Most food processing plants are designed to leverage the maximum number of different products on the fewest pieces of expensive equipment. One challenge for the food industry is changeovers from a product containing allergens to a similar product that does not contain allergens (or the same allergens) produced on the same equipment. Some companies employ precautionary allergen labeling such as “may contain” to all product on the same line or in the same facility; however, this may unnecessarily limit the choices of food-allergic consumers.
Furthermore, the U.S. Food and Drug Administration (FDA) has stated that precautionary labeling cannot be used as a substitute for Good Manufacturing Practices (GMPs), which implies that companies should try to clean between formulations. Other companies follow an allergen validation protocol to demonstrate an effective sanitation changeover and limit the use of precautionary statements to provide the allergic consumer with more food choices.
Historically, companies used a “visually clean standard” for inspections, with allergen checklists to assess the effectiveness of the cleanup.[1] Prior to the development of allergen test methods, companies had no data to verify whether this visual inspection system was effective or adequate to protect the health of food-allergic consumers. A consortium of major food companies sponsored research at the University of Nebraska to develop analytical methods (enzyme-linked immunosorbent assays or ELISAs) to measure allergen residues in food and on equipment. ELISA methods have now been developed for many of the common allergenic foods, and commercial kits are available on the market for most such foods. The University of Nebraska also sponsored research to measure the amount of allergenic food that would be needed to cause even a mild reaction in food-allergic consumers. Using food challenges conducted in allergy clinics has allowed the University of Nebraska to evaluate how much allergenic food can be tolerated without even a mild reaction, and the dosages have been above the limit of detection of the allergen ELISA kits. Now using these same allergen test kits, food manufacturing sanitation procedures can be validated and modified as necessary to produce a changeover product that does not need precautionary labeling. An allergen sanitation validation gives an additional assurance of safety and often supports the adequacy of using the visually clean standard to both plant personnel and allergic consumers.
A Regulatory Perspective
Currently there is no regulatory standard for adequate food allergen sanitation globally. In 1996, FDA issued an Allergy Warning Letter[2] stating that “precautionary labeling should not be used in lieu of adherence to GMPs” and that manufacturers “take all steps necessary to eliminate cross-contamination.” However, no regulatory definitions of these steps or adequate levels have been promulgated to date. The food industry can, however, see similar approaches in the drug industry’s Guide to Inspections of Validations of Cleaning Processes for Pharmaceuticals[3] that FDA published in 1993. The Guide outlines the basics of preventative sanitation programs that a pharmaceutical facility may employ. Similar recommendations were outlined in a document produced by the University of Nebraska’s Food Allergy Research & Resource Program for use by food manufacturers: Components of an Effective Allergen Control Plan: A Framework for Food Processors. This guide also states that “FDA does not intend to set acceptance specifications or methods for determining whether a cleaning process is validated. It is impractical for FDA to do so due to the wide variation in equipment and products used throughout the bulk and finished dosage form industries… Some limits that have been mentioned by industry representatives in the literature or in presentations include analytical detection levels such as 10 ppm…and organoleptic levels such as no visible residue.” FDA food division researchers have studied the adequacy of food allergen sanitation and stated that visual inspection and ELISA allergen kits were the most sensitive methods for detecting the presence of allergen residues compared with ATP swabs.[4, 5]
Choosing Methods
In most cases, companies should use a quantitative ELISA method to validate sanitation that is specific for the allergen to be measured. For example, if the facility is validating the absence of peanuts, it should use a quantitative peanut ELISA kit versus a total protein or an ATP swab due to the increased specificity and sensitivity of the quantitative peanut ELISA kit. Facilities should validate that the peanut can be detected in the peanut-containing food matrix or on the swab prior to use with the non-peanut product (i.e., the positive control that confirms that the ELISA kit is fit for the purpose). Some research has been done on the use of other methods such as polymerase chain reaction and liquid chromatography coupled to mass spectrometry, but these methods are certainly not suitable for routine analysis in the food industry. Thus, the preferred method for food companies remains quantitative allergen-specific ELISAs because they are relatively simple and sufficiently sensitive to ensure that products with no detectable allergen residue by ELISA are safe for food-allergic consumers. Once the initial allergen validation is completed, the use of qualitative ELISA formats such as lateral flow strips and allergen-specific swabs may be employed in a facility as a more cost-effective method of ongoing monitoring. However, it should be noted that historical “visually clean standards” have generally been supported with these allergen test kit validations and therefore may be adequate for ongoing monitoring.
