In January, the U.S. Food and Drug Administration’s (FDA) Center for Food Safety and Applied Nutrition (CFSAN) issued its Fiscal Year (FY) 2004 end-of-year report. The “CFSAN Report Card,” summarizes all significant accomplishments from CFSAN’s annual list of foods program priorities. The CFSAN Report Card is issued as part of the Center’s continued commitment to work together with its stakeholders to ensure the safety and security of the U.S. food and cosmetics supply.
For the third consecutive year, CFSAN succeeded in meeting its end-of-year goal of accomplishing over 90% of its total number of A-list priorities (151 out of 162 priorities). This article will not address every accomplishment; rather, it will emphasize significant accomplishments from a cross-section of the entire report card, highlighting the various ways in which CFSAN ensures a safe food supply and either directly or indirectly benefits the American consumer.
From Workplan to Report Card
CFSAN’s mission is to protect the safety and security of the nation’s foods and cosmetics and it works to achieve its mission through establishing a set of annual goals that reflect the current needs of its stakeholders (i.e., consumers and the industry that we regulate). These goals reflect our commitment to place the greatest emphasis on work that has the potential to do the most good for the consumer and the overall public health.
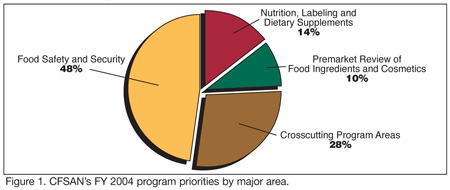 At the beginning of each fiscal year, CFSAN releases a document entitled “CFSAN Program Priorities.” CFSAN’s FY 2004 Program Priorities document was the Center’s largest workplan yet. It consisted of 168 major items known as A-list priorities falling under four major areas: 1) food safety and security; 2) nutrition, labeling and dietary supplements; 3) premarket review of food ingredients and cosmetics; and 4) crosscutting program areas (Figure 1). Food safety and security was the largest area, representing 48% of the program priorities. All four areas, however, are important in ensuring the safety of U.S. foods and cosmetics.
At the beginning of each fiscal year, CFSAN releases a document entitled “CFSAN Program Priorities.” CFSAN’s FY 2004 Program Priorities document was the Center’s largest workplan yet. It consisted of 168 major items known as A-list priorities falling under four major areas: 1) food safety and security; 2) nutrition, labeling and dietary supplements; 3) premarket review of food ingredients and cosmetics; and 4) crosscutting program areas (Figure 1). Food safety and security was the largest area, representing 48% of the program priorities. All four areas, however, are important in ensuring the safety of U.S. foods and cosmetics.
During the course of the year, FDA is often required to respond to numerous unanticipated events that can impact time and resource commitments for priority items. Occasionally, CFSAN will revise its priorities based on these events. The mechanism used to accomplish this is the mid-year review, which provides an opportunity to determine whether CFSAN is on-track in accomplishing its stated goals by the end of the year. During the review, CFSAN can make a realistic determination of what has been accomplished; assess what remaining goals can realistically be accomplished; and identify and remove A-list goals that realistically cannot be reached. As a result of the mid-year review, the total number of A-list priorities in 2004 was reduced to 162.
A Look Back at FY 2004
Of CFSAN’s accomplishments in 2004, highlights include initiatives and measures concerning food security, food safety, obesity, dietary supplements and biotechnology.
Food Security. Food defense remains the central theme of FDA’s public health agenda. The events of Sept. 11, 2001, predicated the need to enhance the security of the U.S. food supply. Congress responded by enacting the Public Health Security and Bioterrorism Preparedness and Response Act of 2002 (the Bioterrorism Act), which was signed into law on June 12, 2002. On June 4, 2004, CFSAN implemented a portion of this law by publishing the final regulation for “Administrative Detention of Food for Human or Animal Consumption Under the Public Health Security and Bioterrorism Preparedness and Response Act of 2002.” Often referred to as the “Administrative Detention” final rule, it provides procedures for the detention of an article of food, if an office or qualified employee of FDA has credible evidence or information indicating that such article presents a threat of serious adverse health consequences or death to humans or animals. This regulation became effective July 6, 2004.
