In September 2002, the U.S. Department of Agriculture's Food Safety and Inspection Service (USDA FSIS) announced its plan to institute a series of additional measures to complement previous policies aimed at the prevention and control of Escherichia coli O157:H7 in ground beef. In issuing its Federal Register notice, the agency stated that these new measures are based on updated information indicating that the incidence of E. coli O157:H7 contamination in raw ground beef is more prevalent than was previously estimated.
Specifically, such information included a February 2000 study by USDA's Agricultural Research Service (ARS), which documented that 28% of the proportion of animals entering the slaughter plant were infected with E. coli O157:H7 and that an average of 43% of carcasses tested positive for the pathogen at various stages of production. In its risk assessment initiative, which ultimately fostered the adoption of the new measures, FSIS also included the latest E. coli O157:H7-associated foodborne illness estimates from the Centers for Disease Control and Prevention (CDC), which have increased to 62,000 illnesses, 1,800 hospitalizations and 52 deaths per year caused by this pathogen.
The new measures to address E. coli O157:H7 contamination in ground beef include the following:
• All beef slaughter and grinding plants are required to acknowledge that E. coli O157:H7 "is a hazard reasonably likely to occur in their operations, unless they can prove otherwise."
• All establishments producing raw beef products must reassess their mandatory Hazard Analysis & Critical Control Points (HACCP) plans and investigate the adequacy of existing pathogen reduction/intervention controls. If controls are not in place or are determined to be inadequate, a pathogen reduction step to reduce the risk of E. coli O157:H7 in the product must be incorporated into the production process.
• FSIS inspectors will now conduct random microbiological verification testing of all beef grinding operations, eliminating previous exemptions of some slaughter plants. The agency also may expand its testing program to include trimmings and carcasses in addition to ground beef.
• FSIS will increase inspections of pathogen reduction and intervention steps to verify that these steps are effective in reducing the incidence of E. coli O157:H7 under actual plant conditions.
While these government-mandated measures will result in increased plant testing for E. coli O157:H7 in beef grinding operations, likely spurring an increase in the demand for sensitive and rapid isolation methods such as real-time polymerase chain reaction (PCR) and immunomagnetic separation (IMS), testing for generic E. coli continues to play a significant role in the meat and poultry industries, as provided by FSIS regulation. The USDA FSIS requires that all poultry and beef slaughter facilities enumerate generic E. coli on carcasses as a means of estimating fecal contamination. This testing is conducted to verify that sanitation standard operating procedures (SSOPs) for equipment and facilities and all process controls used in the plant are effectively working as designed to prevent fecal contamination and to verify process control.
Proper enumeration means that the poultry industry must test 11 to 12 carcasses per day. The level of E. coli that is considered "acceptable" for poultry products is fewer than 100 colony forming units (CFU)/mL of whole carcass rinse (400 mL). A level of 100-1000 CFU/mL is considered marginal, and E. coli levels higher than 1000 CFU/mL are considered unacceptable. For beef carcasses, the level of E. coli that is considered "acceptable" is zero. A level of 1-100 CFU/cm² is considered "marginal," and a level greater than 100 CFU/cm² is considered "unacceptable." If a meat or poultry facility detects marginal or unacceptable levels of E. coli, it must be able to quickly locate the source of the problem and take corrective action. The evolution of rapid, automated enumeration methods is a welcome development in helping industry do just that. In particular, improved electrical and optical methods allow processors to test product and obtain results prior to shipping, avoiding the cost and loss of reputation associated with a recall or foodborne outbreak.
Enumeration Evolution
There are three types of enumeration methods currently in use in the meat and poultry industries, and these differ quite a bit with regard to how one approaches the actual counting of the organism. We can class these into the following main groups: traditional, commonly used rapid and rapid, automated methods. The latter type is instrumental methodology that can be further sub-categorized into impedance/conductance (electrical) and optical methods, and these latter methods are the focus of new advancements in enumeration.
 The meat and poultry industry is well acquainted with the traditional Most Probable Number (MPN) method used to detect and enumerate E. coli as an indicator of fecal contamination or unhygienic processing (Figure 1). This statistical assay method involves traditional serial dilutions and inoculation of the sample in a broth media, followed by an analysis of gas positive tubes and a comparison of phases against statistical tables. This procedure allows the analyst to estimate the most probable number of coliforms, fecal coliforms and/or E. coli present in the sample. The most common MPN method used in food testing is the three-tube MPN test, and although it is considered the gold standard in terms of sensitivity, the method takes a minimum of five days to get results on one test and is expensive to perform in terms of its labor-intensive nature.
