On September 13, 2006, the U.S. Food & Drug Administration (FDA) instructed the public to stop eating fresh spinach because there was an outbreak of Escherichia coli O157:H7 in the nation, and the common factor among the patients seemed to be spinach. The media was in a frenzy over the news, and retail stores were sweeping their shelves of spinach or any products that contained spinach. As a significant producer of spinach, at that point, we were on alert, but knowing that we had excellent food safety protocols, we weren’t concerned that our products might be involved.
However, on September 14, 2006, everything changed. Every detail of that day is engraved in my memory, especially where I was and what I was doing when I got the call. I was walking to our main office to meet with our growers about an Organic Integrity Program we were getting ready to launch. The California Department of Public Health was calling to tell me that our company, Earthbound Farm, was being implicated in the nationwide foodborne illness outbreak associated with spinach. When we got on the call with state and federal officials, representatives of several companies were participating, all anxious to get the news and prepared to defend their companies and brands.
Immediately following the call with regulatory agencies, we engaged our Incident Management Team and plan. The information we had at that point was pretty minimal: Brands we manufactured, along with several others, were being named by patients associated with the outbreak. We left that night poised to act if we needed to, but felt that it was really not focused on any one company yet, so we chose to monitor and sit tight.
The next day, we had another call with the same group of regulators and producers. The number of patients was growing, and they continued to report consumption of several brands. Now the list of associated spinach products was starting to include more brands co-manufactured by Earthbound Farm, and we knew it was time to take action.
We reconvened our Incident Management Team and came out of that meeting with the decision to initiate a recall. At that time, all we knew was that people who had reported eating the spinach we packed had gotten sick. But regulators were acting very quickly to defend public health, and we took our own action because we felt it was the right thing to do. As soon as we announced that we would perform a voluntary recall, the phone calls with other members of industry stopped, and the investigation turned solely on us. It would be 2 weeks before we had a definitive link between the outbreak and our products. It was then that the outbreak strain of E. coli O157:H7 was found in a bag of spinach we manufactured and which was consumed by a patient connected to the outbreak.
Earthbound Farm’s Food Safety Program Prior to the Outbreak
Prior to the outbreak and recall, our company operated at what I believe was at or above industry best practices for food safety. We maintained a Good Agricultural Practices (GAPs) manual that all our growers agreed to follow, we audited against those standards and had several third-party audits to verify compliance. On the manufacturing side, we maintained a Hazard Analysis and Critical Control Points (HACCP) program that was annually reviewed and updated. We monitored all Critical Control Points and were audited by second- and third-party auditors to verify compliance with our program. We passed every audit with high marks (90 percent or higher).
We also participated in several technical groups where we had the opportunity to be engaged in the latest information and food safety strategies in our industry. In fact, we were part of the advisory group that helped craft the Commodity Specific Guidance Document for Lettuce and Leafy Greens that was put out by the United Produce Association. That document was our industry’s attempt to thoroughly identify any risks from farm to fork, and it provided recommendations for “things to consider” as risks were identified along the continuum. As we developed that document and upon its completion, I reviewed each section and felt we were not only addressing the risks but exceeding recommendations.
It’s important to understand that Earthbound Farm not only packed our own brand of organic salad greens but also packed for other major brands (including Dole), both organic and conventional. All of these major brands regularly came to visit our operation, review our food safety plan and decide whether to allow us to pack for them. All allowed us to pack. In fact, we were often praised for our program and its rigorous controls. This spinach outbreak changed all of that.
The Investigation
Investigators from both FDA and the California Department of Public Health arrived at our facility late afternoon on the 15th and did not leave for at least 3 months. The first order of business: They pulled hundreds of environmental samples in the two manufacturing facilities we operated in 2006. They took dozens of product samples from our retained samples for shelf-life evaluation. No samples showed presence of pathogens.
Next, they turned their attention to our program: documentation and practices. They combed over thousands of pages of documents to see if they could uncover the potential moment in time when things went horribly wrong. Although they may have found some minor errors in paperwork and procedure, they turned up nothing that would have led to the contamination event.
