The purpose of food safety programs should be to keep harmful agents, that is, chemicals, allergens, extraneous matter (insects, metal filings, rubber, plastic, etc.) and microbes out of the food supply. This article will focus entirely upon enhancing control of pathogenic bacteria in foods by showing the need for improvement in food safety audits as they relate to microbiological risk.
It is often stated, and likely true, that the United States has the world’s safest food supply. Nevertheless, the Centers for Disease Control and Prevention (CDC) have estimated that every year in the United States there are approximately 76 million cases, 325,000 hospitalizations and 5,000 deaths as a result of foodborne disease.[1] The number of multistate Class I recalls has grown over the past decade, despite apparent improvements in reducing the number of foodborne disease events with several different microorganisms. This is a consequence of improved surveillance capability throughout our nation as local, state and federal agencies share information in the context of FoodNet and PulseNet.
Added to the cost in human suffering from foodborne illness, expensive recalls may also occur when food-processing companies become aware that their product is contaminated with a pathogenic microorganism. If such product results in illness, lawsuits from stricken individuals (including class action suits) and other business losses (and possible lawsuits) from shorted customers may also result. Product contamination events typically result in a plant’s suspending production on implicated lines—sometimes for many weeks—until contamination sites are determined and eliminated, their entire food safety system overhauled and appropriate repairs and modifications to equipment and facilities are made before restarting production. The resulting costs of these activities are often in the millions of dollars, not to mention other losses due to public embarrassment and loss of market share. Consequently, the food industry has implemented a number of approaches to food safety inspection through the years, including finished product testing, good manufacturing practices (GMPs) and Hazard Analysis Critical Control Point (HACCP) verification audits. Each of these approaches has its own flaws but forms a part of a larger picture of food safety.
Finished Product Testing
 Finished product testing is a type of inspection. Prior to widespread understanding and implementation of HACCP in the United States, many companies relied extensively upon end-product holds and testing to provide assurance of the microbiological safety of their products. There is a flaw in this approach.
Finished product testing is a type of inspection. Prior to widespread understanding and implementation of HACCP in the United States, many companies relied extensively upon end-product holds and testing to provide assurance of the microbiological safety of their products. There is a flaw in this approach.
The incidence of foodborne contamination that occurs in the industry is likely to be fairly low. A published example of this is found by Cutter and Henning[2] who reported incidences of Listeria monocytogenes between about 1 and 6% in various ready-to-eat (RTE) meat products (Table 1).
L. monocytogenes is considered by many to be a ubiquitous pathogenic organism in the natural environment and can be found in many food-processing environments.[3] Frequent isolation of Listeria spp. from meat-processing environments has resulted in a viewpoint that complete elimination of this organism from RTE meat and poultry processing environments may not be possible with current technology.[4] Consequently, plant environmental, and therefore processed product, contamination with other less environmentally prevalent pathogens (e.g., Salmonella) can be expected to be a good deal lower.
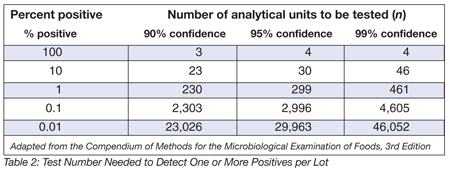 Table 2 below shows that if 1% of a given manufacturing production lot is contaminated, one would need to take at least 299 samples to have a 95% chance of recovering a single positive. It should be pointed out that these tables are based on assumptions of homogeneous contamination, perfect test methods and other considerations that do not reliably occur in the real world. In my experience, finished product contamination is nearly always from the post-processing (e.g., post-CCP) environment[5,6] and is often sporadic.
Table 2 below shows that if 1% of a given manufacturing production lot is contaminated, one would need to take at least 299 samples to have a 95% chance of recovering a single positive. It should be pointed out that these tables are based on assumptions of homogeneous contamination, perfect test methods and other considerations that do not reliably occur in the real world. In my experience, finished product contamination is nearly always from the post-processing (e.g., post-CCP) environment[5,6] and is often sporadic.
