Imagine an orange juice that has been stabilized exclusively by high pressure pasteurization (HPP). The finished juice product will likely present with a fresher taste and a more complete nutritional profile than a comparable but thermally stabilized equivalent. Perfect! This is what the juice company’s customers want—the flavor, texture and color of minimally processed food along with a strong nutritional profile for value-added health benefit. Even better, the high pressure technique not only preserves the desired quality attributes of the product but also kills vegetative bacteria that pose potential spoilage and microbiological food safety hazards. With this non-thermal food preservation method, the juice processor also receives the potential added benefit of increased shelf-life of the product and inactivation of potential pathogens that can cause foodborne illness.
And while there is abundant scientific evidence that HPP and other novel non-thermal processing technologies, most recently pulsed electric field (PEF) systems, in commercial use do provide these benefits to food and bev-erage manufacturers, how does one know with a high level of confidence that the process does not introduce unforeseen negative side-effects in terms of food safety parameters?
The fact is that a significant number of new technologies have emerged in recent years that appear to hold the promise of providing alternatives to heat as a primary means for food preservation. Included among these novel methods are cold plasma, HPP, high intensity pulsed light and PEF. However, before these alternative methods can be used for the mass production of foods, they require validation. That is, the novel process and products manufactured using it must be shown, by objective science and with a high degree of confidence, to be safe for human consumption.
The Case for Process Validation
Process validation within the food processing industry is not an everyday occurrence. As a matter of fact, it is so rare that most food scientists spend an entire career without participating in a process validation team. In general, process validation is reserved for breakthrough technologies or disruptive processing innovations. Research and development outcomes in the food industry generally don’t fit these descriptions. Line-extensions or the repositioning of established product lines don’t require the rigorous scrutiny that defines process validation. Line extensions, more often than not, are based on an existing product or technology that has previously undergone validation. For example, an aseptic canning process or a sterilization procedure will have been validated prior to its initial use. Therefore, line extensions produced or manufactured using these methods would not require validation; rather they require some form of verification.
When a food industry scientist has had experience with process validation techniques, it’s likely that this experience was acquired in the context of conducting a product recall or as a result of participating on a Hazard Analysis and Critical Control Point (HACCP) program team. During a product recall, the collection and analysis of process and product data, along with the other measures undertaken in an effort to understand how, where and when a process may have failed, involve elements of retrospective process validation. During the past 15 to 20 years, ever-increasing numbers of food scientists have become acquainted with process validation methods due to involvement with the HACCP system. HACCP requires that the development teams validate the HACCP plan prior to its introduction onto the factory floor. Typically though, food industry personnel are more likely to be involved with process verification activities rather than validation procedures. The differences between these methods are enormous.
Process validation, according to the U.S. Food and Drug Administration (FDA), requires the establishment of documented evidence which provides a high degree of assurance that a specific process will consistently produce a product meeting its pre-determined specifications. Process validation is generally understood to indicate that a process, procedure or method has been subjected to such a level of scrutiny that its outcomes are predictable. By contrast, process verification, again, according to FDA, is achieved through confirming by examination and provision of objective evidence that specified requirements of a process or procedure have been fulfilled.
In other words, process verification demands objective data that will confirm that a process is operating within predetermined limits or specifications. The differences between validation and verification are subtle and often confusing. Succinctly, process verification should only occur after a process has been properly validated. This point of difference is clearly illustrated by the monitoring activities that are an integral and essential element of the HACCP system. Collecting and examining the data emanating from a critical control point (CCP) constitutes the act of providing objective data to confirm that the critical limits, the predetermined food safety specification, for the CCP are being met. Monitoring, verification in this case, assumes that the established critical limits have previously been validated.
Consider the impact or possible outcomes of a HACCP system in which a critical limit has been arbitrarily established and not properly validated. For example, during pressure assisted thermal sterilization (PATS) of foods, the product’s initial temperatures (IT) in concert with a specified applied pressure are critical to attainment of sterilization temperatures. Therefore, if the critical limit for product initial temperature is established, capriciously at a minimum of 100°C (212°F) when the specified operating pressure is 380 mpa, it is highly unlikely that this pressure temperature combination will yield the desired sterilization temperature of 121°C. A validation procedure developed around these critical process parameters would have revealed an insurmountable flaw in these specifications and demonstrated the instability of the process. Furthermore, validation would have shown the proposed pressure assisted thermal sterilization process to be at risk of producing toxic food. The capability of the critical limits with respect to mitigating the identified hazard must be validated before they can be verified.