Once they got manufacturing codes from packages that case patients had provided them, they shared them with us so we could perform a traceback. Remember, FDA acted very early in the investigation and did not have any evidence to implicate us other than what was being reported by outbreak patients. Despite the rumors that we did not have good traceability, we were able to provide the investigators with the information they requested within 2 hours of every request. Originally, we were provided with three code dates, which traced back to 12 farms. Then it was narrowed to two codes and eight farms. Eventually, they settled on one code and four farms. Every time we provided new information about implicated farms, FDA sent a troop of investigators out to the farm, sampling and scouring each record for evidence of contamination. They finally focused on one of the four farms implicated in the last code, which led to samples from the ranch (but not the field where the spinach was growing) that matched the outbreak strain (one of the top 10 most common strains of E. coli O157:H7).
The investigators claimed victory with these positives from that one farm; it left me with doubts and skepticism. For example, this happened to be the only one of the four that was in transition to certified organic land (meaning it was farmed organically, but the spinach had to be sold as conventional); all the other farms were conventional. Did they think there was higher risk on this farm? Their sampling regime pulled hundreds of samples from the Paicines Ranch, whereas all the other ranches had dozens pulled. By simple statistics, one would deduce that the more samples you pull, the more you will find. In addition, the positive samples were actually found almost a mile away from the spinach field. What was the vector for contamination from that point to the spinach itself? While you may have heard that wild pigs or groundwater contamination was the source of contamination, these are only theories, with no laboratory evidence to support them.
A Re-Envisioned Food Safety Program
The longer the investigation went on, the surer we were that we did not do anything “wrong” that led to the contamination of the fresh spinach recalled. With this understanding, knowing that we were operating at industry best standards, it was clear that we needed to completely re-envision how food safety is approached in fresh produce: from field selection all the way to putting the product on a truck. We let no area pass free from scrutiny. We knew that to bring a truly revolutionary approach to our food safety program, for a product that has no kill step, we would need to look outside of our industry. We quickly accepted the fact that testing, while not the framework of a robust food safety program, was indeed an important tool to help us validate our upstream processes. We were also hopeful that it would enable us to compile enough data points to perhaps help us identify where our greatest risks were. We were committed to going forward with that strategy rather than waiting for academia or government.
Another important trait of our new program is that we decided not to focus solely on the agent of concern from the outbreak (E. coli O157:H7) but to broaden our scope to include all organisms of concern that it was practical to include in our program. As a result, we not only test for E. coli O157:H7 but also look at the broader family of E. coli bacteria that make humans sick: non-O157 STECs (Shiga toxin-producing E. coli) and Salmonella. Later, we would add Shigella to our raw and finished product testing as the technology of testing improved.