Clearly, one cannot rely on finished product testing to guarantee food safety. This is one reason why appropriate implementation of HACCP is important. However, there is a role to be played by finished product testing in the context of HACCP plan verification (HACCP principle No. 7). Nevertheless, finished product testing is one small piece of a much larger food-safety puzzle.
HACCP Verification Audits
HACCP is regarded by many as the pinnacle of the food safety system built upon laws and regulations, GMPs and Sanitation Standard Operating Procedures (SSOPs), respectively. The principles of HACCP are well known and include:
1. Hazard Analysis and Identification of Preventatives
2. Identification of Critical Control Points (CCPs)
3. Establishment of Critical Limits (CL) for CCPs
4. Monitoring of CCPs
5. Corrective Actions for Critical Limit Deviations
6. Establishment of Record Keeping System
7. Establishment of Verification Procedures
However, serious risks can be missed when one seeks to audit HACCP in isolation from the rest of a plant’s food safety system. The importance of recontamination of food after a CCP cannot be underestimated.[7] I recall one checklist for a HACCP verification audit many years ago of a company that received previously unwashed tomatoes in field-soiled boxes. These boxes of tomatoes were placed in a controlled atmosphere for ripening, removed at the appropriate time, and the tomatoes were placed on a conveyor that transported them through a proprietary machine that washed the tomatoes. Critical operating parameters for this machine (temperatures and chemical strength) were identified as CCPs that were appropriately controlled and monitored. Subsequently, the tomatoes were placed, by a worker with a large, cloth, blood-stained “bandage” over a cut finger, back into the soiled boxes. A review of the plant records indicated that it had fulfilled all seven elements of HACCP. No objective observer would argue that the product was free of environmental contamination. Clearly there was a failure of appropriate GMP-based prerequisite programs. Nevertheless, our observations were disputed as irrelevant to the HACCP verification. This highlights the importance of combining the two approaches and takes us back to GMPs and GMP auditing.
GMPs and GMP Auditing
GMPs are described in the Code of Federal Regulations (21 CFR 110). Establishment of GMPs is considered prerequisite to HACCP. The industry typically interprets these items and develops audits for them. Common elements that are reviewed in such audits follow:
• Buildings and Grounds
• Equipment Controls and Maintenance
• Pest Control Programs
• Employee Practices
• Operational Practices and Procedures
• Raw Materials
• Processing
• Packaging
• Storage and Distribution
• Sanitation, Cleaning and Housekeeping
• Temperature Control Methods
I have observed situations in which companies receive high GMP audit scores but still have very challenging microbiological control issues, resulting in potential contamination risk to finished product. This was dramatically proved to me over a decade ago when I spent 1.5 days doing a very thorough GMP audit with a colleague. We took a lot of time reviewing records, procedures and programs—in short, a lot of “paper chasing” —but spent a comparatively small amount of time walking through the plant. No sampling of the plant environment was done. The plant scored 95% and had no “automatic failures.” Subsequent to the audit, plant personnel indicated that they had a struggle with endemic Salmonella and asked that we stay another 3.5 days to investigate the situation, which we did. We went through the operations very carefully, sampling many things including but not limited to traffic patterns (e.g., forklift wheels, wet tracks made by wheeled vehicles, workers’ hands/gloves, workers’ shoes and shoe coverings, footbaths, floor mats and carpet tops and undersides, etc.), equipment after rigorous teardown, underneath various areas (e.g., product conveyors) and other equipment previously missed in our GMP audit. We also sampled broken, wet insulation, dust accumulations, partially caulked and rusted seams for bulk receiving tanks, drains, air handling unit catch pans, coils and “p” traps. In short, we took samples of the plant in a comprehensive manner. Our focus was “investigatory.”
The contrast between the two approaches was dramatic. One approach (the GMP audit) relied upon filling out a checklist with heavy attention on paperwork documenting the existence of programs (not necessarily their performance). The other approach was investigational; for example, observations and sampling were done of likely microbial growth niches in the plant environment and our thinking about routes of transmission heavily emphasized time spent physically in the plant. (These niche sites result from unsanitary operations, maintenance/repair practices and equipment and facility design.) This year, I had a couple of similar experiences in facilities where several third-party auditors from different auditing firms scored the food processing plants very high, yet my investigational approach revealed serious opportunities for product contamination with endemic pathogens. How can this be?