This rule, in conjunction with the interim final rule for food facility registration and the interim final rule for prior notice of imported foods and the recordkeeping final rule (which published in November 2005) are all crucial measures to ensure the security of the nation’s food supply. Additionally, FDA conducted a vulnerability assessment for food terrorism, published numerous guidance documents to industry, and compliance policy guidances for FDA staff to assist the implementation of the four sections of the Bioterrorism Act. The agency is currently working to publish in FY 2005 the final versions of the food registration and prior notice rule.
Food Safety. Ensuring a safe and secure U.S food supply requires adequate and appropriate compliance and outreach programs for all foods. The Center accomplishes this through inspections, both domestic and foreign, sampling and analysis of food products, conducting outreach addressing good manufacturing practices and good agricultural practices, assisting other countries to produce safe food, training food handlers, educating consumers, reviewing food ingredients before they appear on the market, developing and implementing standards for safe foods, and conducting food-related research. In each of these areas the Center either met or exceeded these specific goals.
As part of the effort to ensure that food regulations are up to date, FDA is taking a fresh look at the current food Good Manufacturing Practices (cGMPs), which have been in place since 1986. In the years since the cGMPs were published, there has been a steady increase in reactions to undeclared food allergens and the continued emergence of new pathogens such as Listeria monocytogenes. Based in part on information gathered at three public meetings this year, CFSAN concluded that a modernization of the food cGMPs is needed.
Another area of food safety that requires adequate and appropriate preventative measures is fresh produce. Fresh produce continues to be an important component of a healthy diet. Unfortunately, the U.S. has witnessed a number of foodborne illness outbreaks related to fresh produce in recent years. Because of the importance of fresh produce in a healthy diet and continuing outbreaks associated with the consumption of fresh produce, FDA developed the “Produce Safety Action Plan.” This plan is intended to further minimize foodborne illness associated with the consumption of fresh produce. The plan is designed to target microbial food safety hazards—such as bacteria, viruses and parasites—in or on produce consumed in the U.S., whether produced in the U.S. or abroad. FDA believes that each entity involved in producing, packing, processing, transporting, distributing, or preparing fresh produce has a responsibility to conduct its activities so as to reduce, control, or eliminate microbial contamination in produce. Thus, the Action Plan extends to all parts of the food chain from the farm, through the retail environment, to the home. It is intended to cover fresh fruits and vegetables, both in their unpeeled, natural form and raw products that have received some minimal processing (such as peeling, chopping, or trimming). The Action Plan is not intended to cover frozen fruits and vegetables, fruit and vegetable juices, or other commodities such as tree nuts that are neither fruits nor vegetables and not typically regarded as produce.
One particular food identified by FDA that has a demonstrated need for improved preventative controls is shell eggs. From 1990 to 2001, 14,319 illnesses were attributed to Salmonella Enteritidis (SE) associated with shell eggs. Prior to FY 2004 FDA published refrigeration and labeling rules designed for eggs. These were post-production efforts to reduce the levels of SE. On Sept. 22, 2004, FDA published a proposed rule on egg safety entitled “Prevention of Salmonella Enteritidis in Shell Eggs During Production.” The proposal, if adopted, will require shell egg producers to implement measures to prevent SE from contaminating eggs on the farm. FDA expects that the requirements will ultimately generate public health benefits through a decrease in the numbers of SE-associated illnesses and deaths caused by consumption of shell eggs.
In FY 2004, FDA and the U.S. Environmental Protection Agency (EPA) issued a joint consumer advisory about mercury in fish and shellfish. The advice is for women who might become pregnant, women who are pregnant, nursing mothers and young children. The purpose of the advisory is to inform these groups on how to get the positive health benefits from eating fish and shellfish while minimizing their mercury exposure. This is significant because it is the first time FDA and EPA have combined their advice into a single, uniform advisory. Previously, FDA issued an advisory on commercially caught fish, while EPA issued advice on recreationally caught fish.
Obesity Prevention. Obesity is a pervasive public health problem in the U.S. Since the late 1980s, adult obesity has steadily and substantially increased. The problem of obesity has no single cause; rather, it is the result of numerous factors acting together over time. On March 12, 2004, FDA’s Obesity Working Group publicly released the “Calories Count” report. This report includes recommendations to strengthen food labeling, to educate consumers about maintaining a healthy diet and weight, and to encourage restaurants to provide calorie and nutrition information. It also recommends increasing enforcement to ensure that food labels accurately portray serving size; revising and reissuing guidance on developing obesity drugs; and strengthening coordinated scientific research to reduce obesity and to develop foods that are healthier and low in calories.