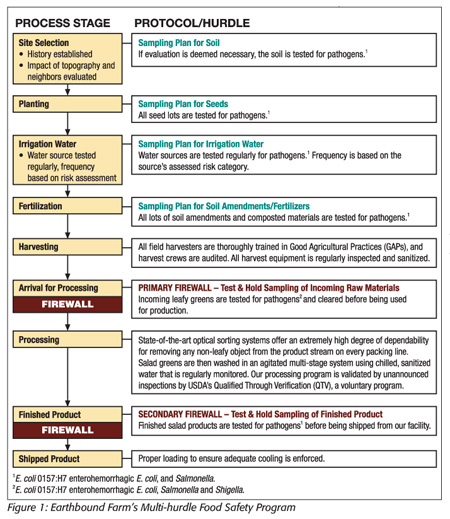 For the program, knowing we had no kill step, we knew we’d need to develop a multi-hurdle process (Figure 1). We started by performing a risk-based hazard analysis of every aspect of production and put controls in place to address the risks identified. Many of the enhancements focused on testing for the presence of the pathogens noted above: testing of farm inputs (e.g., water, seeds, fertilizer, compost), testing of raw material and testing of finished goods. While the enhanced program relies heavily on testing, it should be said that we never looked at testing as the answer to our issue; it was and is just another hurdle in a multi-hurdle approach. It is important to note that we did not throw out our GAP program; we added to it for more strength. We did not change or throw out HACCP; we simply added to the controls around it to build in greater strength. The following is a list of the significant enhancements we made to our program:
For the program, knowing we had no kill step, we knew we’d need to develop a multi-hurdle process (Figure 1). We started by performing a risk-based hazard analysis of every aspect of production and put controls in place to address the risks identified. Many of the enhancements focused on testing for the presence of the pathogens noted above: testing of farm inputs (e.g., water, seeds, fertilizer, compost), testing of raw material and testing of finished goods. While the enhanced program relies heavily on testing, it should be said that we never looked at testing as the answer to our issue; it was and is just another hurdle in a multi-hurdle approach. It is important to note that we did not throw out our GAP program; we added to it for more strength. We did not change or throw out HACCP; we simply added to the controls around it to build in greater strength. The following is a list of the significant enhancements we made to our program:
In the field, we enhanced our program as follows:
• Increased personnel and presence in the field. It is important that we have a relationship with our suppliers that goes beyond a contract and third-party audit. We create the relationship that allows us to know we are getting what we need and rely upon unannounced inspections from Earthbound Farm employees to ensure that promise is delivered.
• Increased our ability to know what is going into the field. We now perform pathogen testing of all inputs going into the field. This would include all seed, soil amendments (fertilizers), water and even transplants. If we have knowledge of what’s coming in, we should be able to reduce the risk to the crop.
• Increased our scrutiny of our approved supplier lists. We go beyond simply approving a grower or a ranch: We break our list down by field lots and release them daily. As a result, we have a very dynamic approved supplier list that is updated daily.
In the plant, we enhanced our program as follows:
• Added a raw product test-and-hold program. As product comes into our facility, everything is placed on hold. We break a field down into four-pallet production units and perform n=60 (150 g total) random sampling. Those samples are tested for E. coli O157:H7, non-O157 STECs, Salmonella and Shigella. We must have negative results for all of these to release the product into processing. We utilize PCR methodology and can obtain a negative in 12 hours and a positive in 16 hours. If evidence of any of the pathogens is found, the entire lot is destroyed.
• Added more robust controls to our processing line. We put metering belts on all of our lines to control the amount of product allocated to each line at any given time, preventing overloading of the wash systems. We added laser sorting to all of our lines for foreign object removal. We increased filtration ability with self-cleaning, in-line filtration (two types per line). We removed thousands of feet of plumbing by reconfiguring our water chilling systems. Shortening our plumbing reduces the opportunity for bacteria to be harbored in our system.
• Increased our ability to narrow the scope of an identified problem. In our outbreak, we had folks consuming product well past the noted expiration date on the package, and we implicated product and growers from an entire shift. We now have very clear language on our packaging and website that instructs the consumer to eat the product up to the expiration date on the package but no longer (we went from “Best if Used By” to “Use By”). We also added a time stamp to every package that allows us to trace the product back to the minute we made it.
• Added a finished goods test-and-hold program. We break our processing day down into 2-hour increments and perform n=60 sampling on each of our 15 pack lines. We test for the same organisms as we do in raw product testing and will not release the product without the negative results. As in raw product testing, any product with evidence of pathogen presence is destroyed.
Today and into the Future
September 2011 marked our 5-year anniversary of the outbreak and recall that changed our paradigm forever. We have generated more than 800,000 data points from our testing. On the raw side of testing, we have recorded more than 650 positives from all growers, all regions and all products. We have seen positives for E. coli O157:H7, non-O157 STECs and Salmonella.
