log CFU/g reduction of L. monocytogenes during a 28-day storage period at 4 °C and 10 °C.[6]
Preparation of nanoparticle-containing edible films: The method for preparing soy protein edible films was standardized by Eswaranandam et al.4 A 10 percent solution of soy protein isolate in deionized water is prepared and stirred at room temperature for 1 hour for total dissolution of the protein. Glycerol, used as a plasticizer, is then added at a 35 percent (w/w) protein basis, and the solution is stirred for 30 minutes. After homogenizing for 2 minutes to ensure complete homogeneity of the sample, the solution is heated at 85 °C for 30 minutes and filtered through cheesecloth. Antimicrobial-containing nanoparticles are added at the desired concentration to the edible film protein solution and stirred for 1 hour. The solution is centrifuged at 10,000 rpm for 15 minutes to remove air bubbles and cast at uniform thickness on plastic sheets (i.e., to act as models for edible films) using a drawdown machine or used directly for coating meat (i.e., dipping meat in an edible film solution). While plastic sheets can be dried in humidity chambers (45 °C/40% relative humidity/4 h) to obtain edible film models for testing textural properties of the films, antimicrobial properties of nanoparticles in edible films can be tested in meat models.
Technology Potential
Edible films potentially could be developed that contain antimicrobially encapsulated nanoparticles for improving the safety of meat. This multiple-hurdle technology is commercially feasible, economical and environmentally friendly, owing to the readily available ingredients for making edible films and the biodegradable quality of edible films as opposed to non-edible synthetic films. This technique can offer decontamination of surface pathogens and protection from recontamination by pathogenic bacteria during both pre- and postpackaging of meat and serve as an anchor for the controlled release of antimicrobials for an extended period of time to maintain product quality and extend shelf life. We can expect that such new systems will have either substantially higher antimicrobial activity or higher stability than free antimicrobials. Because of the small size of the capsules trapped within a transparent edible film, no change in appearance or texture of foods should be observed. This research could dramatically improve the safety of meat products.
 Navam Hettiarachchy, Ph.D., is a university professor in the department of food science at the University of Arkansas, Fayetteville. She earned a Ph.D. in molecular biochemistry at the University of Hull, England.
Navam Hettiarachchy, Ph.D., is a university professor in the department of food science at the University of Arkansas, Fayetteville. She earned a Ph.D. in molecular biochemistry at the University of Hull, England.
 Madhuram Ravichandran is a Ph.D. student in the laboratory of Dr. Hettiarachchy and received an undergraduate degree in biotechnology.
Madhuram Ravichandran is a Ph.D. student in the laboratory of Dr. Hettiarachchy and received an undergraduate degree in biotechnology.
2. Athanasiou, K. A., G. G. Niederauer and C. M. Agrawal. 1996. Sterilization, toxicity, biocompatibility and clinical applications of polylactic acid/polyglycolic acid copolymers. Biomaterials 17(2):93–102.
3. Padgett, T., I. Y. Han and P. L. Dawson. 1998. Incorporation of food-grade antimicrobial compounds into biodegradable packaging films. J Food Prot 61(10):1330–1335.
4. Cagri, A., Z. Ustunol and E. Ryser. 2001. Antimicrobial, mechanical and moisture barrier properties of low pH whey protein-based edible films containing p-aminobenzoic or sorbic acids, J Food Sci 66(6):865–871.
5. Eswaranandam, S., N. S. Hettiarachchy and M. G. Johnson. 2004. Antimicrobial activity of citric, lactic, malic, or tartaric acids and nisin-incorporated soy protein film against Listeria monocytogenes, Escherichia coli O157:H7 and Salmonella Gaminara. J Food Sci 69:79–84.
6. Sivarooban, T., N. S. Hettiarachchy and M. G. Johnson. 2008. Transmission electron microscopy study of Listeria monocytogenes treated with nisin in combination with either grape seed or green tea extract. J Food Prot 71(10):2105–2109.