New food processing technologies based on high hydrostatic pressure (HP) in combination with high temperature (HT) and pulsed electric field (PEF) treatment of raw materials and food produced thereby have gained increasing attention during recent years. This has also resulted in an integrated EU-supported project “Novel Q” (www.novelq.org), comprising the analysis of potential chemical reactions in the food materials treated with HP/HT and/or PEF techniques.
Background
Processing of fruits and vegetables used for human consumption comprises different technologies used for various purposes. Several technologies are based on heat transfer operations such as microwave and thermal processing for sterilization or pasteurization to reduce problems caused by microorganisms, or post-harvest autolyses such as non-enzymatic and enzyme-catalyzed reactions. The major emphasis of food processing is thus preservation or shelf-life extension. Included herein is the prevention of undesirable changes in the concentration of bioactive components affecting the wholesomeness of the food, its nutritive value, its health-promoting properties and its sensory qualities.
It is, however, difficult to avoid the unwanted chemical reactions affecting food quality parameters when only HT processing is used. Therefore, recently, increasing focus has been devoted to new processing technologies, such as HP, HP/HT and PEF treatment of raw materials and food.[1–5] Introduction of new processing technologies also requires analytical procedures for the determination of which biomolecules are affecting food quality. Such methods or procedures are considered to be important tools for following the chemical reactions and changes in native plant components as a function of processing conditions, and for final documentation of product quality,[6] particularly the identification of biomarkers for specific chemical reactions.
The present work focused on increased knowledge of chemical reactions resulting from the HP/HT and PEF processing of fruits and vegetables. Plant materials of four different genera and families were selected for the studies: apple, carrot, tomato and broccoli.
The overall chemistry of the plant material, aside from its origin and chemo-taxonomical composition,[7] is also defined by the plant part and influenced by several factors, such as growing conditions, ripeness, harvest conditions and post-harvest treatments.[8] Identification of biomarkers requires specific procedures for precise quantification of their concentration and possible fate during chemical reactions.[9–12]
The aim is to analyze compounds with structures that allow chemical reactions to occur as a function of processing conditions. Two approaches were used. Studies to identify specific biomolecules and their transformation products were performed. Since it is likely that not all chemical reactions are measurable by such an approach and some reactions may simultaneously affect the concentrations of several compounds (thereby masking the effects on a single biomarker),[10] analytical fingerprinting was also performed to identify unspecific changes in the concentration of low molecular weight (LMW) compounds present in the tested materials.[13,14] The goal was to identify patterns that may be used to indicate both known and unknown chemical reactions as well as synergistic effects on the LMW compounds that result from both enzymatic and non-enzymatic catalysis.
Results and Discussion
Sampling and Pretreatment of Samples
Among the key problems related to evaluation of chemical reactions resulting from HP/HT and PEF treatment of fruits and vegetables (solids and fluids) are the use of proper sampling techniques. This is caused by variations in individual types and concentrations of native LMW compounds and enzyme activities that often can be appreciable, even among fruit and vegetables of the same plant variety and among different parts of the plants.[6,7]
Processing Conditions
Mild PEF treatments (4 kV/cm) increase cell permeability up to 50% in apples, leading to a decrease in total polyphenolics due to release of external liquid.[15] At 2 kV/cm, the higher permeability caused a three-fold increase in extraction of flavonols from apple skin.[16] Food processing is performed with high-PEF treatment (20–80 kV/cm and short pulses; 1–10 µs) to inactivate microorganisms.[17] The right combination of high electric-field strength and total treatment time will ensure enzymatic inactivation without any over-processing.[18,19] For this investigation, several treatment conditions were applied to carrots and tomatoes. Selected combinations of voltage (0–20 kV) and temperature (20–40 °C) were applied.