One of the most significant benefits of our program is the fact that with every positive comes a thorough investigation of the field that product came from, in real time! Without a testing program like this, we would never be able to truly get at the root cause of the issue. Because the test is rapid, the product that is cut in the morning is tested that same afternoon with results that evening. If it’s a positive, our team is back in the field the next day looking at the field and precise area from which we cut the product to see if they can find anything that could have led to the contamination. Of the 650+ positives, we have a handful of events wherein we believe we have found the cause and eliminated the risk. Even when we don’t find the root cause, however, we are building a database of information that may be helpful to industry, academia and government. The data we’re tracking include product type, organism, region, soil type, temperature, humidity, inputs, animal activity, etc. Although we may not see its value today, tomorrow it may be the key, so it goes in. We are starting to see some interesting information come out of the data. For example, we definitely have a higher incident rate in the summer months than any other month of the year. We also see a strong correlation between heat index and the positive rate.
When we announced that we were launching a test-and-hold program, the criticism from our peers freely flowed. We heard remarks such as “You can’t test safety into the product,” “You’ll never find it given your sampling plan,” “You’ll see too high of a false positive/negative rate,” “Since the methods weren’t AOAC approved, we don’t respect the result anyway.” I could continue, but you get the point; people were very skeptical and many remain skeptics. However, the conversation is now moving toward the value of the data that we have generated because of the program. Many of those who criticized us early on are asking politely to have a look at the data.
We see positives on the finished goods side as well. Fortunately, we see a much lower incident rate—approximately 10 per year. This shows that our wash system reduces the risk, but it does not eliminate it. We don’t sample every leaf of the inbound raw material, so it is possible to have low-level contamination get past our raw program. When we get a finished goods positive, we tear our process apart to ensure we did not contaminate the product in our process. Over the past 5 years, we have never found this to be true.
Our test-and-hold program is designed to catch gross contamination and serve as a verification of upstream efforts. It is not the answer to food safety; each test is merely its own hurdle in a multi-hurdle approach. We developed our sampling protocols based on our outbreak information and believe we have the strongest program in the industry.
It’s important to say that our program is by no means static. We will continue to improve and evolve the program as the data and science suggests. One of our strategies was that where there was no good data, we would sample aggressively to build the data quickly and get to the answers faster than if we were to wait for government or academia to find the answers for us. Government and academia have very important roles in helping us get to the answers, but they can be hampered by funding and politics. We looked outside our industry for innovative approaches and attacked those areas that were unknown to us with testing. For example, in the sprout industry, seeds are considered the vector for contamination. Baby greens are not far from a sprout, so why wouldn’t we consider them a risk as well? Because of this, we mandated that all seeds going into our fields would be tested. To date, we have seen no positives and are considering dropping the seed testing from our program as we see very low risk coming from that area.
Is our program perfect? No. Is it better than it was prior to September 14, 2006? Yes! Many in our industry continue to ignore the data we have generated and instead are relying on growers to keep contamination out of consumers’ salads. Our product is grown outdoors with so many risks of contamination that I believe it’s impossible to prevent. Someday, we’ll get to where we know what’s creating these pathogens in our environment, but until we do, we have to test. We are signatories to the Leafy Greens Marketing Agreement (LGMA; www.caleafygreens.ca.gov/), as are 99 percent of the California and Arizona lettuce growers. Even with this excellent program in place, we continue to get positives. Even with our enhancements that go beyond LGMA, we get positives.
We must recognize that pathogens exist in our environment. We must recognize that the vectors of contamination are numerous and impossible to keep out of the field. We must recognize that the contamination is likely to occur on the raw material and implement programs that address these realities. We have done so, and I sleep a lot better at night knowing that we have.
Will Daniels is the senior vice president of operations and organic integrity at Earthbound Farm and is responsible for operations (manufacturing, distribution, facilities), food safety, food quality and the company’s organic integrity program. He serves on the board of directors of California Certified Organic Farmers (recently as president), the board of directors for the California Leafy Greens Marketing Agreement and the editorial advisory board for Organic Processing Magazine.
Nationwide Produce Outbreak: A Moment You Never Forget