Only a few of the commercial ELISA kits have undergone the extensive AOAC validation procedures. However, in practice, these methods have been successfully applied for a variety of allergen residues in an even wider variety of food matrices. The choice of the most appropriate ELISA kit for a specific use must be carefully made and then evaluated using positive controls to ensure that the method will yield reliable results. For example, milk may be added to foods in a variety of forms, including nonfat dry milk (NFDM), casein, whey, etc. Some of the milk allergen ELISA kits measure total milk, some measure casein and some measure ß-lactoglobulin. Thus, depending on the nature of the milk ingredient, one of these kits may be more appropriate than another. Furthermore, ELISA kits for milk can be calibrated in terms of ppm NFDM, ppm casein or ppm ß-lactoglobulin. It is possible to use a conversion factor to adjust the results obtained as ppm casein into ppm NFDM, but the conversion factors can be debatable. Another confounding factor is that milk and some other allergens may undergo significant conformational and degradation changes during cooking or fermentation to render the allergen residue less detectable by ELISA kits while still potentially causing allergic reactions. To validate this processing effect in a facility, samples should be taken before and after processing to determine the detectability in the specific food matrix.
Sampling Procedures
Never do testing until you have a plan about what to do if you encounter a positive result. Planning for allergen testing requires clear communication and coordination with senior management to hold or destroy product, pending results of the testing. Some companies employ a testing plan termed “safe mode” wherein they run the same allergen product before and after sanitation so that if the swabs indicate inadequate cleaning, they can proceed to ship and have not put the consumer at risk. They can then modify the sanitation procedures prior to the next allergen validation testing. Management should plan to run the formula with the highest percentage of allergen to effectively assess the sanitation. Consideration should also be given to the form of the allergen, as peanut butter may be cleaned differently than peanut granules. Particulate materials can present a sampling challenge in which numerous samples may need to be tested to offer assurance that some sample would contain a particle if any were present. Management should also consider the method of sample shipping, laboratory scheduling and availability that may impact turnaround time of the results. Prior to testing, the swabs (certified allergen-free) from the kit manufacturer must be ordered and available for use (note: other swabs or sponges may actually contain the allergen due to recyclable materials or microbiological media in sponges). Other items to order include disposable gloves, phosphate buffer (certified allergen-free), labels for samples and a shipping container.
Prior to the testing, identify all equipment and/or solutions contacting the allergen product. Prior to the sanitation and before discarding food contact solutions, swab each piece of equipment with a separate swab at multiple sites including crevices, as the size of the surface area is not important for qualitative testing. Recording whether there is visual allergen present on the equipment will allow comparison to test results later to support a visually clean standard. Ensure that all locations that have visual product residue are swabbed prior to sanitation, which can verify that the method can detect the allergen and this can identify equipment that may harbor more allergen. After sanitation, use separate gloves to swab each piece of equipment again and mark as “after sanitation” with the equipment name placed in a sealed plastic bag. These allergen kits are very sensitive and able to detect transfer of peanut from one sample to another, so the use of gloves and separate bags is critical. Other samples to consider include final rinse water from clean-in-place systems, utensils for ingredients or product sampling, gloves or hands from assembly operations and internal surfaces of disassembled equipment. Ensure that all crevices and potential niches are swabbed prior to reassembly when possible. Lab analysis should begin within 48 hours of sampling. Report results as in Table 1.