Pasteurization relative to HP treatments can be achieved by combining a pressure of between 250–500 MPa and temperatures greater than 50 °C,[20] whereas sterilization is obtained with temperatures above 70 °C.[21] Here, selected combinations of pressures (0–700 MPa) and temperatures (20–110 °C) were used. All processing treatments were performed by The Technical University of Berlin, Germany; The Catholic University of Leuven, Belgium; and University of Lleida, Spain, as partners involved in the EU project “Novel Q.”
Storage after Processing: Autolysis Processes
A critical factor in connection with these analyses is the possibility of a non-enzymatic or enzyme-catalyzed autolysis process[22] following plant membrane destruction and contact between enzymes and substrates in aqueous media. These types of processes are often the main cause of transformations of native LMW molecules into anti-nutritional or toxic compounds, as well as the basis for various types of wanted or unwanted smell, taste, and color changes (browning).[12,13,23] To filter out such effects of processing, the background noise from natural variations must be overcome.
Due to the aforementioned factors, the main variations detected by target analysis and fingerprinting turned out to be a function of sample variation and not of processing at the conditions tested in this study (vide supra).
Internal or External Standards
Quantitative analyses of individual LMW components need internal standards with the same functional groups as those in the compounds considered. If these are not available, it is necessary to use external standards.[6] Internal standards have been used for quantification of analyzed compounds, and they were added to the material prior to extraction. Trigonellinamide, nor-Val, maltitol, 2-deoxy-D-ribose and glucosibarin + glucotropaeolin were used for determination of, respectively, amines/alkaloids, phenolics, amino acids, oligosaccharides, reducing carbohydrates and glucosinolates.
Selection of Specific LMW Biomarkers
Sucrose and the raffinose family of oligosaccharides are markers of non-enzymatic and enzyme-catalyzed hydrolysis, which transforms non-reducing carbohydrates into reducing carbohydrates. Reducing carbohydrates and non-protein-bound amino acids are precursors of potential browning reactions, Maillard/ Heyn’s products, and/or acrylamide formation.[24–26] Natural antioxidants have been used as markers of reactions leading to product color changes. Changes in enzymatic activities have been investigated by assessment of activities and specificities for peroxidase (POD) and polyphenol oxidase (PPO).[12,13,23] The glucosinolates and their products, as well as myrosinases, have been used as efficient biomarkers of processing effects on broccoli.[27] Methods of analysis for some lipid-soluble compounds have been developed as well.[28–33]
Determinations of enzymatic activities in the test materials were based on analyses of enzyme substrates present in the raw plant extracts as a function of time, but also on the determination of enzyme activities in the starting material and in the processed plant materials. More specific and informative enzyme assays, such as on-line micellar electrokinetic capillary chromatography (MECC) determination of substrate degradation and products of myrosinase,[34,35] POD and PPO[12,13] and phenylalanine/tyrosine ammonia lyase (PAL), have recently been developed.[11] Since several POD and PPO substrates were present in the plant extracts, partial purification of the enzymes was needed to remove unknown substrates from the enzyme extracts. This purification was performed by salting out the enzyme proteins. The semi-purified enzyme activities were thereafter determined in assays using specific substrates in known concentrations.[12,13]
Hydrolysis of Sucrose
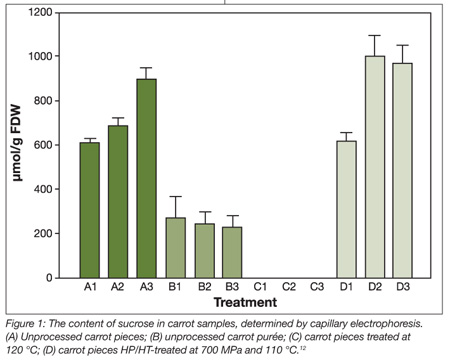 Comparison of sucrose content in carrot pieces and purée indicates that sucrase activity occurs in the purée in which a lower content of sucrose is detected (Figure 1). In carrot pieces processed at 120 °C, sucrose had been degraded completely in non-enzyme-catalyzed hydrolysis, due to the thermodynamically unstable glycoside bond. However, applying pressure of 700 MPa at 110 °C did not affect the sucrose level in the carrot pieces.