By way of another example, consider the efficacy of the cleaning procedures that are routinely used for treating surgical instruments. Historically, the procedures have targeted the inactivation of those bacteria and viruses that are frequently implicated in the transmission of nosocomialy acquired diseases. Contemporary literature, on the subject of hospital acquired diseases, report 90,000 deaths per year by this mode. Contaminated surgical equipment is implicated as a major contributor to this morbidity. Due to the emergence of prion diseases, new prion-specific cleaning procedures are required.
Prions are much more robust and resistant to cleaning and sanitizing agents than bacteria and viruses. Owing to the pernicious nature of prion diseases, and the likelihood of their transmission via contaminated surgical instruments, it is imperative that cleaning procedures can be relied upon to consistently diminish this risk. This outcome can only be assured with the use of validated prion-specific cleaning procedures. In a much heralded case, the surgery of a major U.S. hospital is reported to have used conventional cleaning procedures for treating prion-contaminated instruments and thereby exposed a significant number of surgery patients to prion-mediated disease. Subsequent to this incident and on receiving reports of similar episodes, the World Health Organization intervened by proposing new procedures that had been shown, by way of validation, to be effective in controlling the spread of prion disease in the surgery.
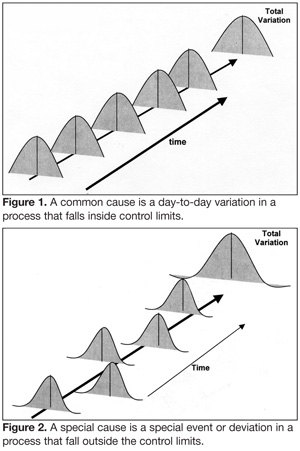 Process validation is the application of scientific methods to a process or procedure in an effort to illuminate the nature and sources of variation inherent in that process. Processes are classified as either stable or unstable base on variation (Figures 1 and 2). Excessive variation occurring outside either the upper or lower specification limits characterize an unstable, or special cause, process. An unstable process is unreliable and prone to failure. By contrast, stable processes, also referred to as common cause processes, exhibit variation around an average that does not exceed specification limits. Stable processes are exceedingly reliable.
Process validation is the application of scientific methods to a process or procedure in an effort to illuminate the nature and sources of variation inherent in that process. Processes are classified as either stable or unstable base on variation (Figures 1 and 2). Excessive variation occurring outside either the upper or lower specification limits characterize an unstable, or special cause, process. An unstable process is unreliable and prone to failure. By contrast, stable processes, also referred to as common cause processes, exhibit variation around an average that does not exceed specification limits. Stable processes are exceedingly reliable.
Another point about stable and unstable processes is that they are frequently confused, one for the other. Dr. Deming, the great process control guru, has reported two kinds of mistakes: a bad mistake is reacting to a variable outcome as if it came from a special cause, when in truth it is the result of a common cause process; a catastrophic mistake is reacting to an outcome as if it came from a common cause process, when in actually it is derived from a special cause process. Mistaking an unstable process for a stable process can have devastating consequences, especially if the process outcomes have a food safety component. Consider again, the example of the contaminated surgical instruments. What if the surgery had concluded that the prion contamination was a common cause event; i.e., normal and acceptable variation in the cleaning procedures that did not require special actions or new procedures?
Process Validation Strategies
Increasingly, due largely to increasing concerns for food safety, the food industry is recognizing the necessity for validating its procedures and processes. The three reasons most often cited as justification for process validation are:
• Safety, quality and effectiveness must be designed and built into the product
• Safety cannot be inspected or tested into the finished product
• Each step of the manufacturing process must be controlled to maximize the probability that the finished product meets all safety, quality and design specifications.
It is interesting that these rationales are based on the notion that it is control of the process that results in the production of the right product with the right attributes. Dr. Deming recommended investing in testing and inspecting manufacturing processes, rather than on testing finished products. Validation is about just that. It is the method that establishes that a process can be relied on with a high degree of certainty to delivery a safe product.
Listed below are examples of the types of processes and procedures that have historically required validation. It is noteworthy that in each case the expected outcomes of the process have a direct relationship with the safety or public health status of the product.
• Sterilization processes
• Aseptic filing processes
• Sterile package sealing processes
• Heat treating processes
• Certain cleaning processes (prions)
• Novel processing techniques
There are three commonly used strategies for process validation: retrospective, concurrent and prospective.