Dietary Supplements. FDA continues to respond to consumer demand for protection from hazards while at the same time recognizing consumer desire for access to safe dietary supplements. While there may be benefits from some of these products, the potential for exaggerated claims, unpredictable composition and toxicity are of considerable concern to FDA.
Of particular concern is the presence of unsafe dietary supplement ingredients. Until early 2004, dietary supplements containing ephedrine alkaloids were widely sold in the U.S. Scientific evidence and numerous reports of serious adverse events, including death, following consumption of dietary supplements containing ephedrine alkaloids, have raised concerns about those supplements containing these ingredients. Consequently, FDA has taken a number of actions in an attempt to protect the public from the risks of these products. On Feb. 11, 2004, FDA published a final regulation declaring dietary supplements containing ephedrine alkaloids, better known as “ephedra,” as adulterated under the Federal Food, Drug, and Cosmetic Act. FDA took this action based upon the well-known pharmacology of ephedrine alkaloids, the peer-reviewed scientific literature on the effects of ephedrine alkaloids, and the adverse events reported to have occurred in individuals following consumption of dietary supplements containing ephedrine alkaloids.
Another issue that raised safety concerns in FY 2004 was the marketing of dietary supplements containing androstenedione, or “andro.” FDA learned of and sent warning letters to 23 firms that were actively manufacturing, marketing, and/or distributing androstenedione as a dietary supplement. The warning letters asked each firm to cease distributing products sold as dietary supplements that contain androstenedione and warned them that they could face enforcement action if they did not comply with this letter. FDA took this action because there is inadequate information to establish that a dietary supplement containing androstenedione will reasonably be expected to be safe. This product is marketed as a “performance enhancer” advertised to promote muscle growth, improve muscular strength, reduce fat and slow aging. Studies show that sufficient quantities of androstenedione, taken over a long period of time may have serious adverse health affects.
Food Biotechnology. The use of biotechnology is now routine in the production of drugs and is expected to become more important to food producers, as well. More than 45 plant-derived foods produced using bioengineering are now marketed in the U.S., most containing genetic characteristics that resist pests. Others with additional traits, such as higher nutrient content or improved flavor, are expected soon. FDA has oversight of the safety of all bioengineered foods, although that oversight does not extend to the safety of pesticidal proteins engineered into food plants, which are regulated by EPA.
As part of FDA’s effort to enhance information provided to the public in FDA’s evaluation of foods derived from bioengineered plants, CFSAN has developed a web page that shows the list of biotechnology consultations that CFSAN has completed. It can be viewed at www.cfsan.fda.gov/~lrd/biocon.html. CFSAN also developed a guidance concerning the potential presence of unintended varieties of bioengineered plant foods that may be present in the food supply, but that have themselves not completed all regulatory steps for marketing. The guidance was published in the Federal Register in November of FY 2004.
Here at Last!
From 1996 until the fall of 2004, relocating CFSAN staff to College Park was the Center’s most prominent crosscutting management initiative. A great deal of effort and resources were devoted to this goal. In September 1996, Maryland officials held a groundbreaking ceremony marking the beginning of the construction of the new College Park home for CFSAN. Five years later, on Oct. 1, 2001, the Harvey H. Wiley Building was dedicated, providing new offices for over 80% of CFSAN’s staff.
In the fall of 2004, construction of a second CFSAN building, called University Station, and located next door to the Wiley Building, was completed. The relocation of nearly all CFSAN staff to a single location marks the close of a significant milestone in the goal of developing and maintaining a world-class organization at CFSAN. Now that the relocation of staff is complete, this monumental effort will no longer compete for time and resources and the Center looks forward to FY 2005 as its most successful year yet.
Donald Carrington is a program analyst in CFSAN’s Office of Management Systems. He has more 30 years’ experience working for FDA and has been involved with the development, monitoring and the reporting of CFSAN’s program priorities since its inception.
FDA’s Food Program Priorities: A Look Back at 2004 Accomplishments