Comparison of sucrose content in carrot pieces and purée indicates that sucrase activity occurs in the purée in which a lower content of sucrose is detected (Figure 1). In carrot pieces processed at 120 °C, sucrose had been degraded completely in non-enzyme-catalyzed hydrolysis, due to the thermodynamically unstable glycoside bond. However, applying pressure of 700 MPa at 110 °C did not affect the sucrose level in the carrot pieces.
Browning Reactions, Maillard/Heyn’s Products and/or Acrylamide Formation
HT treatment of fruits and vegetables may also affect the content of reducing carbohydrates and both the free and protein-bound amino acids by browning reactions (e.g., the Maillard reaction) or the formation of Heyn’s products. In some applications, these reactions are required to ensure the right color or the right taste or smell, as in caramel or during frying. On the other hand, maintaining freshness of fruits and vegetables requires preventing the formation of such reactions. Here, reactions of this kind were not identified, indicating that the processing conditions used for the HP/HT and PEF treatments were sufficiently mild to avoid Maillard reactions and formation of Heyn’s products.
Analysis of free amino acids in the analyzed fruit and vegetable samples revealed that asparagine was the most abundant free amino acid in the samples. A reaction between carbohydrates and free asparagine is the initial step in the formation of acrylamide[24–26,36] and has been shown to occur in heated foodstuffs.[37] Relatively high concentrations of unbound glucose were also present, thus the precursors of acrylamide formation were present in the samples. The results from glucose and asparagine analyses showed no indication of acrylamide formation during HP/HT or PEF treatment under the processing conditions in this study. The formation of acrylamide only takes place at very high temperatures (< 120 °C)[36,38] in accordance with the present results, even though the temperature at or near the electrodes in the PEF treatment chamber may be higher than the average sample outlet temperature.[39]
Polyphenols, POD and PPO
PPO and POD are responsible for enzymatic browning in fruits and vegetables or dark color formation different from the aforementioned browning reactions.[40] PPO, however, in most cases, is more sensitive to heat and PEF treatment than POD.[41] To distinguish between the activity and specificity of these two enzymes, a specific assay has been developed.[13,23] High-intensity PEF treatment (35 kV/cm for 1,500 µs, applying 5.5-µs bipolar pulses at 200 Hz) of tomato juice resulted in a 96.6 % inactivation of POD, and complete inactivation of PPO.[23]
In carrots (Nepal) and apples (Golden Delicious), the qualitatively dominant phenolic compound was chlorogenic acid.[12] With chlorogenic acid as a specific substrate for PPO, its on-line MECC determination makes it an efficient biomarker for processing effects,[13] and thereby an indirect marker of PPO activity.
Glucosinolates and Glucosinolate Degradation Products
In broccoli samples, cell disruption leads to reactions between myrosinase and glucosinolates, which can result in a great number of transformational products, depending on pH, metal ions and other reaction conditions.[27,34,35,42] Among the reactive degradation products are the isothiocyanates, some of which are potential anticarcinogenics.[43] HP/HT treatments under mild conditions appear to have a significant effect on cell permeabilization without affecting myrosinase activity.[27] Hence, HP/HT treatments (100–500 MPa) can induce enzymatic hydrolysis of glucosinolates and thereby yield isothiocyanates, some of which are health-promoting[27] and some of which are anti-nutrients.[44]
Analytical Fingerprinting of LMW Compounds
Analytical fingerprinting was based on chemometric analysis of electropherograms from capillary electrophoresis of LMW raw extracts of the various processed and unprocessed plant materials. Principal component analysis (PCA) was performed to see whether specific degradation patterns could correlate to the processing parameters. PCA analyses of data on all water-soluble LMW compounds detected by MECC from HP/HT processing of carrots led to a marker for thermal treatment. This marker, still unidentified, appeared in high concentrations in samples treated with temperatures at or above 110 °C and was not affected by application of pressure.[14]
Conclusions
Chemical reactions in processed fruits and vegetables occur in native matrix systems at room temperature and atmospheric pressure, when enzymes and their substrates come into contact in appropriate reaction media. The biomolecules/LMW components and their related enzymes are physically separated by membranes in the undamaged tissues. However, under some processing conditions, enzymes and their substrates are mixed, and this provides the basis for the initiation of autolysis processes—that is, degradation or transformation of the native components in non-enzymatic or enzyme-catalyzed reactions.
