Last month, I went to the dentist for my routine 6-month checkup. Maintaining good oral health has always been important to me and was stressed in our family when I was growing up. As my father used to quip, “Be true to your teeth or your teeth will be false to you.”
The dental hygienist cleaning my teeth asked if I brush my teeth manually or if I use an electric toothbrush. I informed her that I use an electric brush, but she recommended a different brand that vibrates at a higher speed because it is more effective at preventing plaque formation. She explained that plaque is a dental biofilm ecosystem, a coating on the teeth that forms when bacteria, often Streptococcus or Neisseria, adhere to the tooth surface. The coating hardens after about 48 hours, and within 10 days, plaque becomes dental calculus, or tartar, which is difficult to remove and can ultimately result in damage to the tooth, the surrounding soft tissue and other health concerns. Plaque results from inadequate brushing and flossing, so the combination of deep flossing below the gumline and firm, rapid brushing can prevent the formation of dental biofilm.
 Food Industry Concern: Biofilm Formation
Food Industry Concern: Biofilm Formation
Just as all of us are challenged by the formation of biofilm on our teeth, a challenge that food processors and their sanitation teams face is the formation of biofilms on food equipment surfaces. Food manufacturing plants, particularly sanitation and food safety/quality personnel, will want to recognize this potential hazard related to sanitation and that biofilms can have a profound impact on the safety and quality of their products. Biofilm formation can contaminate product through the introduction of pathogenic microorganisms or spoilage bacteria. Biofilms have been described as a “metabolically active matrix of cells and extracellular compounds” or as “matrix-enclosed bacterial populations adherent to each other and/or to surfaces or interfaces.”[1] They may contain spoilage bacteria such as Pseudomonas and Enterococcus spp. as well as pathogens such as Listeria monocytogenes, Staphylococcus aureus, Escherichia coli O157:H7 or Salmonella. They are difficult to remove, are often resistant to normal sanitation procedures and can result in other detrimental process effects. Even when a food surface appears to be clean, the presence of biofilms is a potential hazard that must be eliminated and prevented from reoccurring. Before this can be done, it is important to understand what a biofilm is and how it is formed.
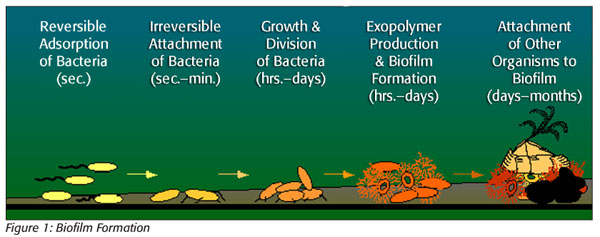
Biofilms begin with a bacterial adhesion, referred to as a conditioning layer, of organic (protein) or inorganic matter forming on an otherwise visually clean food contact surface (Figure 1). The accumulation of organic and inorganic material on processing surfaces creates an environment where bacteria can adhere. Live, damaged or dead cells can attach themselves to the conditioning layer and begin colonization. The conditioning layer starts as a thin, resistant layer of microorganisms, any combination of spoilage and pathogenic bacteria that form on and coat the conditioning layer. As the layers of bacteria attach to the surface and each other, they trap debris and nutrients, and the biofilm begins to take shape. Bacterial appendages (e.g., fimbriae, pili and flagella) may also facilitate the attachment of other cells or materials to form the colony. For example, L. monocytogenes likely attaches to surfaces by producing attachment fibrils. During attachment, cells in the forming colony work together in a coordinated and cooperative fashion, including channeling nutrients to the film and removing waste products.
As the colony continues to attach, there is production of extracellular polysaccharides and changes in cell morphology. Extracellular polysaccharide formation aids adhesion of the cells in the film and protects the bacterial layer against cleaners and sanitizers. The polysaccharide will also trap other cells and debris. Dr. Virginia Deibel states that the extracellular polysaccharide material forms a bridge between bacteria and the conditioning layer with a combination of electrostatic and covalent bonds.[2] Development and growth, without removal intervention, results in the film becoming irreversibly attached to a substratum, interface or to each other, embedded in a matrix of extracellular substance that they have produced. Mature film reaches an equilibrium that delivers oxygen, food and nutrients while carrying away fermentation products and sloughed cells. The outermost slime layer of film serves as a snare that traps additional contaminants and acts as a protectant, sealing the bacteria within so that the protected bacteria can be up to 100 times more resistant to sanitizer. As an example, L. monocytogenes in biofilms is more resistant to sanitizers than those not in films. Inorganic and organic material flowing over the biofilm provides nutrients to the colony. Inside the biofilm, damaged or small cells may have the time to repair themselves and reproduce. The film is irreversible and now requires a special cleaning protocol for removal.