Retrospective validation is a technique for validating a product already in distribution based upon accumulated production, testing and control data. All too many food safety, quality and regulatory specialists are familiar with this method of validation. As previously discussed, it is retrospective validation methodology that is frequently used for analyzing process failures that result in product recalls. With this method accumulated production, testing and control data are examined in an effort to determine when the process went out of control and the production of defective products commenced.
Concurrent process validation may also be familiar to food industry scientists. This technique is based on the simultaneous collection and evaluation of data emanating from a process concurrent with its application. Concurrent validation methodologies are frequently used in the context of proving the efficacy of a change or modification to an established and previously validated process.
By way of example, let’s examine the impact of changing the concentration and contact time of a chemical sanitizer on the inactivation kinetics of a microorganism targeted by the processes. Contact time and sanitizer concentration are critical for achieving the desired inactivation effect. Thus, it is reasonable to conceive that data collected during a real-time disinfection process may be used to validate the efficacy of that modified procedure. It must be noted, however, that the microbiological status of the equipment that is the subject of the validation trials remains in question until all data have been collected and analyzed.
Prospective validation is a rather different process and requires a deliberate, planned approach to data collection and evaluation. New methods, new procedures and novel processes require prospective validation. An ultrasound assisted process for low temperature pasteurization of egg albumin, because of the paucity of information in the literature relating to its efficacy, requires a very rigorous, forward-looking evaluation to determine that the process can be relied upon with a high degree of confidence to deliver a safe food. The prospective validation process, in this case, should be completed, all data collected, analyzed and reviewed, before a decision is taken to accept the use of this technology in the mass production of human food.
Each of these validation strategies shares in common the need for the precise evaluation and qualification of the processing equipment, the manufacturing processes and the finished product.
Installation performance qualification (IPQ) establishes by objective evidence that all critical aspects of the process equipment and ancillary system installations are consistent with the manufacturer’s approved specification and warranties. Examples of applicable elements of the IPQ validation protocol centers on the equipment design. This information includes the materials of construction and description of sanitation and maintenance of the equipment. The installation conditions including; wiring diagrams, utilities, plumbing, ventilation, isolation and other external requirements are integral and necessary components of the protocol. Additional elements included in the protocol are equipment calibration requirements, reliability specifications, safety features, and software and programming parameters. The IQ validation requires all vendor documentation, specifications and conditions recommended for installation (e.g., temperature, humidity, dust level limits), and necessary on-hand spare parts.
The operational performance qualification (OPQ) establishes by objective evidence process control limits and relevant action levels which result in an output that meets all predetermined requirements. The OPQ protocol represents an even longer list. Information includes the human factors impacting the process, such as training requirements for workers and possible ergonomic issues. The process operating procedures are required, preferably in specific detail, along with process change control procedures. Software parameters and screening experiments to establish the key process parameters are also required. Also necessary are the control and operational limits (i.e., time, temperature, conductivity, pressure, flow rates, electrical field density and line speed). Short-term stability and capability (e.g., control charts and other monitoring devices), raw material and packaging specifications (e.g., input variation), and material-handling specifications (e.g., temperature, time, shelf-life, storage conditions) are also elements of the operational performance protocol. The action levels should be evaluated, established and documented during process validation to determine the overall robustness of the process and its ability to avoid approaching worst case conditions. Useful tools for this phase of testing include Failure Mode and Effects Analysis and Fault Tree Analysis.
The product performance qualifications (PPQ) establishes by objective evidence the process is consistently capable of yielding a results or product that will meet predetermined specifications. In this phase of qualification the key objective is to demonstrate that the process will produce acceptable product under normal operating conditions. The PPQ is concerned primarily with attainment of the critical characteristics and properties of the food product.
Critical product characteristics may include organoleptic or other physical factors affecting food quality or food safety. The PPQ protocol will likely include finished product specifications, pH, water activity, conductivity (redox potential), titratable acidity, viscosity, moisture, solids, degree Brix (refractive index), particle-to-liquid distribution ratio, levels of microorganisms (i.e., pathogens, surrogates or spoilage agents), and the chemical residue profile (e.g., indicators and toxins).