The meat and poultry industry is well acquainted with the traditional Most Probable Number (MPN) method used to detect and enumerate E. coli as an indicator of fecal contamination or unhygienic processing (Figure 1). This statistical assay method involves traditional serial dilutions and inoculation of the sample in a broth media, followed by an analysis of gas positive tubes and a comparison of phases against statistical tables. This procedure allows the analyst to estimate the most probable number of coliforms, fecal coliforms and/or E. coli present in the sample. The most common MPN method used in food testing is the three-tube MPN test, and although it is considered the gold standard in terms of sensitivity, the method takes a minimum of five days to get results on one test and is expensive to perform in terms of its labor-intensive nature.
The second group of methods constitutes the most commonly used methodology for enumerating E. coli from broiler chicken carcasses and ground beef. These rapid detection methods, Petrifilm (3M Co.) and SimPlate (BioControl), are based on the detection of the production of an enzyme (glucuronidase) by E. coli, and are available as commercial test kits. Both Petrifilm and SimPlate assays can detect moderate to high levels of glucuronidase-positive E. coli and offer the benefit of results in 24 hours, which allows the plant to get required results and release the product the same day.
The third group is comprised of rapid, automated enumeration methods, which differ from the commonly used rapid methods in that they are not involved in the detection of glucuronidase, but rather, are based on the detection of metabolic by-products in a medium. Thus, these automated methods are able to detect any and all bacteria present in the sample, even if they are injured, because bacteria will repair themselves and grow to produce enough metabolites so that they are picked up by the instrument. Both impedance and optical enumeration methods provide very rapid results (between 2 and 10 hours) as compared with the commonly used rapid methods, but they also offer high sensitivity in terms of detection of lower levels of E. coli. In fact, one could say that the sensitivity of the assay itself is one cell, although the actual detection threshold of the instrument is 10[6]. With both systems, the more contaminated the sample, the quicker they will detect. If the sample is highly contaminated, it may take as few as two hours to obtain results.
There are other similarities and some important differences between the impedance and optical techniques that bear further discussion. The impedance method is a well-known technique involving the opposition to flow of an alternating electrical current in a conducting material. As bacteria multiply, they convert large molecules into smaller, more mobile metabolites that change the impedance of the medium. These metabolites increase the conductance and decrease the impedance of the medium. When microbial populations reach 10[6] to 10[7] cells/mL, the change in the impedance of the medium is sufficient to be detected by the instrument. At this point, a detection time is noted. The detection time is inversely proportional to total count.
The Bactometer (bioMerieux) and the RABIT (Don Whitley Scientific) systems are instruments based on the impedance technique. The optical technique can be performed on the BioSys optical instrument system (distributed by BioSys, Remel and Foss). This instrument allows the user to directly add swabs or sponges without dilution to a pre-filled disposable test vial containing agar and liquid broth. Vials are placed in the automated instrument, which then detects optical changes in the semi-fluid zone that separates the liquid medium containing the sample from the reading area. This color reaction is recorded by a PC-based system that includes both calibration and histogram software, which trends and provides statistical information in a graphical format. The operator can simultaneously monitor from one to 128-plus samples.
In addition to the very rapid results provided by both of these automated methods, these systems can analyze as many times as many samples as the conventional MPN methods (at a ratio of 6:1 versus 10:1), are extremely accurate, exhibit much less error than in plate counting and require minimal training.
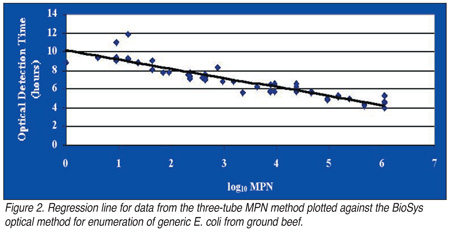 While both systems display a high sensitivity appropriate for the meat and poultry industry to use as a means of enumerating low levels of E. coli from whole broiler carcasses or ground beef, there are a few caveats to consider. Currently, for example, low levels of E. coli are common on broiler carcasses because the acceptable level as dictated by USDA FSIS is less than 100 CFU/mL of rinse. Approximately 90% or more of commercial broiler carcasses fall into this category. This means that the enumeration method used must have the capability to detect E. coli at lower levels. In one enumeration methods comparison study conducted in our laboratory using the BioSys optical and the Bactometer conductance methods, more variance was associated with broiler and ground beef samples that contained lower numbers of E. coli. The variance associated with low numbers of E. coli using the Bactometer method appeared to be much greater than that observed using the BioSys method. For one sample of ground beef, the BioSys optical method was able to enumerate E. coli; whereas the MPN method was unable to detect the presence of E. coli on this sample (Figure 2).