I think there is more than one answer to this question, but I propose the following reasons based on observation:
1. Checklist-based audits can be ineffectual when they are approached with what I call a “checklist mindset.” A checklist mindset argues against an investigational mentality. To explain, we live in an ever-increasingly fast-paced society. Sometimes I am asked, “How long will it take to investigate this production line?” An individual with an investigatory mindset will not be able to answer the question definitively. A true investigator does not know ahead of time what he/she will turn up or how difficult the sampling process may be, nor will he/she know all the equipment that will need to be broken down for sampling and how those activities will impact the timeframe of the audit/investigation. A checklist mindset results in an expedient audit that is over when the checklist is completed, allowing the auditor and selected plant personnel to move on to other responsibilities. A third-party auditor or his/her employer may have an incentive to move on to another audit and thus be able to bill another job. The faster the audit is accomplished, the more jobs can be done. If audit pricing is on a per-audit basis rather than on a time (e.g., per hour) basis, then a clear incentive exists for the auditor to finish the audit quickly. Another incentive to finish checklists quickly occurs when the auditor has a quota of audits that must be accomplished in a given timeframe. Such audits have little incentive built into spending the necessary time observing sanitation, plant/line start-ups, operations and plant/line shut-downs to think about and investigate the microbial ecology of a food manufacturing facility and its potential risk to food contamination. Hence, a lot of potential risks can be missed. Auditors who have booked multiple companies in the same week have an incentive to finish in time to meet their travel schedule and commitments to other plants.
2. Many checklist-based audits result in a great deal of looking at appropriate paperwork (e.g., policies, SOPs) while de-emphasizing time in the plant. Again, important in-plant observations can be missed. For example, the written SSOP, the sanitation approach described by the sanitation supervisor and the one that actually occurs in the plant are sometimes three different things in my experience! All three should be assessed in a comprehensive risk assessment. Be wary of an auditor that does not use a flashlight or bend down to look underneath equipment during their audit. He/she could have a “checklist mindset.”
3. There are substantial differences between auditors. Some have a great deal of experience, some do not. Some auditors have experiences in one industry that are employed auditing other industries. Try to use one who has experience in your industry. This is not always possible. But try to get one who cares about your facility, has experience with in-plant microbial control and is willing to spend the time to observe and think about the potential risks in the plant.
4. Audit scores have been used as performance criteria for plant managers and other personnel. Hence they have become sources of discussion and negotiation during the auditor’s exit review. Clarification of issues is good, but negotiation of food safety scores can be a “slippery slope.” A walk-through microbiological risk assessment, on the other hand, need not be scored. A plant with an audit score in the high 90th percentile out of a possible 100% may look great on paper but that score is worthless if the plant has endemic Salmonella or L. monocytogenes in Zone 2 (near product surfaces) and its products are at significant risk of contamination. Having the right systems is a good thing, but is less important than if those systems are really controlling the plant environment.
The Missing Element
If finished product testing, checklist mindset GMP auditing and similar approaches to HACCP verification audits are not the answer, then what is? I do not advocate that finished product testing, GMP audits and HACCP audits be eliminated. These are all portions of a much larger food safety system that all plants should embrace. We should not “throw out the baby with the bathwater.” I am proposing strengthening these audits so as to get a better idea of actual microbiological food safety risk in the food processing plant environment. In my view, a better measurement of microbiological food safety would occur if a food processor were to combine a comprehensive food safety system review with an in-factory walk-through “risk assessment,” including environmental sampling for appropriate indicator organisms.