The strategies and method of validation notwithstanding, it is a fairly accepted practice that the validation processes adhere to the following guidelines:
• Form and train a multi-functional team in validation procedures
• Develop a master validation plan
• Develop process validation protocols
• Initiate trials according to protocol
• Analyze the data and report findings
Formation of a team is critical for seldom is process validation the purview of a single field of endeavor or specialty. On the contrary, process validation should be conducted by a team of specialists. Teams most often are comprised of representatives from manufacturing, research and development, food safety and microbiology, packaging, and operations. A team assembled to validate a PEF process might, for example, include physicists, organic chemists, physical chemists, microbiologists, electrical engineers, mechanical engineers, toxicologists, food scientists, regulatory and other health and safety specialists. The contrasting views and opinions of the team, when understood in the context of process validation, portend a successful outcome for the processes under evaluation.
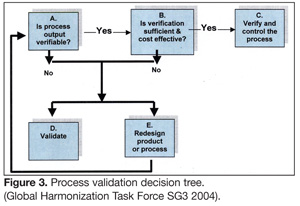 Another very important aspect of process validation whether or not on it is justified. Using a process validation decision tree is a useful tool in deciding on whether or not a process needs to be validated (Figure 3). Developing a master validation plan is a critical first activity for the validation team. The master validation plan is a document that provides a statement of purpose, intent and justification. A statement of purpose must clearly delineate in broad form of the outcomes of the validation process. For example, a team that is assembled to investigate the feasibility of using PEF for inactivation of pathogenic bacteria in apple juice will decide on the intended approach to process validation and include this in the master validation plan. If the team states that they will use a retrospective approach, then an overview of the activities that will continue the validation process should be clearly stated.
Another very important aspect of process validation whether or not on it is justified. Using a process validation decision tree is a useful tool in deciding on whether or not a process needs to be validated (Figure 3). Developing a master validation plan is a critical first activity for the validation team. The master validation plan is a document that provides a statement of purpose, intent and justification. A statement of purpose must clearly delineate in broad form of the outcomes of the validation process. For example, a team that is assembled to investigate the feasibility of using PEF for inactivation of pathogenic bacteria in apple juice will decide on the intended approach to process validation and include this in the master validation plan. If the team states that they will use a retrospective approach, then an overview of the activities that will continue the validation process should be clearly stated.
Justification is also a key activity because validation procedures tend to be very expensive. Often, the cost of validating novel technologies is a prohibitive undertaking for a single company. Therefore, consortiums are frequently formed for purpose of sharing in the economic burden of demonstrating proof of the method or process. As consortia or individual companies they all want to know that after investing several hundred thousands of dollars that at the end of the process there will be a clear and economic benefit to the organization.
The master validation plan in many respects is analogous to the writing of an abstract for research project. This document provides an overview of the validation activities. Moreover, it provides a statement of expected outcomes. The master validation plan serves the purpose of making sure that all concerned stakeholders have a clear view as to what is going to be undertaken. The plan needs to be transparent and provide information necessary for making decisions about going forward with the project. It is also recommended that the master plan be peer reviewed prior to its being accepted. Moreover, if regulatory authorities or agencies are to be involved with the outcomes of the validation process, then they too must be involved early on in the process and the master validation plan is an excellent mechanism by which to invite their participation.
The process validation protocols are an extension of the master validation plan. The protocols, however, provide the materials and methods of validation. This document elaborates in great detail all procedures that will be used in the validation process. The validation protocols also establish criteria for success and the level of statistical confidence that is required for declaring success or failure. For example, if the protocol stipulated the use of count reduction studies for demonstrating the lethality of a specific process. It is likely then that the protocol would specify the food (testing medium), the microorganism (pathogen or surrogate), the package, the method of delivering the lethal effect, as well as the methods for recovery and enumeration of the test organism. The protocol should also specify, in advance, the required levels of inactivation that are required. Additionally, for statistical purposes, the protocol establishes the number of experiments that are to be conducted.
Once the protocols have been established, reviewed and accepted by the team the validation process can begin in earnest. It is the responsibility of the team to ensure that the terms of reference and all procedures delineated in the protocols are explicitly adhered to. Otherwise, it would be nearly impossible to evaluate the experimental outcomes and provide a meaningful objective assessment of the processes’ reliability or reproducibility.
Proper analysis of the data as alluded is critical to demonstrating “proof of the process.” For this reason and others, it is recommended that the validation team include a statistician or someone well-versed in statistical process control (SPC). The success criteria established by the team in advance of initiating the validation process are found only through the detailed analysis of the data.