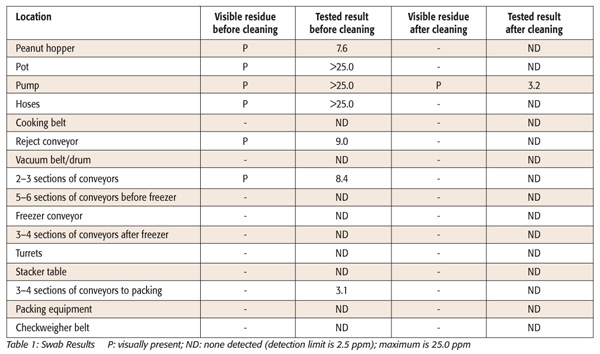
There are basically three options for the next steps: 1) continue operation after sanitation with the exact same allergen product previously sampled so that positive swab results do not impact that product and plan for the next validation; 2) wait for all allergen swab sample results prior to start-up; samples taken after sanitation should be “None Detected” or “BLQ, below the limit of quantitation” prior to start-up of a nonallergen product. If the after-sanitation results are positive, immediately communicate so that the plant-intensive sanitation corrective action can take place, modify Sanitation Standard Operating Procedures (SSOPs) and retest until acceptable results are obtained or allergen-label as appropriate; or 3) if swab results after sanitation are positive at a low level, continue with product testing focusing on the first product after start-up to assess the risk. Positive swab tests do not necessarily mean that the product will be positive, since swabs are so sensitive, the results are not quantitative and the surface areas and contact times are variable.
After successfully completing the swab validation testing, proceed to product sampling. Manufacture the product through the same equipment that was previously swabbed and validated for food contact surfaces. Mark or indicate in some manner the first product produced through the line and sample adequately. Ship product to the lab in the temperature state as labeled (e.g., Keep Refrigerated or Frozen). Product from the entire sample lot must be held awaiting the lab results, destroyed or labeled as containing the allergen. Successful allergen validation results should be summarized in a report including the version of SSOPs that was validated, which products and lines were validated and the lab results. Some companies may employ a “push-through” method where the first 5 minutes (or more) of product after changeover is always discarded. However, this method should be validated sufficiently to ensure that the time or volume discarded is adequate. Quantitative ELISAs can also be used for this purpose. Use push through for 5 minutes, then 10 minutes, then 15 minutes, and sample at each time followed by testing. Once you get to the “None Detected” or “BLQ” result, then the validation of the push-through procedure is completed upon adequate documentation of this approach. Typically, dust in the air from dry products with adjacent lines does not accumulate to a significant level as to cause the nonallergen product to test positive. This may be facility dependent, however, and should be verified. Dust can be an issue when the adjacent line is idle, but swab testing will reveal if sufficient allergen residues have been deposited on the idle line to require sanitation prior to start-up.
This comprehensive sanitation validation testing can be used initially upon start-up of a new line to assess the need for labeling and the risk for the facility. Ongoing monitoring may include only visual inspection if the validation results support that approach, revalidation periodically or if the SSOP changes significantly, or routine monitoring in the case of packaging claims such as peanut-free or when consistent sanitation is especially difficult. Periodic revalidation can establish a history to confirm that SSOPs are consistently applied over time.
Consumer Protection with Thresholds
The food industry currently uses allergen ELISA kits to validate that the sanitation after allergen products is effective to eliminate the allergen from nonallergen-labeled products. In support of this approach, one can compare the detection limit for the peanut ELISA kits at 2.5 ppm (mg per kg of food) to the published threshold studies wherein the first dose of peanut to elicit a mild reaction was 0.4 mg whole peanut. Since threshold doses are expressed in amounts (i.e., mg) and ELISA kits measure concentrations (e.g., 2.5 ppm), the amount of food eaten that contains a particular concentration becomes critical to the evaluation of the risk. So if 0.4 mg whole peanut were contained in a 50-gram serving of a food, it would equate to 8 ppm. Using a probabilistic risk assessment model with 450 peanut-allergic patients, the lowest elicitation dose of 0.4 mg was encountered with four of the 450 subjects.[6] Thus, a food containing 8 ppm peanut would put a rather low percentage of peanut-allergic consumers at risk of a mild reaction if they ate 50 g of the product. Of course, the risk would increase if one of these highly sensitive peanut-allergic consumers ingested an even larger amount of that food. But if a food is tested to have less than 2.5 ppm peanut, then the food would be predicted to be safe for the vast majority of peanut-allergic individuals even with rather high consumption levels.