Microbiological Walk-through Risk Assessment Approach
With this approach, the auditor carefully observes plant operations (e.g., cleaning and sanitation, startup and operations) and takes appropriate samples. Given legal liabilities associated with sampling and disposition of the product, these samples could be tested for appropriate indicator organisms. However, it seems appropriate that if plant personnel know there could be potential for Salmonella contamination, then Zone 3 and 4 samples may be considered for such testing by an outside laboratory. However, a great deal of information can also be gained with simple tests [e.g., aerobic plate counts (APCs), Enterobacteriaceae (EB) counts, etc.]. These “indicator” tests can provide a good measurement of the efficacy of sanitation and control of the process and environment during operations. For example, an effectively sanitized surface should have undetectable EB, as these organisms are easily killed by sanitizers. Other indicators could include a test for “Listeria-like” microbes in RTE meat-processing facilities. These organisms are recoverable after enrichment and streaking onto modified Oxford or other appropriate media. Coliform testing in plants that make non-sterile dry infant formula is also of value, given this industry’s concerns about Enterobacter sakazakii (a coliform bacterium).8 The presence of an APC of 100,000 on a large (e.g., 1-ft2 area) environmental surface is likely to be reflective of an area where microbial control efforts have failed and microbial growth has occurred. The choice of test(s) will be dependent on the site and its relative risk to the product, with appropriate consideration given to the disposition of the finished product. This approach calls for an experienced auditor with an understanding of likely microbiological risks associated with the type of production environment being audited and the implications of such testing on the final disposition of the finished product. The testing to be done should be discussed and agreed upon with the plant management before the samples are sent to an outside laboratory. The tests selected may need to be determined after the auditor has walked through the plant at least once.
In-plant Microbiological Risk Assessments
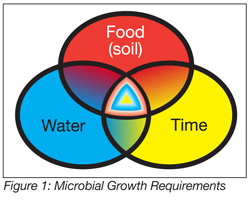 The approach I am advocating is quite different than the way the term is often used in academia. It has little to do with statistics and probability, but a great deal to do with observations in the plant related to where microbes have grown in localized niches. It is certainly impractical, and likely impossible, to expect food-processing facilities to be sterile. However, microbial growth can be controlled. Microbial growth niches occur when moisture, nutrition, time and growth-conducive temperatures occur in a non-sterile environment (Figure 1). Adequate nutrition for microscopic creatures is typically in abundance in food-processing environments. Time abounds, particularly in areas missed or inaccessible, for cleaning. Microbial growth can occur over a wide range of temperatures between approximately 32 and 140 °F. Moisture is absolutely necessary for microbial growth, hence, its control is essential. Sampling sites should be selected where it is likely that moisture is entrapped (e.g., wet insulation, roof leaks, water marks, hollow conveyor rollers, duct-taped pipe leaks, sandwiched areas associated with equipment, such as slicers, conveyor guides, product scrapers, etc.). A list of such sites related to Listeria niches is found in Kornacki and Gurtler.[3]
The approach I am advocating is quite different than the way the term is often used in academia. It has little to do with statistics and probability, but a great deal to do with observations in the plant related to where microbes have grown in localized niches. It is certainly impractical, and likely impossible, to expect food-processing facilities to be sterile. However, microbial growth can be controlled. Microbial growth niches occur when moisture, nutrition, time and growth-conducive temperatures occur in a non-sterile environment (Figure 1). Adequate nutrition for microscopic creatures is typically in abundance in food-processing environments. Time abounds, particularly in areas missed or inaccessible, for cleaning. Microbial growth can occur over a wide range of temperatures between approximately 32 and 140 °F. Moisture is absolutely necessary for microbial growth, hence, its control is essential. Sampling sites should be selected where it is likely that moisture is entrapped (e.g., wet insulation, roof leaks, water marks, hollow conveyor rollers, duct-taped pipe leaks, sandwiched areas associated with equipment, such as slicers, conveyor guides, product scrapers, etc.). A list of such sites related to Listeria niches is found in Kornacki and Gurtler.[3]
Gabis and Faust[9] wrote that “the probability of product contamination from the environment is dependent upon a number of variables…” They listed those as follows:
1. Proximity of microbial growth niches to the product stream
2. Number of niches in the factory
3. Spatial relationships of niches and the product stream
4. Microbial population in niches
5. Degree of niche disruption during operations
6. Exposure of the product stream to the environment
Consequently, sites conducive to growth (e.g., where moisture has collected or been entrapped) can be broken down into those areas which are of high, medium and indirect risk of contaminating the product stream based upon observations and testing. These areas can be defined as follows:
High risk – an area or practice that may directly contaminate the product from a microbial growth niche either continually or intermittently.
Medium risk – similar to “high risk,” but mitigating factors (e.g., further heat processing) may reduce risk by an undetermined amount.