Summarizing and reporting the results are also essentials elements of the validation process. At the conclusion of the validation trials a final report summarizing and referencing all protocols and results should be assembled. The documents should derive conclusions related to the validation status of the process that has been evaluated. The report must make clear whether or the process is stable and capable of delivering the desired outcome with a high degree of statistical confidence. The report should also delineate in very clear language the minimum operational requirements necessary for achieving the desired results or product. The summary report should be reviewed and accepted by the validation team prior to its being presented to management, regulatory officials or other stakeholders.
Validating Confidence
Process validation, the process of establishing and using scientific methods to assure that a process can be relied upon with a high degree of confidence to produce a product or results that meet predetermined specifications for food safety, is becoming increasingly important as a strategic element in assessing and managing food safety risk. The critical control points of a HACCP plan must be validated before the plan is rolled out onto the production floor. That is, the hazard containment elements of the plan must be shown to be effective. As shown, novel processes for preserving foods and food manufactured using unconventional methods also require validation through a deliberate, well-planned set of experiments that have been designed explicitly to demonstrate the stability and capability of the process.
It is a generally accepted principle, that food safety can neither be tested nor inspected into a food product. Said in another way, if the process is not right the product won’t be right (safe). Process validation aims to establish by scientific methods that a process is capable, reliable and reproducible. It must also be kept in mind that process validation is absolutely process and product specific. The protocols written for a cold plasma process will not be valid for use with another novel preservation method. However, the validation framework and guidelines as discussed are applicable to all processes and procedures. Indeed, quantifying the probabilities and magnitude of adverse effects of a failed process and the myriad resulting consequences constitutes the heart of process validation.
It has been noted that validation is an expensive exercise. The expense, due to public health implications of delivering an unsafe food into the marketplace, is justified. The uncertainty surrounding new preservation techniques demands that they are prospectively validated. Food safety management is about prediction and prediction is dependent on objective data that have been properly gathered and expertly analyzed.
Larry Keener is the owner and general manager of International Product Safety Consultants, Inc. (IPSC), Seattle, WA, a global leader in providing food safety and food technology services in the areas of food security, microbiology, regulatory affairs and crisis management to clients in Asia, Africa, Australia, Europe, Central and South America, Canada and the U.S.
Most recently, Larry is co-founder and co-chair of the Global Harmonization Initiative, an organization founded to promote harmonization of food safety legislation and regulations. He is a member of the Institute of Food Technologists and is chair-elect of IFT’s Non-Thermal Processing Division. Larry is a member of the editorial advisory board of Food Safety Magazine, for which he is a frequent contributor. He can be reached at Lkeener@aol.com.
Bibliography
Dailey, K.W. The FMEA Pocket Handbook. DW Publishing, USA. 2004.
Deming, W.E. The New Economics for Industry, Government, Education. MIT Press, Cambridge, MA. 1993.
Haimes, Y. Risk Modeling, Assessment, and Management. Wiley and Sons. NJ. 2004.
Harry, M. and R. Schroeder. Six Sigma. Random House, NY. 2000.
Hojo, T. Quality Management Systems: Process Validation Guidance. Global Harmonization Task Force, Study Group #3, Edition 2. 2004.
Keener, L. Handbook of Hygiene Control in the Food Industry, Chapter 28. Woodhead Publishing. Cambridge, UK. 2005.
Keener, L. Process Validation Methodology. 2nd Innovative Foods Center Conference, Australia. 2004.
Keener, L. and D. Hoover. Process Validation Procedures. IFT Nonthermal Processing Division Workshop, Philadelphia, PA. 2005.
Klein, A. “Reused Devices, Surgery’s Deadly Suspects,” Washington Post. Dec. 30, 2005.
McDermott, R.E., et al. The Basics of FMEA. Productivity Press. Portland, OR. 1996.
Shaw, A. Guideline on General Principles of Process Validation. U.S. FDA, Washington, DC. 1987.
Shewhart, W.A. Statistical Methods from the Viewpoint of Quality Control. Dover Publication. New York, NY. 1986.
Taylor, W.A. Methods and Tools for Process Validation. Global Harmonization Task Force Study Group #3. Taylor Enterprises, USA. 1998.
World Health Organization. WHO Infection Control Guidelines for Transmissible Spongiform Encephalopathies. Report of a WHO consultation. Geneva, Switzerland, 23–26 March. WHO/CDS/CSR/APH/2000.3. 1999.