Food processing based on heat transfer processes is commonly used and adapted to create a desired food quality. The use of HT in food processing also has some drawbacks, including unwanted chemical reactions that impact the quality, texture and freshness of the food ingredients.
The novel processing technologies HP/HT and PEF considered in this work operate with relatively low temperatures and thereby heat transfer, but still under conditions where unwanted chemical reactions can occur. This means that the components and their chemical reactions are different for the various plants and plant families considered. However, it seems reasonable that neither PEF nor HP/HT induce unknown types of reactions, but only those reactions associated with the ubiquitous compounds and allelochemicals/xenobiotica in the matrix systems.
In connection with studies of effects on enzymes and chemical reactions from HP/HT and PEF treatments of the studied fruits and vegetables, highly specific methods for on-line determination of substrate transformation and the simultaneous production of products have been developed. This has been the case for the enzyme systems PAL, PPO, POD and myrosinase. High-intensity PEF (35 kV/cm, 150 µs, 5.5-µs bipolar pulses at 200 Hz) treatment of tomato fruit juice revealed 96.6% inactivation of POD and complete inactivation of the more labile PPO. Mild PEF treatment (2–4 kV/cm) resulted in increased permeability of LMW phenolic compounds through the membranes. It was also found that mild HP/HT seems to have a beneficial effect on broccoli cell permeability of glucosinolates without affecting myrosinase activity. HP/HT (100–500 MPa) can, however, induce myrosinase-catalyzed hydrolysis of glucosinolates in cruciferous vegetables.
Analytical determination of the individual native molecules in the considered matrix systems—including their potential reactions and final products in the processed food products—is considered a useful tool for the development of novel processing technologies (HP/HT and PEF) and their introduction into production units. ♦
The authors of this article participate in the EU-funded “Novel Q” project, which aims to remove hurdles to the application of novel food processing methods. The authors wish to thank their partners—the research groups of Professor Dietrich Knorr, The Technical University of Berlin, Germany; Professor Marc Hendrickx, The Catholic University of Leuven, Belgium; and Professor Olga Martin-Belloso, University of Lleida, Spain—for the processed samples they provided for this study. Financial support from the EU Commission in relation to the project FP6 “Novel Q” 015710-2 is kindly acknowledged.
Keld Ejdrup Andersen, Ph.D.; Heidi Blok Frandsen, M.S.; Jens Christian Sørensen, Ph.D.; and Hilmer Sørensen, Ph.D., are in Biochemistry and Natural Product Chemistry, Department of Basic Sciences and Environment, Faculty of Life Sciences, University of Copenhagen, Thorvaldsensvej 40, DK-1871 Frederiksberg C, Denmark.
1. Zhang Q., G. V. Barbosa-Canovas, and B. G. Swanson 1995. Engineering aspects of pulsed electrical field pasteurization. J. Food Engineer. 25:261-281.
2. Doernenburg H. and D. Knorr. 1998. Monitoring the impact of high-pressure processing on the biosynthesis of plant metabolites using plant cell cultures. Trends Food Sci. Technol. 9:355-361.
3. Wouters P. C., I. Alvarez, and J. Raso. 2001. Critical factors determining inactivation kinetics by pulsed electrical field food processing. Trends Food Sci. Technol. 12:112-121.