Indirect risk – any situation or condition likely to develop into a microbial growth niche (e.g., standing water in a hallway) that may migrate to another site and become a growth niche capable of inoculating product at a detectable level.
This approach causes the auditor to focus his or her observations in the plant on likely contamination risks to finished product. It is important to recognize that test results from samples taken in a plant are, at best, merely a snapshot of microbiological conditions at those sites at the moment of sampling. Consequently, data from testing of such samples taken should not weaken the force of the initial observation, since a high-risk condition may require more time after the sample is taken to develop the microbial populations that may result in direct measurable contamination of product. However, the data may reveal areas that were not obviously a risk during the initial walk through. In my experience, the biggest surprises often come in the form of pre-operational surface sampling results. Oftentimes, high counts may be found on surfaces that look visibly clean and dry after cleaning and sanitization and before product startup. Frequently, this phenomenon results from inadequate use of hot water during cleaning, something that may or may not have been caught during the observation of sanitation.
In summary, current checklist approaches may not identify actual microbiological risks, especially when the auditor is not sampling for them. A walk-through microbiological risk assessment with environmental sampling and testing would provide valuable information about the relative risk of a process and should be used to supplement the current checklist-based approaches. Such an approach could help avoid the types of food safety surprises we have seen in recent years.
The next issue of this publication will discuss how to avoid some of the pitfalls of the walk-through risk assessment approach.
Jeffrey L. Kornacki, Ph.D., is President of Kornacki Microbiology Solutions, Inc., a McFarland (Madison), WI-based company. Dr. Kornacki has performed in-factory microbiological investigations, risk assessments and food safety-related audits for 20 years in approximately 500 food-processing facilities in a variety of capacities with different organizations. Between 2001 and 2003, he was an Assistant Professor at the University of Georgia’s Department of Food Science and Center for Food Safety prior to starting several food safety consulting companies. He remains an Adjunct Assistant Professor in the University of Georgia’s Department of Food Science and is also Adjunct Faculty with Michigan State University’s National Food Safety & Toxicology Center. He has researched, published and spoken widely on pathogen control, testing and sampling in food production facilities. Kornacki Microbiology Solutions (KMS) provides in-plant services including microbiological risk assessments, troubleshooting and CCP validation. They also develop applied microbiology research protocols, offer public and private short courses and provide expert witness testimony for their clientele. They can be found at www.kornackifoodsafety.com.
References
1. Mead, P. S., et al. 1999. Food-related illness and death in the United States. Emerging Infectious Diseases 5:607-625.
2. www.fsis.usda.gov/OPPDE/Nis/Outreach/Listeria.htm#Top.
3. Kornacki J. L. and J. B. Gurtler. 2007. Incidence and Control of Listeria in Food Processing Facilities, in Listeria, Listeriosis and Food Safety, 3rd Ed. Edited by Ryser, E. T. and E. H. Marth. Boca Raton, FL: CRC Press.
4. Tompkin, R. B., V. N. Scott, D. T. Bernard, W. H. Sveum, and K. S. Gombas. 1999. Guidelines to prevent post-processing contamination from Listeria monocytogenes. Dairy Food Environ Sanit 19:551-562.
5. Allan, J. T., Z. Yan, and J. L. Kornacki. 2004. Surface material, temperature, and soil effects on the survival of selected foodborne pathogens in the presence of condensate. J. Food Prot 67:2666-2670.
6. Allan, J. T., Z. Yan, L. L. Genzlinger, and J. L. Kornacki. 2004. Temperature and biological soil effects on the survival of selected foodborne pathogens on a mortar surface. J. Food Prot 67:2661-2665.
7. Reij, M. W. and E. D. Den Aantrekker. 2004. Recontamination as a source of pathogens in processed foods. Int J Food Microbiol 91:1-11.
8. Gurtler, J. B., J. L. Kornacki, and L. R. Beuchat. 2005. Enterobacter sakazakii: A coliform of increased concern to infant health. Int J Food Microbiol 104:1-34.
9. Gabis, D.A. and R.E. Faust. 1988. Controlling microbial growth in the food-processing environment. Food Technol Dec:81-82, 89.