Biofilms form at a slow but steady rate and, much like tooth tartar, become harder to remove over time. They can form on almost any surface but are most likely to form on rough, penetrable surfaces, such as those that are scratched, pitted, corroded or cracked.[3] They can attach to all types of surfaces in food plants, from stainless steel, especially on abraded or scratched surfaces, to polypropylene. Biofilms may form in hard-to-reach areas, such as the undersides of conveyor belts and seals. For this reason, it is necessary to regularly inspect and change equipment parts such as gaskets, O-rings and piping. Where possible, food plants should identify and eliminate areas that cannot be thoroughly cleaned through sanitary design of equipment. Extended production runs with minimal cleanup in between also increase the chances and frequency of films developing due to increased organic material contact time, opportunity for organisms to multiply and formation of the conditioning layer. These longer runs also cut into valuable sanitation time and reduce the ability of sanitarians to do their job as designed, causing them to rush or take shortcuts. Additional factors that contribute to biofilm formation include the lack of stringent cleaning regimes, pH extremes and high contact-surface temperatures that denature proteins, facilitating the formation of a conditioning layer, a low fluid flow rate and nutrient availability.
Challenges Associated with Biofilms
There are several problems associated with biofilms, not the least of which is product contamination. Product contamination occurs from sloughing bacteria that are shed periodically by the film and can reattach on equipment somewhere else in the product flow or can make their way into food product. If these are spoilage organisms, product shelf life may be reduced, and consumer purchases, especially repeat purchases, may decline. However, if they are pathogens, the product may be considered adulterated and subject to recall, or it may be responsible for a foodborne illness outbreak. Any company that has been involved in a recall or whose product has been associated with an illness can attest to the fact that biofilms are damaging to the business and extremely expensive. Organisms within the film are also more heat resistant, so sloughed cells from films that form before cooking may not be destroyed as readily by the lethality process.
Just as dental plaque damages teeth through the production of acids as a by-product of bacterial metabolism, biofilm formation comes with associated problems, such as accelerated deterioration of equipment through corrosion from cellular byproducts. There may also be a reduction in the efficacy of heat transfer and impairment of detection devices as the film disrupts transmission. As previously indicated, attached cells can develop increased resistance to cleaning chemicals and sanitizers possibly due to protection provided by the polysaccharide layer. The reduction in effectiveness of chemicals may be the result of cells layering and the reduction of exposed surfaces on which the chemicals make contact. As an example, cells of L. monocytogenes in biofilms have been found to be more resistant to sanitizers than nonattached cells.
Evidence of Biofilm
There are several means of determining that a biofilm has begun to form on a food contact surface. Detection may be via the use of several senses. Visual signs include a “rainbow” appearance on stainless steel, and tactile senses will detect a slimy feel on otherwise clean-appearing equipment surface. Although sour or off-odors may not indicate the presence of biofilms, they may indicate that a piece of equipment is not being cleaned thoroughly and that there is a potential for biofilm formation.
From an analytical standpoint, another indicator of biofilms is a sporadic spike in environmental test results due to bacterial sloughing. These indications may be found through generic microbiological tests such as aerobic plate count or through environmental pathogen testing for plants conducting Listeria swabs. An increase in the bacterial counts or positive findings may indicate the formation of a biofilm and bacterial sloughing. Unfortunately, if they are not detected soon enough, the result may be sporadic product microfailures or decreased product shelf life. However, if you are already at a point where product has begun to fail shelf life or demonstrate higher than normal bacterial counts, it would be wise to consider the possibility of biofilm formation and apply control measures to eliminate it. Adenosine triphosphate (ATP) bioluminescence devices can be used to detect the presence of organic materials; unfortunately, ATP may not detect the presence of mature biofilms. The reason for this is that embedded cells do not move as much due to nutrient availability and use less energy, thus producing less ATP. Therefore, the device may deliver a “Pass” reading on a surface where there is a biofilm.