While both systems display a high sensitivity appropriate for the meat and poultry industry to use as a means of enumerating low levels of E. coli from whole broiler carcasses or ground beef, there are a few caveats to consider. Currently, for example, low levels of E. coli are common on broiler carcasses because the acceptable level as dictated by USDA FSIS is less than 100 CFU/mL of rinse. Approximately 90% or more of commercial broiler carcasses fall into this category. This means that the enumeration method used must have the capability to detect E. coli at lower levels. In one enumeration methods comparison study conducted in our laboratory using the BioSys optical and the Bactometer conductance methods, more variance was associated with broiler and ground beef samples that contained lower numbers of E. coli. The variance associated with low numbers of E. coli using the Bactometer method appeared to be much greater than that observed using the BioSys method. For one sample of ground beef, the BioSys optical method was able to enumerate E. coli; whereas the MPN method was unable to detect the presence of E. coli on this sample (Figure 2).
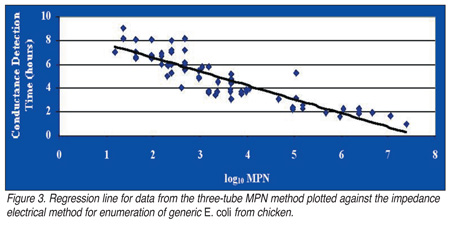 In the same study, the Bactometer method was found to be optimal in terms of regression statistics for enumerating E. coli from ground beef (r = -0.96) and the regression line for data from the three-tube MPN method plotted against the impedance electrical method for enumeration of generic E. coli from chicken was also optimal (Figure 3). However, on ground beef samples that contain lower concentrations of E. coli (< 2 log[10] CFU/mL), the conductance method became more erratic; hence, enumeration of very low numbers of E. coli from ground beef using this method should be conducted with caution.
In the same study, the Bactometer method was found to be optimal in terms of regression statistics for enumerating E. coli from ground beef (r = -0.96) and the regression line for data from the three-tube MPN method plotted against the impedance electrical method for enumeration of generic E. coli from chicken was also optimal (Figure 3). However, on ground beef samples that contain lower concentrations of E. coli (< 2 log[10] CFU/mL), the conductance method became more erratic; hence, enumeration of very low numbers of E. coli from ground beef using this method should be conducted with caution.
Another difference between the two rapid automated methods is that the Bactometer impedance method can display electronic drift, which essentially means that the instrument cannot detect the location of the inflection point in the curve. The optical method does not utilize electrical measurements, and thus does not exhibit electronic drift.
In addition, the types of samples one is analyzing must also be considered. With the Bactometer, if the microbiologist places salts in the media to select for certain kinds of bacteria it will disrupt the impedance curve. Also, gas producers will make bubbles in the media using impedance and that will disrupt the curves, as well. The electrical signal results in a jagged line instead of a nice, smooth curve and the instrument cannot interpret these curves.
The primary caveat with the BioSys optical method is that any foods that are high or low in pH may influence the reading, because it is it a pH-based method and the medium is pH sensitive. Thus, to get an accurate reading of food samples with low pH such as highly acidic foods, like lemons, citrus foods or juices, the analyst must adjust the pH before placing the sample in the instrument. The optical method works well for meat and poultry products, which are separated from the actual sample. In the Bactometer, the sample is in contact with the electrodes, and thus any particulate matter, salts, or fatty compounds in the meat sample that can get in between the electrodes in the impedance sample is going to affect the reading. The BioSys optical instrument features an agar plug at the bottom of the instrument where the actual reading takes place, and no contact takes place between the food sample suspended in the liquid at the top and the agar plug at the bottom and there is no influence on the reading. In this way, the optical method enables the user to analyze salty or opaque samples.
Conclusion
Enumeration methods have improved dramatically in the past few years, becoming more rapid, more automated and more accurate. In particular, the automated impedance/conductance and optical methods for enumerating Escherichia coli from broiler carcasses and ground beef perform very well in comparison to the most commonly used methods for enumerating E. coli in the meat and poultry industries. Moreover, although these instruments have a capital cost associated with their purchase ($23,000 to $60,000), the cost per test ($1.00 to $2.00, including labor) is much lower than the cost per test than the standard plate count method. The labor and time required to prepare samples using the rapid automated methods are significantly reduced, as well, because only one dilution is required.
Scott M. Russell, Ph.D., is associate professor in the Department of Poultry Science at the University of Georgia, Athens, GA., where he has conducted research and provided extension services to the poultry industry for nine years. His main areas of interest are developing rapid methods for identifying and enumerating pathogenic and spoilage bacteria from foods of animal origin, and identifying methods for eliminating pathogenic and spoilage organisms from poultry during rearing and processing. Contact him at srussell@arches.uga.edu.
>