In summary, allergen validation studies provide a valuable tool to protect allergic consumers from reactions and to minimize unnecessary precautionary labeling statements to provide allergic consumers with more food choices. These allergen validation studies also provide the food industry and regulatory bodies with scientific support for the visually clean standard.
Tracie Sheehan, Ph.D., is president of TGS Quality Consulting LLC.
Joseph Baumert, Ph.D., is assistant professor in the department of food science & technology and co-director of the Food Allergy Research & Resource Program at the University of Nebraska–Lincoln. He can be reached at jbaumert2@unl.edu.
Steve Taylor, Ph.D., is professor in the department of food science & technology and co-director of the Food Allergy Research & Resource Program at the University of Nebraska–Lincoln. He can be reached at staylor2@unl.edu.
References
1. Deibel, K., T. Trautman, T. DeBoom, W. Sveum, G. Dunaif, V. Scott and D. Bernard. 1997. A comprehensive approach to reducing the risk of allergens in foods. J Food Prot 60(4):436–41.
2. FDA. 1996. Allergy Warning Letter. NOTICE TO MANUFACTURERS Label Declaration of Allergenic Substances in Foods.
3. FDA. 1993. Guide to Inspections of Validation of Cleaning Processes for Pharmaceuticals. Division of Investigations, Office of Regional Operations, Office of Regulatory Affairs.
4. www.charm.com/resource/file/186.
5. Jackson, L.S., F.M. Al-Taher, M. Moorman, J.W. DeVries, R. Tippett, K.M. Swanson, T.J. Fu, R. Salter, G. Dunaif, S. Estes, S. Albillos and S.M. Gendel. 2008. Cleaning and other control and validation strategies to prevent allergen cross-contact in food-processing operations. J Food Prot 71(2):445–58.
6. Taylor, S.L., D.A. Moneret-Vautrin, R.W.R. Crevel, D. Sheffield, M. Morisset, P. Dumont, B.C. Remington and J.L. Baumert. 2010. Threshold dose for peanut: Risk characterization based upon diagnostic oral challenges of a series of 286 peanut-allergic individuals. Food Chem Toxicol 48:814–9.
SIDEBAR
Allergen Testing Validation
Allergic consumers rely on having accurate and complete food labels to avoid potentially life-threatening reactions. Food manufacturers use many allergen control tools to help them provide those labels, including commercial allergen test kits. While kit makers have been highly successful in producing kits in multiple formats that are robust, simple and rapid, in the end, the accuracy and reliability of the results obtained depends on how the kits are used. A food manufacturer using allergen test kits must evaluate factors such as whether the target of detection is appropriate (whole foods or specific proteins, native or denatured, etc.), the design of appropriate sampling plans (where and when to sample, number of replicates, etc.) and to how to validate a test for a specific application. A well-designed sampling plan may be particularly important in situations where cross-contact occurs sporadically or where the allergen is particulate (e.g., chopped nuts). Foods are complex mixtures, and the properties of each mixture change during processing and storage. Method validation helps ensure that the results obtained with a specific test kit in a specific situation accurately reflect the properties of the food or ingredient being tested. Multi-lab collaborative studies provide assurance that kits can perform at a defined level of reliability, but validation of the complete assay process in each specific situation makes it possible to show that this level of performance and reliability has actually been achieved. Validation can be used to show that an assay process (starting with the sampling technique and finishing with the final assay readout) can actually detect the allergen of interest if it is present and (for quantitative assays) that the measured allergen concentration accurately reflects the amount in the food or ingredient. It is important to consider using actual food samples in validation studies to account for matrix effects that depend on specific food formulations, the effect of processing on the analytic target and on extraction efficacy, and potential positive or negative interferences. Spiking of extracts with known material and the use of test kit controls are important for verifying the day-to-day use of an assay, but a more complete analysis can help validate an assay before it is used routinely.
—Steven M. Gendel Food Allergen Coordinator Food and Drug Administration, Center for Food Safety and Applied Nutrition