Biofilm Removal
There should be no good reason for the films to build if there is good sanitary equipment design and regular and thorough cleaning to remove surface soil and subsurface film. Food manufacturing equipment poses many sanitary design challenges. Equipment has hollow rollers and tubing, welds, joints and scrapers that make cleaning difficult. Once biofilms have established themselves on a surface, they are harder to remove as they develop over time and require more aggressive action to eliminate. Fortunately, the original biofilm attachment is weak and easy to remove through proper sanitation procedures. Therefore, the film soil must be removed, and the most effective method of cleaning is a standard process, described below:
Dry clean. This is done to remove as much visible soil or product material as possible. This may involve scraping, brushing, vacuuming, sweeping or shoveling to remove large particles.
Potable rinse. The temperature of rinse water to remove initial soil should be between 120 and 130 ºF to break down fats, but should not exceed 140 ºF to prevent creating baked-on soil conditions or mineral scale formation that will make removal of the biofilm even more difficult.
Apply detergent. In general, the use of a chlorinated alkali or a combination of oxidative agents and acids, such as hydrogen peroxide and peracetic acid, is recommended to break the chemical bonds of food soils. Depending on the food materials that the plant makes, the processes involved and the soils created, it may be necessary to work closely with the sanitation chemical supplier to determine the specific combinations of cleaning chemicals and sanitizers to use. Application of the recommended chemicals over an extended exposure time (> 5 min.) will also be necessary to allow them to begin breaking down and removing the coating layer. Mechanical action (scrubbing on surfaces) or agitation, such as in a clean-out-of-place tank for small parts, is the most effective means of biofilm removal and must be applied to completely remove the top layers of soil and subsurface attachment conditioning layer. Scrubbing with abrasive pads or brushes will help break down the films for removal; however, the scrubbing must not be so intense as to etch or scratch the equipment surface. Etching the surface only creates additional niches where films can form and makes removal more difficult.
Final warm water rinse. Again, the rinse water temperature should be approximately 140 ºF to remove all cleaning chemical and bound soils.
Sanitize. Some bacterial cells may remain after cleaning, so the application of sanitizer should reduce remaining bacterial count to negligible levels. Cleaning must be thorough, since simply applying sanitizers to soiled surfaces is ineffective and a waste of money because the efficacy of sanitizer is reduced by the presence of soil. However, once the soil is removed and the biofilm is exposed, a higher concentration of sanitizer should be applied because lower levels will be less effective at killing the microorganisms in the film. As an example, apply sanitizer at 800–1,000 ppm and allow it to work for a period of time. Use a clear rinse and reapply sanitizer at the allowable level of 200 ppm without rinsing off. On weekends, apply a quaternary ammonium compound at a level of 800–1,000 ppm and leave it on over the weekend to take advantage of its residual property. Acid-based sanitizers may be used to remove mineral film, and ozone use should also be considered as it is a strong oxidant that acts quickly against a wide array of microorganisms and has not been shown to result in organism resistance.
Inspect: This will be using physical senses, ATP bioluminescence or microbiological testing to verify that the cleaning has been effective.
Current Research
The U.S. Department of Agriculture Agricultural Research Service (ARS) has conducted studies on biofilm formation and composition, including means of prevention and removal. ARS has already determined that a strong negative electrostatic charge to biofilms on stainless steel may reduce bacterial surface contamination. In the study, researchers at the Meat Quality Research Unit found that stainless steel surface-finishing treatments, such as polishing, sandblasting and grinding, reduced buildup of biofilms. They indicated that electro-polishing, placing the stainless steel in an acid bath and running an electric current through the solution, prevented biofilm formation. Bacteria are negatively charged, and the current through the acid media may change the charge on the metal, reducing the ability of bacteria to attach and form biofilms. A study by Dr. Michael Doyle at the University of Georgia Center for Food Safety, funded by the American Meat Institute, has shown that strains of lactic acid bacteria can inhibit growth of Listeria in a biofilm over extended time. The lactic acid bacteria did not grow at 39 ºF but did produce an anti-Listerial metabolite to keep levels of Listeria low.
The future application of the extensive research information may lead to additional methods of both preventing and controlling biofilms. Until then, food manufacturing plants will have to rely on methods that include sanitary design, effective sanitation and monitoring for changes in the production environment.
 Michael Cramer is the senior director, food safety and quality assurance, for Windsor Foods.
Michael Cramer is the senior director, food safety and quality assurance, for Windsor Foods.
2. Deibel, V. and J. Schoeni. 2002/2003. Biofilms: Forming a defense strategy for the food plant. Food Safety Magazine 8:49–54.
3. Mejias-Sarceno, G. 2011. Inadequate sanitation results in biofilm formation. Food Safety Magazine 17:16–18.