4. Góngora-Nieto M. M., D. R. Sepulveda, P. Pedrow, G. V. Barbosa-Canovas, and B. G. Swanson. 2002. Food processing by pulsed electrical field: Treatment delivery, inactivation level, and regulatory aspects. Lebensmittel Wissenschaft und Technologie. 35:375-388.
5. Ludikhuyze L., A. Van Loey, S. C. Indrawati, and M. Hendrickx. 2003. Effects of combined pressure and temperature on enzymes related to quality of fruits and vegetables: from kinetic information to process engineering aspects. Crit. Rev. Food Sci. Nutri. 43:527-586.
6. Sørensen H., S. Sørensen, C. Bjergegaard, and S. Michaelsen. 1999. Chromatography and Capillary Electrophoresis in Food Analysis. 1st Ed. (Royal Society of Chemistry, UK).
7. Sørensen H., J. C. Sørensen, and S. Sørensen. 2001. Phytochemicals in food. The plant as chemical factories. In: Biologically Active Phytochemicals in Food. Analysis, Metabolism, Bioavailability and Function. (The Royal Society of Chemistry, UK), pp. 3-12.
8. Hansen M., P. Møller, and H. Sørensen. 1995. Glucosinolates in broccoli stored under controlled atmosphere. J. Am. Soc. Hortic. Sci. 120:1069-1074.
9. Andersen, K. E., C. Bjergegaard, P. Møller, J. C. Sørensen, and H. Sørensen. 2005. Compositional variations for α-galactosides in different species of Leguminosae, Hordeum and Brassicaceae – A chemotaxonomic study based on chemometrics and high performance capillary electrophoresis. J. Agric. Food Chem. 53:5809-5817.
10. Petersen I. L., H. C. B. Hansen, H. W. Ravn, J. C. Sørensen, and H. Sørensen. 2007. Metabolic effects in rapeseed (Brassica napus L.) seedlings after root exposure to glyphosate. Pestic. Biochem. Phsiol. 89:220-229.
11. Andersen K. E. H. B. Frandsen, J. C. Sørensen, and H. Sørensen. 2009. PAL activity assay based on online determination of Phe and cinnamate. In Proceedings of EuroFoodChemXV Congress, Copenhagen. pp. 16-19.
12. Andersen K. E., et al. 2009. Chemical reactions in fruits and vegetables as a function of processing. . In Proceedings of EuroFoodChemXV Congress, Copenhagen. pp. 20-23.
13. Andersen, K.E., Frandsen, H.B., Sørensen, H., Sørensen, J.C. and Sørensen, S. 2007. On-line electrokinetic capillary chromatography determination of polyphenol oxidase activity and specificity in fruits and vegetables. In: Proceedings of EuroFood Chem XIV Conferences, Paris, August 29-31, 2:508-511.
14. Andersen K.E., Frandsen H.B., Van der Plancken I.,Grauwet T., Hendrikx M., Sørensen J.C., and Sørensen H. 2009. Combined MECC and PCA fingerprint analyses as a tool for revealing marker molecules for novel HP/HT processing of carrots. . In Proceedings of EuroFoodChemXV congress, Copenhagen (ISBN 978-87-993033-4-2) 5-8 July (2009) II, 269-272.
15. Balasa A., et al. 2009. Pulsed electric fields processing and its potential to induce stress in plant systems. In Proceedings of EuroFoodChemXV Congress, Copenhagen. pp. 198.
16. Turk M., E. Vorobiev, and A. Baron. 2009. Pulsed electric field of apple peels improved the extraction of flavonol components by diffusion at mild temperature. In Proceedings of EuroFoodChemXV Congress, Copenhagen. pp. 3-6.
17. Mosqueda-Melgar J., R. M. Raybaudi-Massilia, and O. Martín-Belloso. 2008. Combination of high-intensity pulsed electric fields with natural antimicrobials to inactivate pathogenic microorganisms and extend the shelf-life of melon and water melon